Introduction
Melissodes Latreille is a common genus of wild bees from the tribe Eucerini occurring in the New World (Laberge, 1956a; Laberge, 1956b; Laberge, 1961; Wright et al., 2020). As of now, a total of 129 species have been described in this genus making it the second largest in the Eucerini tribe (Wright et al., 2020). 26 of these species have been documented in Oregon (see table 1) and 8 have known ranges that overlap portions of Oregon (see table 2). Melissodes are known to be important pollinators in crops such as cotton (LaBerge, 1956), alfalfa (LaBerge, 1956), muskmelons (Winfree, et al., 2007), watermelon (Campbell et al., 2018; Winfree, et al., 2007), canola (O'Brien & Arathi, 2018), and coffee (Ngo et al., 2013), but most chiefly so in sunflowers (Parker et al., 1981). Parker (1981) documented that a species of Melissodes (M. agilis) is a more proficient pollinator in sunflowers than that of the western honeybee. Although their importance has been expressed in agricultural settings (Parker, 1981) and likely in wild ecosystems due to their abundance (Laberge, 1956a), identification of Melissodes beyond genus is often scarce due to the subtle characters that differentiate between species. The genus was last revised in a three-part series by W. E. Laberge (1956a; 1956b; 1961) as well as a later correction to the M. (Callimelissodes) subgeneric key (Laberge, 1963), in which detailed species-specific treatments, generic and subgeneric keys, and distributions were first presented, representing a cornerstone for current-day Melissodes knowledge. However, the keys presented in these revisions can be, at times, slightly ambiguous and overwhelming due to the sheer number of species and intraspecific variation. As a genus, Melissodes are widespread and range over the entirety of the New World, but species differ largely in what areas they occupy (Laberge, 1956a; Laberge, 1956b; Laberge, 1961; Wright et al., 2020). Many species are restricted to South America and Central America, which, when trying to identify North American species using a key designed to include all 129 species, drastically inflates the amount of information needed to identify an individual. The same can be said for species whose ranges are restricted to specific portions of North America that differ from the location in which a specific individual was found. Herein, a key developed from that of Laberge (1956a; 1956b; 1961; 1963) using the same structure and traits but incorporating subgeneric splits and only including species who occur, or are likely to occur, in Oregon is presented. Although using a regional key such as this may not yield new state records for Oregon, apart from those whose ranges overlap this area and are included in the key, it may assist in ease of identification beyond genus allowing for species-level studies and less chances for drastic misidentification due to individual variation. Along with this, species descriptions and comparisons, derived from that of Laberge, (1956a; 1956b; 1961) are given to further aid in identification.
Table 1. A table of Melissodes species with their citations to the resource which documents their occurance in Oregon.
| Species | Citations |
|---|---|
| Melissodes agilis | (Laberge, 1961; Ikerd, 2019; Johnson, 2020; Best et al., 2022a; Texas A&M University Insect Collection, 2023; Best, 2026; Illinois Natural History Survey, 2026; The International Barcode of Life Consortium, 2026) |
| Melissodes ablusus | (DeBano et al.) |
| Melissodes bimatris | (Laberge, 1961; Ikerd, 2019; Johnson, 2020; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; MT James Entomological Collection, Washington State University, 2026; The International Barcode of Life Consortium, 2026) |
| Melissodes clarkiae | (Laberge, 1961; Best et al., 2022a; Bentley & Osborn, 2026; Best, 2026; The International Barcode of Life Consortium, 2026) |
| Melissodes communis | (Laberge, 1956a; Ikerd, 2019; Johnson, 2020; Best et al., 2022a; Ikerd & Engler, 2023; Bentley & Osborn, 2026; Best, 2026; The International Barcode of Life Consortium, 2026) |
| Melissodes dagosus | (Laberge, 1956b; Ikerd, 2019; Ikerd & Engler, 2023; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; The International Barcode of Life Consortium, 2026) |
| Melissodes glenwoodensis | (Laberge, 1961; Ikerd, 2019; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; The International Barcode of Life Consortium, 2026) |
| Melissodes lupinus | (Laberge, 1961; Ikerd, 2019; Johnson, 2020; Graham et al., 2021; Best et al., 2022a; Ikerd & Engler, 2023; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026; The International Barcode of Life Consortium, 2026) |
| Melissodes lustrus | (Laberge, 1961; Ikerd, 2019; Johnson, 2020; Best et al., 2022a; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026) |
| Melissodes lutulentus | (Laberge, 1961; Ikerd, 2019; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026) |
| Melissodes menuachus | (Laberge, 1961; Ikerd, 2019; Illinois Natural History Survey, 2026) |
| Melissodes metenuus | (Laberge, 1961; Ikerd, 2019; Johnson, 2020; Graham et al., 2021; Best et al., 2022a; Ikerd & Engler, 2023; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026) |
| Melissodes microstictus | (Laberge, 1961; Johnson, 2020; Best et al., 2022a; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026; MT James Entomological Collection, Washington State University, 2026; The International Barcode of Life Consortium, 2026) |
| Melissodes pallidisignatus | (Laberge, 1961; Ikerd, 2019; Johnson, 2020; Graham et al., 2021; Best et al., 2022a; Texas A&M University Insect Collection, 2023; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026) |
| Melissodes paululus | (Ikerd, 2019) Note, Laberge (1961) had Oregon within M. paululus’ range, just none had been collected. |
| Melissodes plumosus | (Laberge, 1961; Ikerd, 2019; The International Barcode of Life Consortium, 2026) |
| Melissodes pullatellus | (Laberge, 1961; Bentley & Osborn, 2026; Grinter et al., 2026) |
| Melissodes rivalis | (Laberge, 1956b; Ikerd, 2019; Johnson, 2020; Best et al., 2022a; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026; Motz, 2026; The International Barcode of Life Consortium, 2026) |
| Melissodes robustior | (Laberge, 1961; Ikerd, 2019; Best et al., 2022a; Bentley & Osborn, 2026; Best, 2026; Johnson, 2026; MT James Entomological Collection, Washington State University, 2026; The International Barcode of Life Consortium, 2026) |
| Melissodes saponellus | (Laberge, 1961; The International Barcode of Life Consortium, 2026) |
| Melissodes semilupinus | (Laberge, 1961; Ikerd, 2019; Johnson, 2020; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; The International Barcode of Life Consortium, 2026) |
| Melissodes stearnsi | (Laberge, 1961; Bentley & Osborn, 2026; The International Barcode of Life Consortium, 2026) |
| Melissodes subagilis | (Laberge, 1961; Ikerd, 2019; Johnson, 2020; Illinois Natural History Survey, 2026) |
| Melissodes tepidus | (Laberge, 1956a; Ikerd, 2019; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026) |
| Melissodes tristis | (Ikerd, 2019) |
| Melissodes utahensis | (The International Barcode of Life Consortium, 2026) |
Table 2. A table of Melissodes species with their citations to the resource which documents their ranges which overlap Oregon (i.e. species with a likely occurance in Oregon that have yet to be documented).
| Species | Citations |
|---|---|
| Melissodes bicoloratus | (Ikerd & Engler, 2023) |
| Melissodes compositus | (Ikerd, 2019) |
| Melissodes druriellus | (Ikerd & Engler, 2023) |
| Melissodes grindeliae | (Laberge, 1961) |
| Melissodes minusculus | (Ikerd, 2019) |
| Melissodes nigracauda | (Washington State Department of Agriculture, 2025; MT James Entomological Collection, Washington State University, 2026). Likely the same specimen. |
| Melissodes verbesinarum | (Laberge, 1961; Ikerd, 2019; MT James Entomological Collection, Washington State University, 2026) |
| Melissodes vernalis | (Laberge, 1961) |
How to use this resource
This field guide is designed to aid in identifying Melissodes species in Oregon. This includes comparisons of similar genera such as Eucera and Diadasia, which have overlapping ranges and similar morphologies, comparisons of similar species within the genus, and regional keys. Ranges have been determined using historic taxonomic literature in tandem with GBIF occurrence records (see tables 1 & 2). This data was also used to evaluate distributions, phenology, and host-plant relationships. Each species has a full treatment used to assist in identification, locality, morphology, and more (species treatments are outlined below).
Species treatments
Each species description consists of what follows:
Morphological descriptions, particularly of key diagnostic features with accompanying photos of the feature; distribution maps plotted in Leaflet using OpenStreetMap (OSM)
derived from authoritative taxonomic literature (Laberge, 1956a; 1956b; 1961) and recent sampling from GBIF occurrence records to produce an up-to-date range; phenological graphs created using data from
GBIF occurrence records; flower records and/or preference if lists are too extensive; when applicable, notes concerning taxonomic history and/or distinctive identifiers; two
"Feild Marks" and "Similar species" sections (one per sex given the sexual diamorphism) unless stated otherwise; a
similar naming convention to that used by the North American Native Bee Collaborative (2020) will be adapted for this guide, that being at the beginning of each species treatment,
a group-name is given to species of similar morphologies (if a group-name is absent, then the species does not specifically resemble another and instead, the word “distinct”
will take the place of group-name). For most species, as there is insufficient data, phenological graphs will consist of two lines, one representing phenology based on Oregon
captures, and the other based on captures across each species respective range. Each species "Field Markers" section is based on the information given about both sexes from the genus treatment;
if not familiar with Melissodes as a genus, reading the genus treatment is recomended before subsequent species treatments. Morphological descriptions for each species are derived from that of Laberge (1956a; 1956b; 1961).
Precursing each of the species treatments, the genus will be treated. This will not include a
group-name as no other genera are described within this guide; similar genera will be listed instead.
Note: according to The International Barcode of Life Consortium (2026), M. micheneri, M. personatellus, and M. utahensis have been found in Oregon (once per species except the latter). However, the two former species
are omitted in this field guide above as the only identifying features are DNA barcodes and their current known ranges (without these datapoints) do not overlap or include Oregon. M. utahensis, the sole exception to this
rule, is included in Table 1 as three records, all from largely differing areas of Oregon, have been collected and for the reasons outlined in its species treatment. Although M. micheneri and M. personatellus may have been
collected in Oregon, the lack of sufficient data in regards to their identification methods, as well as Oregon being vastly out of their range, makes it challenging to be certain (see more in the M. utahensis species treatment).
Reference Images
As can be determined below, subtle differences are used in delimiting species of Melissodes. To describe these delimiting characters, terms have been applied to specific anatomical features and structures. Below is a list of Melissodes images with labeled anatomical features, the terms used to describe these features will be used throughout the guide; definitions for these terms are also in the glossary with parenthesized use cases.
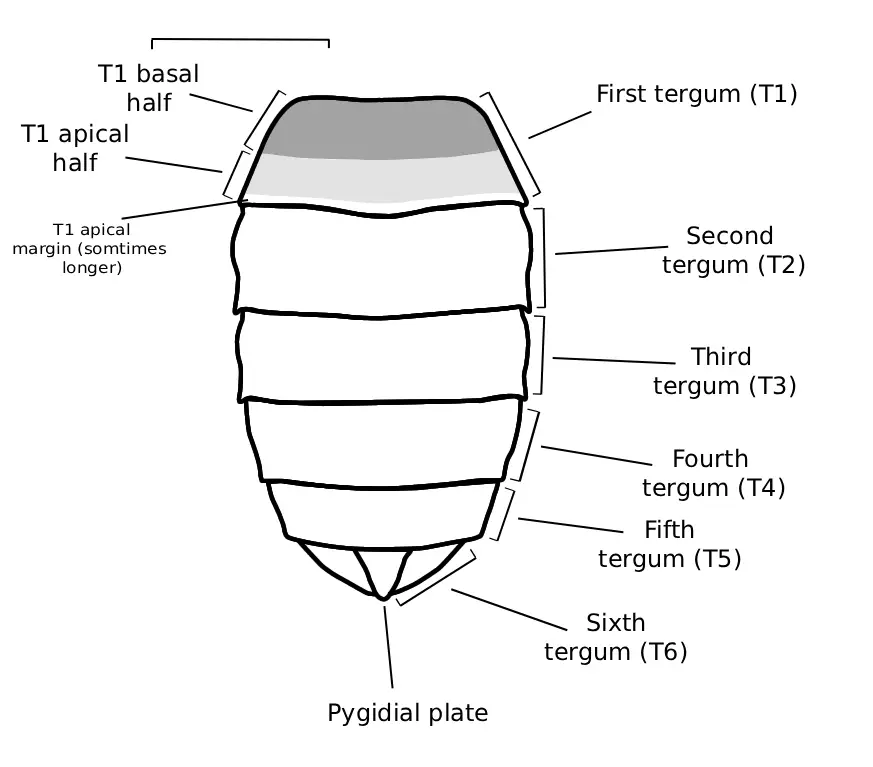
Fig. 1. Fig. 1. An illustration of the dorsal metasomal structure of a female Melissodes that has been redrawn and relabeled from that of Laberge (1956a). Only the first tergum is regionally labeled, but T2-T6 follow the same patterns (male Melissodes have 7 terga). Originally presented as "Fig. 20. Diagram of metasoma (approximately X 11) of M. communis showing areas of vestiture. Regional terms are on the left and descriptive terms referring to vestiture are on the right. Similar types of shading indicate similar types of pilosity."

Fig. 2. Diagrams representing the facial anatomy of a Melissodes. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 3. Diagrams representing the thoracic anatomy of a Melissodes. Photo credits: Christopher Wilson (All Rights Reserved).
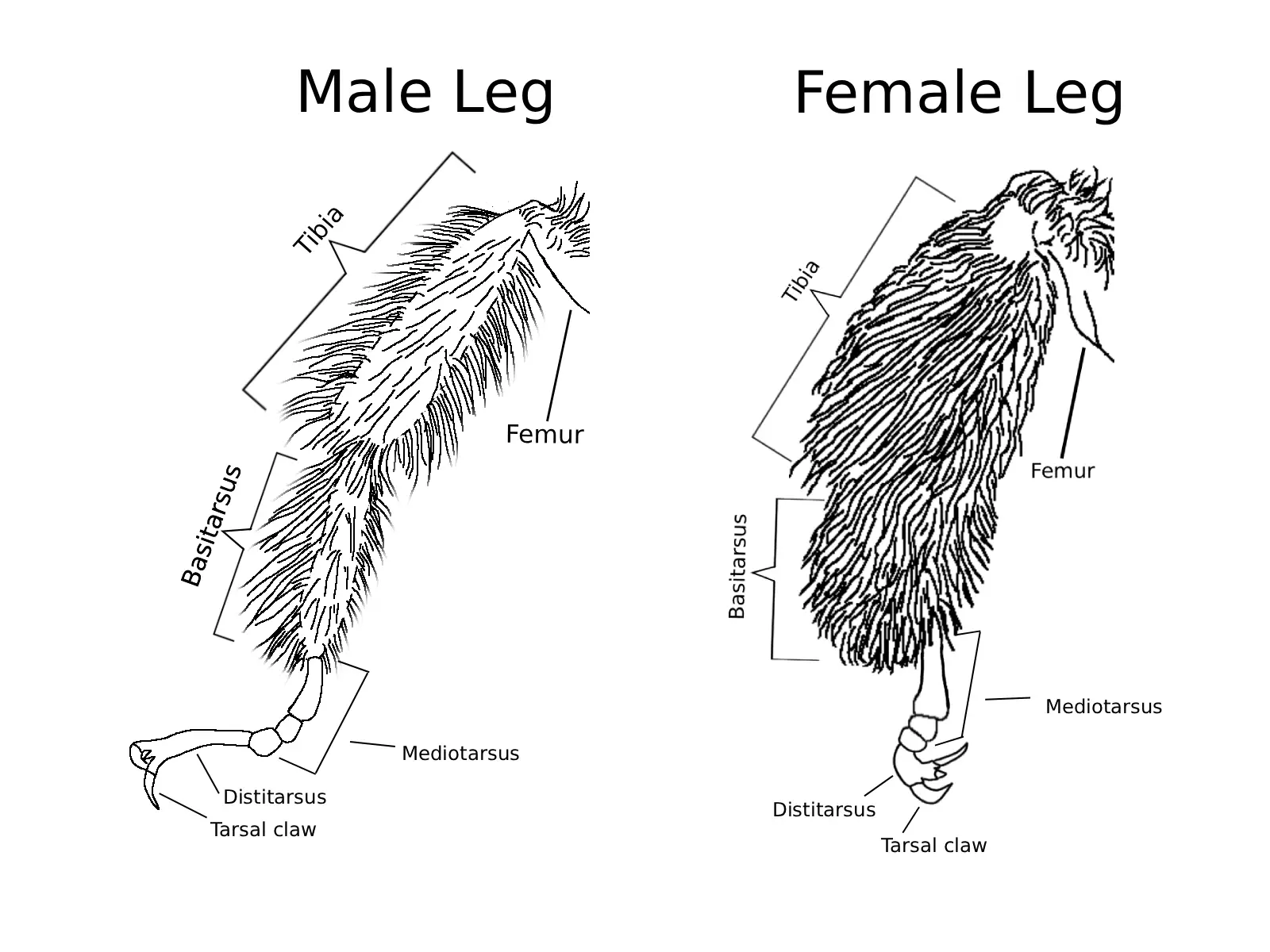
Fig. 4. Diagrams representing the leg anatomy of a Melissodes. One diagram per sex is given due to their sexual diamorphism.
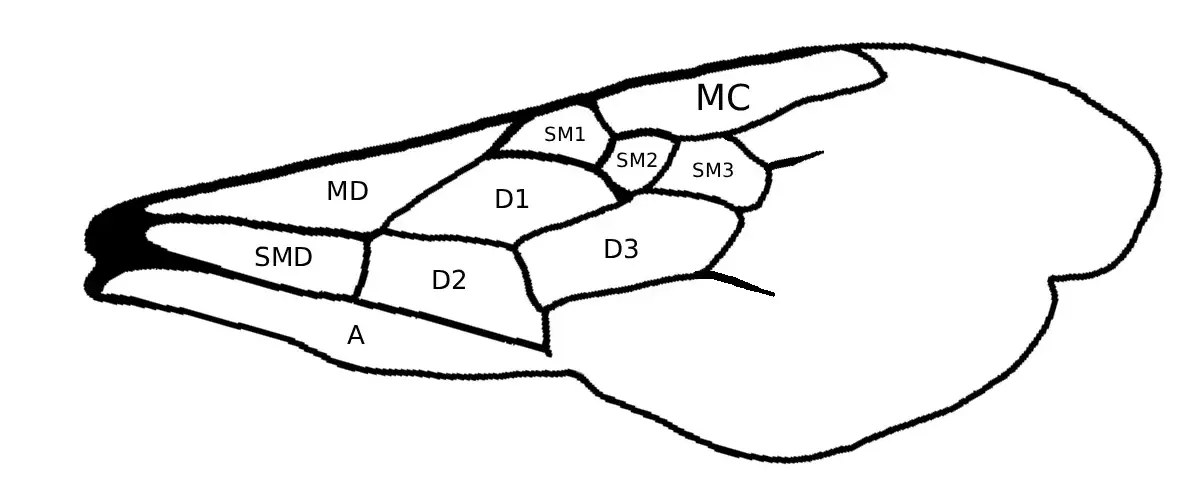
Fig. 5. Wings of a Melissodes (cells shown by capital letters). Cells: A, anal; D, discoidal: MC, marginal; MD, median; SM, submarginal; SMD, submedian.

Fig. 6. Labeled images illustrating the ventral structure of a Male Melissodes' metasoma. The sterna are labeled S1-S6; note that this is a male, females have only 5 sterna.
Identification
Because of their sexual diamorphism, two keys for the species of the genus Melissodes in Oregon are listed below (male and female keys). These keys are developed from those of Laberge (1956a; 1956b; 1961; 1963), but include subgeneric splits and only Melissodes that are known to inhabit, or have ranges that overlap, Oregon. If a couplet regarding a certain character seems ambiguous, check morphologically similar species in their respective treatments.
Key to the bees of the genus Melissodes in Oregon
Males













Females
Melissodes

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Animalia
Arthropoda
Insecta
Hymenoptera
Apidae
Eucerini
Melissodes
Melissodes are perhaps one of the most commonly found wild Apid bee genera in Oregon apart from Bombus and Ceratina (Best et al., 2021b; 2022b). In general, the genus is comprised of very setaceous, robust, and medium sized bees with the largest Oregonian Melissodes (M. glenwoodensis) in length, measuring 11-14 mm. in the female sex and 10-15 mm. in the male sex (Laberge, 1961; for reference, Apis melifera measures about 13.2 mm. on average; see DeGrandi-Hoffman et al., 2004). Males and females can be readily distinguished from one another by the increadibly long antennal length of the males and the scopae on the hind legs and robustness of the females. Melissodes are ground-nesting bees (Laberge, 1956a; Laberge, 1956b; Laberge, 1961; Michener, 2007) that are most active during the fall months, with peak activity occurring in August and July (see, in part, Fig. 12). This genus is widespread across the entirety of Oregon, but each species differs slightly in range (see species treatments). Overall, Melissodes as a genus are oligolects of Asteracae, and often nest near large quantities of these flowers (Parker et al., 1981), which seems reasonable as its been reported that some species home ranges are short, likely around 14-16 meters (Foy, 2025).
Field Markers
♂ Long antenna, often around more than two-thirds of their total body length and almost no malar margin. Very setaceous, and often having a yellow or pale colored clypeus
(although there are exceptions; see M. tristis). The wings have 3 submarginal cells and the eyes tend to have a green color, though not always. The first flagellar
segment is usually incredibly short compared to the rest of the flagellum (except in M. dagosus and sometimes M. rivalis) and the genal area is usually somewhat
narrow laterally (though not always).
♀ Very setaceous and robust with large scopae on the hind legs and almost no malar margin. The wings have 3 submarginal cells and the clypeus is often not very
protuberant (except for M. rivalis). The genal area is usually somewhat narrow laterally (not always) and the vertex is somewhat flat. The tegulae are narrowed anteriorly
and the metasoma is often shorter and distally rounded (not wasp-like).
Similar Genera
Although they are a distinctive genus, Melissodes can superficially resemble other genera, prime examples of which are Eucera and Diadasia. Male Melissodes can reliably be distinguished from that of a male Diadasia by their long antennae and anteriorly narrowed tegulae (Fig. 7), although females can bear more of a resemblance due to their robustness and scopal hair location. Female Melissodes can be differentiated from that of female Diadasia due to the anteriorly narrowed tegulae and flatter vertex outlines when looking in facial view (Michener, 2007; Laberge, 1957; see Fig. 8). Distinguishing between Melissodes and Eucera can be a bit trickier as both genera reside in the same tribe (Eucerini) and therefore males cannot be separated by antennae length alone and females share similar features. The most common and most relioable way to separate both sexes of Melissodes from Eucera is with tegular shape, that being Melissodes tegulae are narrowed anteriorly and Eucera tegulae aren’t (Michener, 2007; Laberge, 1957; see Fig. 9). Males of Eucera often have entirely black antennae with no ventral red markings (not always the case, somes species have red colorations ventrally), highly distinct flagellar segmentation, and wider scapes in contrast to the often red ventral to dorsal antennal markings, less distinct flagellar segmentation, and narrower scapes of Melissodes (Fig. 10; male Melissodes microstictus have entirely black antennae as well, except for a ventral pale macula, but the flagellar segments are less distinct, the genal area is smaller, and the clypeus is less protuberant than that of a male Eucera). Both sexes of Eucera also tend to have larger genal area to eye ratios (not always; Fig. 11), and earlier phenological activity than that of Melissodes (Fig. 12).

Fig. 7. A comparison of the males of Melissodes and Diadasia showing the differances in antennal length and structure of the tegulae (Melissodes on the left, Diadasia on the right). Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 8. A comparison of the females of Melissodes and Diadasia showing the differances in vertex and tegulae structure (Melissodes on the left, Diadasia on the right). Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 9. A comparison of both sexes of Melissodes and Eucera showing the differance in tegulae structure (females above, males blow; Melissodes on the left, Eucera on the right). Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 10. A comparison of males of Melissodes and Eucera showing the differance in antennal coloration and structure (Melissodes on the left, Eucera on the right). Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 11. A comparison of both sexes of Melissodes and Eucera showing the differance in eye to genal area ratios (females above, males blow; Melissodes on the left, Eucera on the right). Photo credits: Christopher Wilson (All Rights Reserved).
Fig. 12. A graph representing the phenology of the genus Melissodes and Eucera in Oregon (blue represents Melissodes and red represents Eucera). The x value is the month, and the y value is the percentage of documented observations occurring in that month. Note the peak activity of Melissodes occurs in fall, much later than Eucera. Melissodes data compiled from (Ikerd, 2019; Johnson, 2020; Best et al., 2021a; Graham et al., 2021; Best et al., 2022a; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; Droege & Maffei, 2025; Gibbs, 2025; Bentley & Osborn, 2026; Best, 2026; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Frost Entomological Museum, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Motz, 2026; MT James Entomological Collection, Washington State University, 2026; The International Barcode of Life Consortium, 2026) and Eucera data compiled from (Ikerd, 2019; Best et al., 2021a; Graham et al., 2021; Best et al., 2022a; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; VanDyk et al., 2023; Gibbs, 2025; Grant et al., 2025; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Orrell & Informatics, 2026a; San Diego Natural History Museum, 2026; The International Barcode of Life Consortium, 2026; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 (see "Dataset Licenses").
Fig. 13. A graph showing the number of Melissodes documentation per species representing the most to least common. The x value is the species, and the y value is the number of documented observations. Data derived from ((Ikerd, 2019; Johnson, 2020; Best et al., 2021a; Graham et al., 2021; Best et al., 2022a; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; Droege & Maffei, 2025; Gibbs, 2025; Bentley & Osborn, 2026; Best, 2026; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Frost Entomological Museum, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Motz, 2026; MT James Entomological Collection, Washington State University, 2026; The International Barcode of Life Consortium, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 (see "Dataset Licenses").
Melissodes agilis

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes agilis
Melissodes agilis, the agile long-horned bee, is known to be one of the most common and widespread North American Melissodes (Laberge, 1961). Unexpectedly, in Oregon, this species ranks fifth most common with a total of 69 publicly documented state records scattered across the majority of the landscape (Fig. 13). Although not a distinctive species in general, especially in the female sex, the only resemblance M. agilis directly bears is to M. trinodis which only occurs in eastern portions of the United States and Canada (Laberge, 1961), and for this reason is labeled distinct. Females are known to nest in and around large quantities of Asteraceae, particularly Helianthus, with daily activity beginning around 6:30 AM to 7:45 AM (likely dependent on location and temperature) and height of activity often in earlier parts of the day (Parker et al., 1981). Parker et al. (1981) found at 9:00 AM, there were twice the amount of females foraging than at 11:00 AM, and at 1:00 PM there were about one-third the amount of females foraging than at 11:00 AM. It seems that most females stay in their nests during the afternoon (Parker, F. D et al., 1981). Males are distinctive in that the galeae are dulled above due to fine reticular shagreening and the wings are yellow to red; the hairs on the head and thorax are minimally pale ochraceous and often pale rusty-red; the mandibular bases have a yellow macula and the labrum has a large mediobasal pale macula (at least equal to, and usually larger than, one-third of the labrum); the clypeus is entirely light excluding the dark spots denoting the tentorial depressions or the anterior notches and margin; the first flagellar segment’s maximum length is less than, or equal to, one-third of the third segment’s minimum length; the penultimate segment is about three times as long (maximum) as it is wide (minimum). Females are distinctive in that the first tergum’s apical impunctate area is medially longer than one-half of the punctate basal area; the third tergum usually doesn't have any dark apical hairs in the apical area; The mesoscutum doesn’t have a dark brown to black patch of posteromedial hairs, or if so, then very few dark hairs are present; the clypeus often protrudes beyond the eye by about one-half of an eye’s width in profile; the apical area of the third tergum bears pale hairs that don’t entirely obscure the surface, differing from those of the distal band in being more erect and less plumose. This species was historically known to be active between the months of April to November with peak activity occurring in July, August, and September (Laberge, 1961). Newer data seem to follow this pattern, however, August generally seems to be the height of activity with the last month of significant records being September (Fig. 19).
Field Markers
♂ Mostly a pale bee but vestiture ranges between light near desaturated gray to fairly vibrant
ochraceous. The eyes are green to grey or greyish blue and the wing veins are yellow. The base of
the mandibles are yellow to cream colored and the labrum is white or cream-colored apart from the
apical margin, which is brown (very rarely is the labrum all brown). The flagellum is red to yellow
on its underside and dark red to brown on top and the apical margins of the terga are hyaline and
colorless to yellow (in some eastern individuals, they can be a translucent brown, though this is
unlikely in Oregon). The first flagellar segment’s minimum length is about one-sixth to one-seventh
the maximum length of the second segment, and the penultimate segment is about three times as long
(maximum) as it is wide (minimum). All flagellar segments do not have longitudinal lateral depressions.
The first flagellar segment’s maximum length is less than, or equal to, one-third of the third
segment’s minimum length. The galeae are dulled above due to fine reticular shagreening and the
wings are yellow to red. Facial and genal area hairs are pale ochraceous to yellow and the vertex
is yellow to bright rufescent (the vertex usually has no brown hairs). The sides of the thorax are
pale ochraceous, and the top is ochraceous to bright rufescent. The first tergum’s basal four-fifths
to five-sixths has long pale hairs that are medially long enough to reach apical margin, although
there are too few to completely obscure the apical area. The apical area is glabrous or has very sparse,
small, appressed brown to yellow hairs basally and laterally. The second tergum’s white to yellow
basal pubescent band is long and the interband zone has long, abundant, suberect, relatively simple,
pale hairs. The distal pale band of the second tergum is yellow to white, often as long or longer than
the apical area medially, and usually it’s narrowly interrupted medially. The apical area of the second
tergum has relatively simple, appressed to subappressed, white to yellow hairs that obscure the surface,
be it not entirely. The third tergum is similar to the second but the basal tomentum is brown and the
hairs on the interband zone are usually scattered with pale pubescence, the distal pale band isn’t
interrupted, and the apical area is shorter. The fourth tergum is similar to the third, but the pale
distal pubescent band is closer to the apical margin. The fifth tergum is similar to the fourth, but
the distal hair band is closer to the apical margin. Terga 6 and 7 have long, white to ochracous,
sometimes yellow, hairs.
♀ The overall vestiture is ochraceous to yellow, usually very few dark hairs if any. The eyes are a
gray to bluish gray and rarely greenish or black with violet reflection and the wing veins are red to
reddish brown. The underside of flagellar segments 3-10 and often the apex of the second are rufescent.
The first tergum’s apical impunctate area is medially longer than one-half of the punctate basal
area. The third tergum usually doesn't have any dark apical hairs in the apical area and instead
bears pale hairs that differ from those in the distal pale band by being more erect and less plumose,
but don’t obscure the surface. The mesoscutum doesn’t have a dark brown to black patch of posteromedial
hairs, or if so, then very few dark hairs are present and the clypeus often protrudes beyond the eye
by about one-half of an eye’s width in profile. Facial and genal area hairs are pale ochraceous to
yellow and the vertex is yellow to bright rufescent (the vertex will sometimes have brown hairs, if
so, they are never abundant). The sides of the thorax are pale ochraceous, and the top is ochraceous
to bright rufescent. The first tergum has long, ochraceous to yellow or slightly rufescent hairs
basally and laterally, except for the apical area which is glabrous or has very sparse, small,
appressed brown to yellow hairs basally and laterally. The second tergum’s white to yellow basal
pubescent band is long and the interband zone has short, appressed to subappressed, relatively
simple, white to pale hairs. The distal pale band of the second tergum is yellow to white, broad
laterally (very rarely reaching apical margin) to narrow medially, and usually it’s narrowly
interrupted medially. The apical area of the second tergum has relatively simple, appressed to
subappressed, white to yellow hairs that obscure the surface, be it not entirely. The third tergum
is similar to the second but the basal tomentum is dark brown as well as the hairs on the interband
zone, the distal pale band isn’t interrupted, and the apical area is shorter. The fourth tergum is
similar to the third, but the pale distal pubescent band reaches the apex across the entire tergum
and occasionally has a minute apicomedial area of brown hairs. Terga 5 and 6 are dark brown except
for white to yellow tufts of hair at extreme sides.
Similar Species
Although distinctive in Oregon, M. agilis can bear somewhat of a resemblance to a few other M. (Eumelissodes) within the same region.
♂ M. agilis superficially resembles a number of other Oregonian Melissodes males, the first of which, as the name implies, is M. subagilis. This species quite starkly differs from M. agilis in the mandibles and labrum having no maculations and the galeae which are rather unshagreened and shiny in the apical half (only delicately shagreened if shagreened at all; sometimes densely tessellated). Another species M. agilis can somewhat resemble is M. menuachus. This species differs from M. agilis in the shiny or delicately shagreened galeae above (Fig. 14) and the longer first flagellar segment (Fig. 15 in pat; F1 minimum length about one-fifth of the second segment’s maximum length; M. agilis’ F1 minimum length about one-sixth to one-seventh of the second segment’s maximum length). M. agilis can appear similar to M. lupinus as well, differing largely in the subgeneric characteristics (M. lupinus is in M. Callimelissodes and M. agilis is in M. Eumelissodes), such as M. lupinus having a convex margin of the fourth sternum and M. agilis having a transverse margin (Fig. 16), but also differing in F5-F9 having longitudinal depressions on the outer surface in M. lupinus, and none in M. agilis (Fig. 17). This is not to say that all other Oregonian Melissodes males bear no resemblance to males of M. agilis, this is just a comparison of species that bear the highest resemblance.

Fig. 14. A comparison of the males of M. agilis (left) and M. menuachus (right) showing the differances in galael texture. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 15. A comparison of the males of M. agilis (left) and M. menuachus (right) showing the differances in first flagellar segment's length and ratio to the second flagellar segment. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 16. A comparison of the males of M. agilis (left) M. lupinus (right) showing the differances in the structure of the fourth sternum. The red line indicates the margin of S4. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 17. A comparison of the males of M. agilis (left) M. lupinus (right) showing the differances in the antennal structure, that being M. agilis has no longitudinal depressions on F5-F9 and M. lunpinus does. Photo credits: Christopher Wilson (All Rights Reserved).
♀ M. agilis can superficially resemble M. robustior due to the shared ochraceous vestiture. Thy can be easily distinguished from one another by the absence of a dark posteromedial mesoscutal patch of hairs on M. agilis, and a presence of one on M. robustior. If the mesoscutal hairs are absent due to wear, the two can also reliably be separated by the absence of dark tegular hairs on M. agilis, and the presence of them on M. robustior. If tegular hairs are also absent, M. robustior’s apical impunctate area of the first tergum extends basally on both sides creating somewhat indistinct lateral rounded projections while M. agilis has basal punctures that extend to the apical area at extreme lateral areas.
Fig. 18. Map showing an estimation for the known distribution of M. (Eumelissodes) agilis in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Johnson, 2020; Best et al., 2022a; Texas A&M University Insect Collection, 2023; Best, 2026; Illinois Natural History Survey, 2026; The International Barcode of Life Consortium, 2026). Data licensed under CC-BY 4.0, CC-BY-NC 4.0, or Public Domain as applicable (see "Dataset Licenses").
Fig. 19. A graph representing the phenology of M. agilis in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Ikerd, 2019; Gibbs, 2025; Scott, 2025; Bentley & Osborn, 2026; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; MT James Entomological Collection, Washington State University, 2026; Oram, 2026; University of Minnesota Insect Collection, 2026). Data licensed under CC-BY 4.0, CC-BY-NC 4.0, or Public Domain as applicable (see "Dataset Licenses").
Flower preference
M. agilis is a known oligolege of the tribes Astereae and Heliantheae (Robertson, 1926), and according to the flower records presented by Laberge (1961) and from more recent reports in literature (see Hogland, 2025a for entire records with updated synonymy), M. agilis seems to have a strong preference toward the genus Helianthus. According to Laberge (1961), of the 2,153 M. agilis collected with floral accounts, 1,608 were found atop Helianthus. This renders Helianthus to be favored 74.69% of the time by M. agilis.
Synonyms
Melissodes aurigenia Cresson, 1878
Melissodes ablusus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes ablusus
Melissodes ablusus is very rare (hence the lack of a common name) with only a single collection from Oregon (DeBano et al., 2024). Before this state record from Wallowa County (Zumwalt, Oregon), it was believed that M. ablusus only occurred in California (Laberge, 1961), and Utah based on a 2014 capture (Brigham Young University, Arthropod Collection, 2026). Although only collected in the Northeastern area of Oregon, due to its coastal presence in California (Laberge, 1961), it seems likely that this species ranges across the entire state. Something of note, M. ablusus was originally described from one female as a race of M. metenuus that resides in California (Cockerell, 1962). This is interesting as M. metenuus is a known inhabitant of Oregon, and therefore, it is possible that the singular report of M. ablusus in this region (which is quite a large northward range expansion), may in fact be a misidentification of M. metenuus. However, given the key-breaks between M. ablusus and M. metenuus for both sexes (the sex of the M. ablusus specimen from (DeBano et al., 2024) was not given) from Laberge (1961), the likely source in which the Oregonian specimen was identified, misidentification between the two seems unlikely. As can be determined from above, females of M. ablusus are similar to that of M. metenuus, but also resemble M. plumosus. Males seem to be similar to that of M. clarkiae based on antennal and flagella structure and, as the males of M. clarkiae are allied with M. metenuus and M. plumosus, it seems as if the males of M. ablusus may be as well. Given the original description presented by Cockerell (1926) was only based on a singular female and no males, descriptions from that of Laberge (1961) regarding males of M. ablusus are purely based on morphological characters, and are therefore tentative. With how few collections there are in general, and only a singular collection in Oregon, a map and phenology chart will be omitted.
Field Markers
Due to both the sexual dimorphism and the female dimorphism, three separate treatments will be listed in this section, (1) males, (2) dark females, (3) light females.
♂ The flagella (except the first completely dark flagellar segment) are orange-red and distinct longitudinal depressions laterally on F4-F10 (usually on F3 and F11 as well). The penultimate flagellar segment’s length
is two times its width or less and the galeae are densely tessellated. The first flagellar segment’s minimum length is less than one-fifth of the second
segment’s maximum length and the fourth sternum’s posterior apical area is widely convex. Terga 2-5 are opaque brown and occasionally translucent and yellowish
apically and the mandibular bases are cream colored (almost as pale as the labrum). The apical margin of the broad flap on the fourth sternum is not emarginate
medially (this would count as weakly medially emarginate on the second couplet), and sterna 3 and 5 are distinctly convex apically. Somewhat pale in vestiture,
with the sixth and seventh terga having brown hairs (not black like M. communis and M. tepidus).
♀ (Dark) The darker females have densely tessellated galeae and measure to be around 9-11 mm. in length. The head hairs are entirely black and the
thorax is a dark brown except for sparse pale-ish brown to ochraceous hairs that surround the base of the wing (these sparse pale-ish brown to ochraceous
hairs are also present dorsally on the metanotum and propodeum). The first tergum’s crowded basal punctures usually extend to the basal half medially and,
in the medial one-third, these punctures are mostly separated by one puncture width or less; minimally basally and sometimes more. The scopae are mostly
yellow to white, only brown near the basitibial plate as well as the apical areas of the basitarsi, and the ultimate flagellar segment is noticeably longer
than it is wide. Terga 2-4 have pale pubescent bands, descriptions of which follow: the basal zone of the second tergum has wide oblique fasciae of pale
pubescence present laterally that can sometimes almost meet medially and reach the apical margin laterally, if this is the case, then the fasciae is dark
brown apicolaterally and medially; The third tergum’s distal pale pubescent band is medially brown and separated from the apical margin by dark brown pubescence
and suberect hairs; the fourth tergum’s apical pubescent band is often dark brown, and occasionally pale brown laterally (only laterally, never medially).
Terga five and six have no lateral pale hairs.
♀ (Light) The lighter females have densely tessellated galeae and measure to be around 9-11 mm. in length (similar to that of the darker females).
The head hairs are usually pale apart from the abundant darker vertex hairs. The hairs on the inner surface of the hind bastitarsi and tibiae are
usually pale-colored red to yellow (if the head hairs and hairs on the inner surface of the hind bastitarsi and tibiae are mostly dark, then the
galeae are always dull; if not, check M. metennus). The thoracic hairs tend to be ochraceous to pale ochraceous, apart from the mesoscutum, which
has a large dark brown patch of hair posteromedially that usually reaches forward to the anterior margins of the tegulae, and the scutellum, which
has a large dark brown medial patch of hair. Terga 2-4 have pale pubescent bands, descriptions of which follow: the second tergum’s basal zone has
long pale ochraceous pubescence and the interband zone has short and relatively simple suberect dark brown to dark ochraceous hairs; the distal,
pale pubescent band of the second tergum is wide and arched (the band is narrowly interrupted medially and reaches or almost reaches the apex of
the tergum laterally); the apical area of the second tergum has short and simple appressed to suberect, brownish ochraceous hairs, apart from the
narrow glabrous margin; the third tergum is similar to that of the second, but the interband zone often has sparse, appressed, pale pubescence, and
the pale distal band is broader, narrowly interrupted medially, and extends to the apical margin laterally; the medial area of the fourth tergum has
a narrow dark pubescent area along the apical margin, and the broad, pale, pubescent band has scattered brown pubescence (the band is located basally
from the narrow dark pubescent area); terga 5 and 6 have dark brown hairs (tergum 5 has small lateral pale tufts). The first tergum’s crowded basal
punctures usually extend to the basal half medially and, in the medial one-third, these punctures are mostly separated by one puncture width or less;
minimally basally and sometimes more.
Similar Species
As stated above, M. ablusus is similar to that of M. plumosus and M. metenuus in the female sex, and M. clarkiae in the male sex. However, another species that is very closely related to M. ablusus in both sexes is M. minusculus. Although Laberge (1961) even notes that M. minusculus may in fact be a color variation of M. ablusus, but later denies this hypothesis based on punctuation and vestiture differences (M. minusculus seems to be consistent with their light vestiture in contrast to the darkness of M. ablusus). Although M. minusculus currently has no Oregon records, a recent collection in Idaho (Ikerd, 2019) seems to indicate a northward range expansion, which likely includes portions of Oregon. For this reason, M. minusculus will be included within the comparisons below. Similar to the field marks above, due to both the sexual dimorphism and the female dimorphism, three separate similar species comparisons will be listed in this section, (1) males (2) dark females (3) light females.
♂ Males of M. ablusus are similar to that of M. clarkiae based on the length on the length of the penultimate flagellar segment (in both species it’s two times its width or less) and the longitudinal depressions on the outer surfaces of F4-F10. However, these two species differ in the galeal sculpturing and the length of the first flagellar segment. M. ablusus have matte and dull galeae due to coarse, dense, and regular tessellation, whereas M. clarkiae have somewhat shiny to shiny galeae, but faintly dulled due to delicate reticular shagreening (the galeal surface is never tessellated). The minimum length of the first flagellar segment in M. ablusus is less than one-fifth of the second segment’s maximum length, whereas in M. clarkiae, the minimum length of the first flagellar segment is more than one-fifth of the second segment’s maximum length. M. ablusus is also quite similar to M. minusculus, though somewhat darker in vestiture. The two can be separated by the hairs on terga 6 and 7, where, in M. ablusus, these are mostly brown, and in M. minusculus, these are white to gold-colored. Although somewhat allied with M. metenuus and M. plumosus, M. ablusus starkly differs in the length of the penultimate segment. That being, in M. ablusus, the length of the penultimate segment is two times its width or less (as stated above), whereas in M. metenuus and M. plumosus, this segment is longer than two times its width.
♀ (Dark) darker females of M. ablusus are similar to that of the M. metenuus females based on the dark vetiture. However, the two differ in the galeal sculpture and tergal punctuation. That being M. ablusus has matte and dull galeae due to coarse, dense, regular tessellation, whereas M. metenuus has shiny to moderately shiny galeae with no, or very little, faint shagreening. Also, M. ablusus tends to have denser punctures on the terga than that of M. metenuus. M. ablusus also bears a resemblance to M. minusculus based on the coarsely, densely, regularly tessellated galeae, but can be separated by the darker vestiture (especially by the dark hairs on the face below the vertex). Another species allied to M. ablusus is M. plumosus, although darker individuals of M. ablusus tend not to superficially resemble M. plumosus. Nonetheless, M. ablusus can be identified from that of M. plumosus much as it is M. metenuus (dull galeae and denser punctures on the terga), but also by the darker vestiture.
♀ (Light) the lighter females of M. ablusus are much more similar to that of M. plumosus than that of M. metenuus due to the lighter hairs (mixed with darker hairs) on the facial areas. Similar to the dark females, lighter M. ablusus can be separated from both of these species by the dull galeae due to coarse, dense, regular tessellation and denser tergal punctures. Also similar to the darker female (due to these characters based on structure and sculpture instead of hair color), lighter M. ablusus bears a resemblance to M. minusculus based on the coarsely, densely, regularly tessellated galeae. However, these two are instead separated by the dark and pale hairs mixed on the face below the vertex in M. ablusus, and the entirely white head hairs (apart from the brown vertex hairs), including the face, in M. minusculus.
Flower preference
Currently unknown.
Synonyms
Melissodes metenua race ablusa Cockerell, 1926; Melissodes ablusa Cockerell, 1926.
Melissodes bicoloratus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes bicoloratus
Melissodes bicoloratus is a somewhat uncommon M. (Eumelissodes) known to inhabit western areas of the United States. Although there are no current Oregon records, this species has recently been collected in Washington (Ikerd & Engler, 2023), a large northward range-expansion that likely includes parts, and most of, Oregon (Fig. ??). Females of this species are very similar to that of M. bimatris and quite closely resemble their darker forms, differing at the scopal hair coloration. Females also resemble M. nigracauda, seemingly to a higher degree than that of the darker M. bimatris, and, based on vestiture alone, can’t be separated from one another. M. nigracauda, similarly to M. bicoloratus, currently hasn’t been found in Oregon, but has been collected in Washington (Washington State Department of Agriculture, 2025). When including this point in a map, and outlining a range based on collection locations, portions of Oregon become overlapped (See “Melissodes nigracauda” species treatment). For this reason, M. nigracauda is included in the foregoing comparisons. Males of M. bicoloratus are similar to that of M. bimatris in vestiture, apart from the apical band not medially obscuring the apical margin of the first tergum, and integumental color, apart from the lighter wing veins. Males are also somewhat similar to M. agilis and M. subagilis due to the aforementioned light wing veins, but can be separated from the former by the dark mandibular bases with no macula and the dark labrum, and the latter by shiny galeae and lateral apical hairs on the first tergum. (see “Similar Species” for more info). M. bicoloratus aales are distinctive in that the body length is approximately 11 mm.; the galeae are shiny with no shagreening above apart from the tips; the apical area of the first tergum is obscured by hairs in the lateral one-thirds or faintly less but not obscuring the apical margin medially; the overall vestiture is extremely pale ochraceous to white; the second tergum’s distal pale pubescent band is usually narrower than the apical area; the dorsal areas of the scutellum and mesoscutum do not have dark hairs; the wing veins are red to yellow; the labrum and mandibles are entirely black; the clypeus is entirely light excluding the dark spots denoting the tentorial depressions or the anterior notches and margin; and the first flagellar segment’s maximum length is less than, or equal to, one-third of the third segment’s minimum length. Females are distinctive in that the upper thoracic hairs (including those of the upper portions of the mesepisterna and posterior areas of the propodeum) are pale ochraceous to faintly rusty-red; the lateral and ventral thoracic hairs are black to dark brown; the scopae are dark brown to black; and the galeae are shiny with no shagreening. M. bicoloratus seems to be active between the months of March and October, with peak activity occurring in July (see Fig. ??).
Field Markers
Due to both the sexual dimorphism and the female dimorphism, three separate treatments will be listed in this section, (1) males, (2) dark females, (3) light females.
♂ Generally, males are fairly pale with distinctly light wing veins (much lighter than those of M. subagilis) as described below. The eyes are
greenish gray and the clypeus is yellow apart from the usually piceous apical margin. The labrum and mandibular bases are entirely black completely
black. The wing veins are reddish yellow to yellow and flagellar segments 1-10 are red to yellow on the underside and dark red to brown on top. The
apices of the terga are colorless hyaline and in length, measuring approximately 11 mm. The galeae are shiny with no shagreening above apart from the
tips and the overall vestiture is extremely pale ochraceous to white. The first flagellar segment’s maximum length is less than, or equal to, one-third
of the third segment’s minimum length. The first flagellar segment’s minimum length is about two-thirds of its maximum length, and about one-sixth the
maximum length of the second segment. The penultimate segment is about three times as long (maximum) as it is wide (minimum). The hairs on both the
head and the thorax are white to extremely pale ochraceous, but there are usually pale ochraceous hairs only on the dorsal area of the thorax. The first
tergum has long basal white to ochraceous hairs and long appressed to subappressed apical pale hairs that reach and obscure the apical margin only at
extreme lateral areas and not medially; lateral pubescence obscuring the apical area is usually less than one-third of the tergum’s width. The second
tergum has white basal pubescence and pale suberect, bristle-like hairs on the interband zone. The distal pubescent band of the second tergum is not
interrupted medially and often narrower than the apical area. The apical area of the second tergum has suberect pale hairs, although they are less
abundant than that of M. bimatris. Terga 3-5 are similar to that of the second tergum except for the interband zones, which in addition to the bristle-like
hairs there are also delicate, sparse, white, appressed pubescence. Also, the distal bands of terga 3-5 become closer to the apical margin with each tergum.
The sixth and seventh terga are covered in yellowish to white pubescence.
♀ The females of M. bicoloratus are quite distinctive compared to almost all other Melissodes due to their dark coloration. The overall vestiture is almost completely dark
except as follows: the dorsum of the thorax, vertex, and upper mesoplural areas, which are ochraceous to near-yellow; the upper areas of the face, the areas around the antennal
fossae, which are usually pale ochraceous. The metasomal hairs are mostly dark, but there are a few paler hairs on the basal one-half ton three-fifth of the first tergum, and
some basal pale hairs on the second tergum as well. The scopae are entirely dark. The wing veins are red to reddish brown and the galeae are shiny with no shagreening
dorsally excluding some faint delicate shagreening in less than, or equal to, the apical half. The mesoscutum is shiny and sparsely or not at all shagreened and has deep large
punctures that vary in diameter and are mostly separated by one-half to one puncture diameter. The underside of flagellar segments 3-10 are rufescent and the eyes are often greenish
gray to dark gray. The terga do not have their pubescent bands complete but instead these “bands” more so resemble lateral fasciae, or are sometimes entirely absent. If one of the
tergal bands is complete, then it’s the second tergum’s band and the hairs on the lower lateral mesepisternal surfaces are dark brown.
♂ To an extent, males of M. bicoloratus can resemble those of M. agilis, M. subagilis, M. lutulentus and M. bimatris. M. bicoloratus can be separated from M. agilis based on the shiny galeae, black mandibular bases, and hairs on the first tergum that obscure the apical area laterally. This species can also be identified from those of M. subagilis in a similar fashion, although, M. subagilis also have black mandibular bases, and therefore this character cannot be used. Moreover, M. bicoloratus is predominantly extremely pale ochraceous to white in vestiture, whereas M. subagilis is often somewhat yellow to ochraceous. M. bicoloratus can quite quickly be separated from those of M. bimatris due to the lack of a distinct apical band across the entire apical margin of the first tergum (in M. bicoloratus, these apical hairs are only present laterally, not medially). Similar to M. bimatris, M. lutulentus can somewhat superficially resemble M. bicoloratus due to the apical band of the first tergum. M. bicoloratus differs from M. lutulentus by the light mesoscutal and scutellar hairs (somtimes there are dark mesoscutal and scutellar hairs on M. lutulentus) and the apical hairs on the first tergum which are medially sparse and/or minutely barbed; only dense and plumose laterally. In M. lutulentus, these apical hairs are appressed, white, dense, plumose, and short, creating a distinct band that reaches the apex across the entire tergum.
♀ The females of M. bicoloratus closely resemble two species, those being M. bimatris and M. nigracauda, but can also somewhat resemble M. pullatellus and M. rivalis, be it not strongly. Females can be reliably and quickly separated from M. bimatris by the dark scopal hairs. However, females of M. nigracauda share the exact same vestiture coloration as M. bicoloratus including the dark scopae, and because of this, they are indistinguishable from one another using setae alone. Although not necessarily identifiable on the wing from each other as immobilization is required, M. biocoloratus can be distinguished from M. nigracauda by the unshagreened shiny galeae. Due to its dark vestiture, M. bicoloratus can somewhat resemble other melanistic Oregonian species, those being M. pullatellus and darker varieties of M. rivalis. Females can be identified from both of these species by the dorsal thoracic areas having ochraceous to near-yellow hairs (no dark hairs). Furthermore, M. bicoloratus differs from M. rivalis by the dark scopae and convergence of eyes towards the mandibles, and differs from M. pullatellus by the aforementioned entirely ochraceous to near-yellow dorsal thoracic hairs.
Fig. ?? Map showing an estimation for the known distribution of M. (Eumelissodes) bicoloratus showing that it's range overlaps portions of Oregon, and therefore, likely occurs within these areas. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Johnson, 2020; Carril et al., 2023; Ikerd & Engler, 2023; Bentley & Osborn, 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenological activity of M. bicoloratus. The x value is the month, and the y value is the number of documented observations. As no M. bicoloratus have currently been foun in Oregon, only one phenological line is given. Data compiled from (Ikerd, 2019; Johnson, 2020; Carril et al., 2023; Ikerd & Engler, 2023; Bentley & Osborn, 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 (see "Dataset Licenses").
Flower preference
Little is known in regards to the flower records and/or preference of M. bicoloratus. All flower records included in this list are from reports in the literature or datasets. Each flower has a parenthesized reference listed after it, corresponding to the literary work or dataset in which it was recorded. Currently, M. bicoloratus has been collected atop Acaciella angustissima (McElrath, 2023; GBIF record 3801940257), Chrysothamnus sp. (Laberge, 1961), Ericameria arborescens (McElrath, 2023; GBIF record 3801376267), Melilotus sp. (Laberge, 1961) Penstemon palmeri (Laberge, 1961).
Synonyms
Melissodes bicolorata Laberge, 1961
Melissodes bimatris

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes bimatris
Melissodes bimatris, the mothered long-horn bee, is incredibly variable in the female sex. As seen in the group-name statement, M. bimatris females can take a dark or light form (Fig. 22; males aren’t nearly as variable). This species is somewhat common to rare across its entire range, and it seems similarly so in Oregon with only 21 publicly documented occurrences placing it 12th most common in the genus (see Fig. 13). Males of M. bimatris resemble M. semilupinus and, depending on which morph, females can either resemble M. bicoloratus and M. nigracuada (dark), or M. semilupinus (light). Males are distinctive in that the mandibular bases are black with no yellow maculations and the labrum is completely black, or sometimes has a small mediobasal pale macula; the first flagellar segment’s minimum length is usually less than one-third of the third segment’s maximum length; the first flagellar segment’s maximum length is more than one-third of the third segment’s minimum length; and the first tergum has an obvious and well developed distal band consisting of appressed or subappressed, dense, white pubescence that reaches and obscures the apical margin across the entire tergum (sometimes the apical band is worn). Females are distinctive in two manners, that being the dark form and light form. The dark females are distinctive in that the entire vestiture is mostly black to very dark brown apart from the ochraceous dorsal thoracic hairs and the ochraceous to fluvo-ochraceous scopae (highly distinctive in Oregon). The light females are distinctive in that the apical area of the third tergum is glabrous, impunctate, and minimally wider than that of the medial width of the second tergum’s distal band; the mesoscutum doesn’t have a dark brown to black patch of posteromedial hairs, or if so, then very few dark hairs are present; the clypeus often protrudes beyond the eye by about one-half of an eye’s width in profile; the first tergum’s apical impunctate area is medially equal to, or less than, one-half of the punctate basal area; and the third tergum usually has long, suberect, pale hairs apical hairs in the apical area. Although not documented across the entirety of Oregon (its most westward collection occurring in Klamath county. Fig. ??; Illinois Natural History Survey, 2026), it seems likely given its coastal presence in California (Laberge, 1961), that M. bimatris’ range overlaps the entire state. This species seems to be active from May to November, but most chiefly so in September (see Fig. 23).
Field Markers
Due to both the sexual dimorphism and the female dimorphism, three separate treatments will be listed in this section, (1) males, (2) dark females, (3) light females.
♂ Generally a pale to ochraceous bee with distinct apical banding on the first tergum. The eyes are green to gray and the clypeus is
yellow apart from the usually piceous apical margin. The mandibular bases are entirely black and the labrum is usually completely black,
although sometimes there’s a small pale maculation positioned mediobasally; the wing veins are black to dark reddish brown and flagellar
segments 2-10 are red to yellow on the underside and dark red to brown on top (first segment completely dark brown). The apices of the
terga are colorless to yellow hyaline. The first flagellar segment’s minimum length is about two-thirds of its maximum length, and about
one-fifth or somewhat less than the maximum length of the second segment. The penultimate segment is about three times as long (maximum)
as it is wide (minimum). Flagellar segments 8-10 or 7-10 are somewhat crenulate, similar to that of the male M. semilupinus. However, no
flagellar segments longitudinally laterally depressed as in many Callimelissodes males or M. paululus. The first flagellar segment’s maximum
length is more than one-third of the third segment’s minimum length. The hairs on both the head and the thorax are white to pale ochraceous,
but on the vertex of the head and the dorsal area of the thorax, the hairs usually become a bit darker. The first tergum has long basal white
to ochraceous hairs and long appressed to subappressed apical pale hairs that reach and obscure the apical margin across the entire tergum as
long as they aren’t worn off. The second tergum has white basal pubescence and pale erect, bristle-like hairs on the interband zone. The distal
pubescent band of the second tergum is not interrupted medially and often narrower than the apical area. The apical area of the second tergum has
many suberect pale hairs. Terga 3-5 are similar to that of the second tergum except for the interband zones, which in addition to the bristle-like
hairs there are also delicate, sparse, white, appressed pubescence; interband zone hairs suberect instead of erect. Also, the distal bands of terga
3-5 become closer to the apical margin with each tergum. The sixth and seventh terga are covered in yellowish to white pubescence. However, two
males have been found to have pale rufescent to yellow-ochre and brownish red pubescence basally on terga 3-5.
♀ (Dark) Females of the dark morph are incredibly easily recongnizable and identifiable via vestiture and macro characteristics as follows.
The eyes are gray (although not from Laberge, 1961, a dark blue eye color is common as well) and the wing veins are black to dark brownish red.
The underside of flagellar segments 3-10 are often rufescent (somtimes dark brown to black) and the apical area of the first tergum has a narrow
hyaline margin. The vestiture is almost completely dark apart from the pale ochraceous to near-yellow dorsal thoracic hairs (sometimes these hairs
occur the base of T2 and T1) and bright ochraceous to fluvo-ochraceous scopal hairs, apart from the darker hairs on the inner surfaces of the hind
basitarsi. The mesoscutum doesn’t have a dark brown to black patch of posteromedial hairs, or if so, then very few dark hairs are present. The first
tergum’s apical impunctate area is medially equal to, or less than, one-half of the punctate basal area. The galeae are shiny and the mesoscutum is
shiny without shagreening. The bands often aren’t complete, but given how distinctive this morph is, the characteristics described are sufficient for
proper identification.
♀ (Light) In contrast to their dark counterparts, light females of M. bimatris are much less recognizable and can easily be mistaken for a few other
Melissodes. The eyes are gray (although not from Laberge, 1961, a dark blue eye color is common as well) and the wing veins are black to dark brownish red.
The underside of flagellar segments 3-10 are often dark brown to black and the apical area of the first tergum has a narrow hyaline margin. The general
vestiture is mostly white to pale ochraceous with darker hairs on the dorsal thoracic and head (vertex) areas. The first tergum’s apical impunctate area
is medially equal to, or less than, one-half of the punctate basal area. The apical areas of the second and third terga have long, pale, suberect hairs and
the mesoscutum doesn’t have a patch of dark brown to black posteromedial hairs (if patch present, then composed of very few dark hairs). The hairs on the
legs (excluding scopae) are mostly pale ochraceous to white, apart from the outer apical area of the fore tibiae and middle tibiae, the fore tarsi, and inner
surfaces of the hind basitarsi, which are dark reddish brown to brown. The scopal hairs are sometimes pale ochraceous but often pale yellow and notably long.
The galeae are shiny and the mesoscutum is shiny without shagreening. The flagella are usually entirely black without red ventral markings and the second
flagellar segment is less than, or equal to, its width. The apical area of the third (and often second) tergum often bears long, suberect, pale hairs with
the band not reaching the apex.

Fig. 20. A juxtaposition of the dark (left) and light (right) morphs of the females of M. bimatris. Photo credits: Lori Weidenhammer (left); Christopher Wilson (right).
Similar Species
While highly distinctive within the males due to the dense hair band obscuring the apical margin of the first tergum along with the lack of mandibular maculations (lack of mandibular maculations separating it from that of M. semilupinus), and the dark females are distinctive due to the fluvo-ochraceous scopae, lighter forms of the females have an incredibly high resemblance to M. semilupinus. Similar to the field marks above, due to both the sexual dimorphism and the female dimorphism, three separate similar species comparisons will be listed in this section, (1) males (2) dark females (3) light females.
♂ Males of M. bimatris are analogous to that of M. semilupinus (as stated above), mostly so due to the apical band across the second tergum (Fig. 22). However, the two can be quite reliably and easily separated from one another by the lack of mandibular maculations on M. bimatris and the presence of them on M. semilupinus (Fig. 23). Due to these distinctive characters, no other Melissodes, except for M. semilupinus, directly resemble M. bimatris (apart from similar vestiture coloration).

Fig. 22. A comparison of the apical area of the first tergum showing the similarities between the males of M. bimatris (left) and M. semilupinus (right). Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 23. A comparison of the mandibular bases between the males of M. bimatris (left) and M. semilupinus (right) showing the lack of maculations on M. bimatris and the presences of maculation on M. semilupinus. Photo credits: Christopher Wilson (All Rights Reserved).
♀ (Dark) The darker females of M. bimatris are quite distinctive and easily recognisable due to their melanistic vestiture in contrast to their light dorsal thoracic hairs. Out of the entire genus, only two other Melissodes share this same coloration, those being M. bicoloratus and M. nigracauda. Interestingly, neither have currently been collected in Oregon, but both have been recently collected in Washington (Ikerd & Engler, 2023; Washington State Department of Agriculture, 2025), a range-expansion that includes parts, and most of, Oregon (see respective species treatments for more information). For this reason, these two species will be included within this comparison. M. bimatris is distinct and easily told apart from both M. bicoloratus and M. nigracauda by the light scopal hairs in contrast to the dark, almost nearing black, scopal hairs of M. bicoloratus and M. nigracauda (Fig. 24). M. bimatris also differs from that of M. nigracauda by the shiny galeae in contrast to the shagreened and dulled galeae of M. nigracauda (M. bicoloratus also have shiny galeae, and therefore, this character cannot be used in comparison between the two).

Fig. 24. A comparison of the scopal hair coloration of the females of M. bimatris (top left), M. nigracauada (top right), and M. bicoloratus (bottom) showing the light hairs on M. bimatris and the dark hairs on M. nigracauada and M. bicoloratus. Photo credits: Lori Weidenhammer (top left); Natural History Museum of Los Angeles County (top right) (CC-BY 4.0); Christopher Wilson (bottom) (All Rights Reserved).
♀ (Light) The light form of the females of M. bimatris are quite similar to that of M. ochreus and M. semilupinus, M. ochreus will be omitted as their current known range doesn't include or overlap Oregon, but comparisons will be made for M. semilupinus. The most notable difference between M. bimatris and M. semilupinus is the second flagellar segment's length. In M. semilupinus, the length of the second flagellar segment is faintly, but noticeably longer than its width, whereas in M. bimatris, the length of the second flagellar segment is less than, or equal to, its width.
Fig. 20. Map showing an estimation for the known distribution of M. (Eumelissodes) bimatris in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Johnson, 2020; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; MT James Entomological Collection, Washington State University, 2026; The International Barcode of Life Consortium, 2026). Data licensed under CC-BY 4.0 (see "Dataset Licenses").
Fig. 21. A graph representing the phenology of M. bimatris in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Ikerd, 2019; Johnson, 2020; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; MT James Entomological Collection, Washington State University, 2026; The International Barcode of Life Consortium, 2026) and range-wide compiled from (Ikerd, 2019; Johnson, 2020; Carril et al., 2023; Texas A&M University Insect Collection, 2023; Gibbs, 2025; Bentley & Osborn, 2026; Brigham Young University, Arthropod Collection, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Kenneth S. Norris Center for Natural History, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026; University of Arizona Insect Collection, 2026a). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 (see "Dataset Licenses").
Flower preference
M. bimatris is a known oligolect of Asteraceae (as are almost all Melissodes) but seems to specifically prefer the genus Chrysothamnus (Laberge, 1961). As Laberge (1961) presented, of the 72 M. bimatris collected with floral accounts, 53 were found atop Chrysothamnus. This renders Chrysothamnus to be favored 73.61% of the time by M. bimatris.
Synonyms
None.
Melissodes clarkiae

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes clarkiae
Melissodes clarkiae is a somewhat distinctive bee that seems to be allied with M. metenuus and M. plumosus in both sexes, and M. ablusus as well as M. minisculus (due to the antennal structure) in the male sex. This species was originally known to inhabit only the coastal areas of Oregon and California (Laberge, 1961). However, a collection in Oregon from 2019 implies a presence further inland that historically assumed (see Fig. ??; Best et al., 2022a). The females are quite distinct from all other Melissodes within their subgenus (Callimelissodes) due to the scopal hairs usually consisting of only one or two branches on either side of the rachis and the rachis protruding beyond the branched area. The males are distinct in F4-F10, and often F3-F11 having longitudinal depressions on the outer surface, the length of the penultimate flagellar segment being two times its width or less, the shiny to somewhat shiny galeae, and the minimum length of F1 being more than one-fifth of F2’s maximum length. Although distinctive in the manner, the males are similar to those of M. metenuus and M. plumosus, but aren’t put in this group due to the length of the penultimate flagellar segment (see more in “Similar Species”). Something of note, due to the simple feebally branched hairs on the scopae, females (unlike many other Melissodes) likely do not carry Asteraceae pollen and therefore likely do not pollinate such plants (Laberge, 1961). While very few records are available with floral data attached, the morphological structure of both the scopae of M. clarkiae and the small dry spherical pollen of Asteraceae plants do not suggest a relationship between the two (the feebly branched scopae likely can’t hold the small pollen grains; Laberge, 1961). As presented by Laberge (1961), M. clarkiae is a probable oligolect of Onagraceae due to its scopal structure lending way in carrying pollen tethered by viscin threads (such as plants in the genus Oenothera; Cruden & Jensen), or larger triangularly shaped pollen (such as the plants in the genera Clarkia, hence the common name; Halbritter, 2016).
Field Markers
♂ The flagellum is almost entirely red-colored ventrally except for the first entirely dark segment. The mandibular bases and labrum are yellow,
but the labrum is marginally darkened. The clypeus is lemon-yellow, but very narrowly darkened posteriorly, and flagellar segments 3-10, usually
the base of F11 as well, have shiny, shallow, longitudinal dorsolateral depressions. The length of the penultimate flagellar segment is equal to,
or shorter than, twice its width (usually faintly shorter) and the ultimate segment’s length is more than three times its width. The fourth sternum
has a wide hyaline flap that is medially incredibly shallowly, or not at all, emarginate, and the fifth and third sterna are noticeably apically
convex. The overall vestiture is mostly cinereous, including the vertex of the head and dorsal thoracic areas. Terga 2-4 have pale pubescent bands,
descriptions of which follow: the overall vestiture of the second tergum is pale ochraceous to white with the basal and distal bands taking on the
latter color (distal band medially interrupted); the apical area of the second tergum bears simple, suberect, often pale ochraceous (sometimes brown)
hairs; the third and fourth terga are similar to that of the second, apart from the distal bands being uninterrupted, the basal tomentum being brown,
and the apical hairs being largely brown (minimally medially and sometimes more); the fifth tergum similar to that of the fourth, except the distal
band often reaches the apex across the entire tergum; terga six and seven are entirely covered in golden-brown to golden-yellow hairs. The eyes are
often bluish gray to gray, and on rare occasions, greenish blue. The galeae are often dull due to dense shagreening.
♀ Somewhat pale in vestiture with some darker patches of hairs on the vertex, the scutellum, and the posteromedial areas of the mesoscutum (mesoscutal
dark patch reaches, or almost reaches, the anterior margins of the tegulae). The scopal hairs are long and usually consist of only one or two branches on
either side of the rachis and the rachis protrudes beyond the branched area. The underside of F3-F10, and usually the apical area of F2, are yellow to
reddish and the galeae are somewhat shiny above, but dulled due to minute reticular shagreening, often becoming less shagreened in the apical one-third,
and have largely spaced punctures with straight hairs arising from them. The mesoscutum is posteromedially, laterally, and anteriorly shiny and often has
a small impuncate area posteromedially. The pygidial plate is narrowly U-shaped and the wing veins are dark brown. Terga 2-4 have pale pubescent bands,
descriptions of which follow: the distal pale pubescent band of the second tergum is wide, often becomes medially interrupted, arched, and laterally reaches,
or almost reaches, the apes of the tergum; the apical area of the second tergum is narrowly glabrous marginally, but anterior to this margin bears suberect to
appressed, simple, ochraceous to brown hairs (more than that of M. lupinus); the third tergum, is similar to that of the second, apart from the wider
uninterrupted distal band that is noticeably spaced from the apical margin (not reaching the apex), and the interband zone, which bears appressed, sparse,
pale pubescence; the pale ochraceous band on the fourth tergum is wide, positioned apically (no pale simple hairs near the medial area of the apical margin).
The fifth and sixth terga are covered in pale brown hairs apart from the large lateral tufts, which are cinereous., and the sternal hairs are golden brown,
but laterally, these hairs become whitish.
Similar Species
M. clarkiae is quite distinctive in both sexes, but most predominantly so in females. In general, the characteristic of simple scopal hairs are quite distinctive throughout the entire genus, but in Oregon, M. clarkiae is the only species with this trait. Males are distinctive in F4-F10 (usually partially developed in F3 and F11) having longitudinal depressions, the penultimate flagellar segment’s length being equal to, or shorter than, two times its width, and the somewhat shiny to shiny galeae that are only faintly dulled due to delicate reticular shagreening (not tessellated). However, due to their allyship, M. clarkiae can superficially resemble M. metenuus and M. plumosus in both sexes, and M. ablusus as well as M. minusculus (due to the antennal structure) in the male sex.
♂ Males can somewhat resemble M. metenuus and M. plumosus, but more strongly so for M. ablusus and M. minusculus due to the distinctive antennal structure they share. M. clarkiae can be separated from M. metenuus and M. plumosus by the length of the penultimate flagellar segment. That being, in M. clarkiae, the length of the penultimate flagellar segment is two times its width or less, whereas in M. metenuus and M. plumosus, the length of the penultimate flagellar segment is longer than two times its width. In regards to M. ablusus and M. minusculus, M. clarkiae can be separated from both by the galeae, which are somewhat shiny to shiny dorsally but faintly dulled due to delicate reticular shagreening (no tessellation), in contrast to the matte and dull galeae due to coarse, dense, and regular tessellation of M. ablusus and M. minusculus. Also, the minimum length of the first flagellar segment of M. clarkiae is less than one-fifth of the second segment’s maximum length, whereas for M. ablusus and M. minusculus, F1’s minimum length is more than one-fifth of F2’s maximum length.
♀ Females are more strongly allied with M. metenuus and M. plumosus than the males, but not by much. M. clarkiae is separated from all Melissodes in Oregon by the combination of the U-shaped pygidial plate and the simple scopal hairs. Due to this, no Melissodes directly resemble those of M. clarkiae, and a further comparison is unnecessary.
Fig. ??. Map showing an estimation for the known distribution of M. (Callimelissodes) clarkiae in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Best et al., 2022a; Bentley & Osborn, 2026; Best, 2026; The International Barcode of Life Consortium, 2026). Data licensed under CC-BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. clarkiae in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Best et al., 2022a; Bentley & Osborn, 2026; Best, 2026; The International Barcode of Life Consortium, 2026) and range-wide compiled from (Ikerd, 2019; Johnson, 2020; Best et al., 2022a; Bentley & Osborn, 2026; Best, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; The International Barcode of Life Consortium, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
M. clarkiae seems to be an oligolect of Onagraceae due to the scopal structure and flower records. However, very few flower records are available (7), so a definitive statement on oligolecey and/or preference seems unjustified. All flower records included in this list are from reports in the literature or datasets. Each flower has a parenthesized reference listed after it, corresponding to the literary work or dataset in which it was recorded. Currently, M. clarkiae has been found atop Clarkia speciosa subsp. speciosa (Laberge, 1961).
Synonyms
None.
Melissodes communis

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes communis
Melissodes communis, the common long-horn bee, is quite distinctive due to the paucity of species in its subgenus (Melissodes s.s.) that occur in Oregon. Males are highly distinctive due to the black hairs on the last two terga, entirely yellow clypeus with no dark margin (excluding the tentorial depressions) ( Melissodes s.s. subgeneric characteristic) and the thin distal band on the second tergum. Females are also distinctive, though like most species in the genus, less so than the males. Females are distinctive in the length of the metanotum, the coloration of the hairs on the inner surfaces of the hind basitarsi, the comparably “impunctuate” apical tergal areas, and the narrow bands on the second and third terga that do not diffuse across most of the tergum. M. communis contains two subspecies (M. communis ssp. communis and M. communis ssp. alopex), only one of which occurs in Oregon (M. communis ssp. alopex), having just a slightly wider distal pale band of the second tergum and white metasomal pubescence, prominently so on the females than that of M. communis ssp. communis. This species, although the vernacular name suggests otherwise, ranks surprisingly low in Oregonian commonality, standing at 30 current publicly available state records and only ninth most collected within the genus. Something of interest, M. communis seems to largely differ from most Melissodes in flower preference, being most often observed atop families other than Asteraceae. This species has been found across almost all of Oregon and undoubtedly occurs throughout the entire state (Fig. ??). Although collections have occurred in March, commencement of activity for M. communis seems to occur between the months of April and May, with peak activity occurring during June, July, and August (Fig. ??).
Field Markers
♂ Sometimes a larger bee, in length, measuring 10-15mm. The flagellum is almost entirely red-colored ventrally except for the first entirely dark segment.
The clypeus and labrum are bright yellow to pale (not darken marginally) and the mandibular bases have yellow maculations that are minimally wider than the
triangular basal depressions and often completely cover the basal half. The first flagellar segment’s minimum length is about one-sixth of the second segment's
maximum length and is also distinctly longer than the pedicle on the same side. The last two terga are entirely covered in black to dark brown hairs rather
than the paler hairs that are common amongst most males. The second tergum’s distal band is narrow and usually equal to, or less than, one-half the width of
the medial portion of the apical area. The vestiture is somewhat pale ochraceous to almost whitish on the metasoma. The thoracic hairs do not have a dark patch,
nor do the head hairs. The wing veins are a somewhat lighter reddish brown and the leg hairs are often pale ochraceous (similar to the thoracic hairs). The
galeae are shiny with no shagreening, apart from the tips, and have widely spaced punctures with hairs dorsally arising from them. The mesoscutum is shiny with
no, or delicate, shagreening and the eyes tend to be greenish.
♀ Somewhat darker (lighter than those of M. bicoloratus but darker than those of M. agilis or M. plumosus) and larger bee, in length, measuring 12-16 mm. The
underside flagellar segments 3-10 are reddish-brown to red and the metanotum is as long as, or longer than, the propodeum’s dorsal surface medially. The first
tergum is usually only narrowly hyaline marginally, or not hyaline at all. The second tergum’s distal pale band is often never medially interrupted and the
anterior margin of this band is straight while the posterior margin is evenly curved. The hairs on the inner surface of the hind basitarsi and tibiae are
reddish-orange to yellow (not distinctly contrasting with the rest of the scopae) and the band on the second tergum is narrow and not diffuse. The apical
areas of the second and third terga are not distinctly medially punctate posterior of the bands, or if there are punctures, then they are no wider than the
base of the hairs that arise from them. The head hairs are mostly white to ochraceous apart from the abundant dark brown vertex hairs (these can at times be
very sparse or absent entirely). The thoracic hairs are mostly pale colored (often ochraceous), but the mesoscutum has a large posteromedial patch of dark
hairs (these can at times be very sparse or absent entirely, similar to those of the vertex), and the scutellar hairs are mostly dark with ochraceous to white
hairs on the periphery. The tergal hairs are mostly whitish, apart from the fifth and sixth terga which are covered in black to dark brown hairs. However, the
band on the fourth tergum is often medially interrupted by a small rectangular or triangular notch of suberect dark brown hairs.
Similar Species
The subgenus in which M. communis resides (Melissodes ss.) in total contains 24 species, however, only two inhabit Oregon. Due to the subgeneric characteristics (metanotum as long as, or longer than, the propodeum’s dorsal surface medially for the females and the dark hairs on the last two terga as well as the non-darkened clypeal and labral margins for the males), this in turn renders M. communis quite distinctive in Oregon with only a single other species that it’s weakly allied with, M. tepidus. Both sexes of M. communis can be separated from both sexes of M. tepidus by the narrower distal band on the second tergum.
♂ In M. communis the second tergum’s distal band is narrow and usually equal to, or less than, one-half the width of the medial portion of the apical area, whereas in M. tepidus, it’s equal to, or almost equals, the medial portion of the apical area. In M. communis, the fourth tergum’s pale band is clearly narrower than fourth times the medial portion of the apical area. However, in M. tepidus, this band more than, or equal to, fourth times the medial portion of the apical area. The second tergum’s distal band on M. communis can be, or almost be, as wide as the medial portion of the apical area, but the fourth tergum’s pale band is always clearly narrower than fourth times the medial portion of the apical area.
♀ In M. communis, the second and third terga have somewhat narrow and thin bands (thinnest on the distal band of T2) and the apical areas of the terga are not distinctly medially punctate posterior of the bands, or if there are punctures, then they are no wider than the base of the hairs that arise from them. In M. tepidus, the bands on the second and third terga are very wide, diffusing across the tergal surface, and the apical areas, located posterior to the bands, are distinctly medially punctate, with punctures that are notably wider than the hairs that arise from them.
Fig. ??. Map showing an estimation for the known distribution of M. (Melissodes) communis in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Johnson, 2020; Best et al., 2022a; Ikerd & Engler, 2023; Bentley & Osborn, 2026; Best, 2026; The International Barcode of Life Consortium, 2026). Data licensed under CC-BY 4.0 or CC-BY-NC 4.0 as applicable (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. communis in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Ikerd, 2019; Johnson, 2020; Best et al., 2022a; Bentley & Osborn, 2026; The International Barcode of Life Consortium, 2026) and range-wide compiled from (Eardley & Ranwashe, 2017; Ikerd, 2019; Johnson, 2019; Johnson, 2020; Best et al., 2022a; Archbold Biological Station, 2023; Carril et al., 2023; Ikerd & Engler, 2023; Purdue Entomological Research Collection, 2023; Texas A&M University Insect Collection, 2023; North Carolina State University Insect Collection, 2024; Veit & McFarland, 2024; Droege & Maffei, 2025; European Bioinformatics Institute (EMBL-EBI), GBIF Helpdesk, 2025; Gibbs, 2025; Holm et al., 2025; Pozo & Comisión, 2025; Quiroz & Comisión, 2025; Scott, 2025; Sánchez & Comisión, 2025; Zhuang, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Best, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026a; Cheadle Center for Biodiversity and Ecological Restoration, 2026b; Clemson University Arthropod Collection, 2026; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Florida State Collection of Arthropods, 2026; Frost Entomological Museum, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Kelly et al., 2026; Kenneth S. Norris Center for Natural History, 2026; Luther College, 2026; Mertz et al., 2026; Mississippi Entomological Museum, 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Natural Science Research Laboratory, Museum of Texas Tech University (TTU), 2026; New Mexico State Collection of Arthropods, 2026; Northern Arizona University, 2026a; Orrell & Informatics, 2026a; San Diego Natural History Museum, 2026; The International Barcode of Life Consortium, 2026; UMMZ & LSA, 2026; University of Arizona Insect Collection, 2026a; University of Arizona Insect Collection, 2026b; University of Kentucky, 2026; University of Minnesota Insect Collection, 2026; Virginia Tech Insect Collection, 2026; Wild, 2026; Soon, n.d.). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Melissodes compositus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes compositus
Melissodes compositus is a less common Callimelissodes that is most closely allied to M. lustrus, but is also related to M. metenuus, and M. plumosus in both sexes. Males are distinctive in the antennal structure, specifically the flagellar depressions and length of the penultimate segment, the glabrous apical tergal areas, and the sculpting of the interband zones on the second and third terga. The females are most closely related to M. lustrus and are distinctive in the sculpture of terga 1-3 and the mesoscutal, scutellar, and galeal sculpture. Historically, this species range was only known to include the states Arizona, Colorado, Montana, New Mexico, and Utah. However, collections from Idaho coupled with Nevadan, Montanan, and Northern Californian collections seem to suggest that there may be a presence of this species in Oregon (Fig. ??). For this reason, M. compositus is included within this guide. As the name suggests, M. compositus likely prefers plants from the family Asteraceae (originally called Compositae), but the paucity of floral data leaves uncertainty. As none have been collected in Oregon yet, only one line is given for the phenology graph, this line representing the range-wide phenology of this species. It seems as if M. compositus is active between the months of May and October, with peak activity occurring in August.
Field Markers
♂ Overall, a pale bee with translucent-brown to yellow apical margins that are highly shiny and glabrous. The underside flagellar segments 2-11
are red to yellow (these segments are dark brown on top) and the mandibular bases, clypeus, and labrum (the labrum having its margin not darkened)
are yellow. The eyes are bluish gray to gray, and on rare occasions, greenish blue, the wing veins are black to brown, and the apical areas of
terga 2-5 are usually yellow-brown hyaline. The first flagellar segment’s minimum length is more than one-fifth of the second segment’s maximum
length, and segments 4-10 (sometimes also on 11 and poorly defined and short on 3) have shiny, shallow, longitudinal dorsolateral depressions.
The length of the penultimate segments is more than twice, but less than three times, its width and the ultimate segment’s length is approximately
three times its width. The interband zones of the second and third terga have round, deep, abundant punctures that are mostly separated by
noticeably less than a single puncture diameter. The head hairs are mostly dark ochraceous to pale ochraceous apart from the vertex, which often
has some darker hairs. The thoracic hairs are also mostly dark ochraceous to pale ochraceous apart from the dorsal areas, which tend to bear darker
hairs. The tergal hairs are mostly pale ochraceous to white, with the second, third, and fourth terga having glabrous apical areas; the fifth
tergum, which reaches the apex across the entire tergum; and the sixth and seventh terga, which have some pale brown to dark ochraceous hairs,
predominantly medially and basally. The legs have white to pale ochraceous hairs, differing at the inner surfaces of the tarsi, which are pale
orange to yellow.
♀ General vestiture is somewhat pale (lighter than those of M. communis ) and the underside of F3-F10, and usually the apical area of F2 are
yellow to reddish. The basal puncture on the first tergum do not exceed the basal one-third and the galeae are moderately shiny above, and delicately
reticularly shagreened, but never densely tessellated. The ultimate flagellar segment is longer than its width, appearing more rounded than flattened,
the pygidial plate has an elongated U-shape, and the scopal hairs are plumose. The scopal hairs are mostly pale ochraceous to white and the head
hairs are often pale apart from the abundant darker vertex hairs. The thoracic hairs are laterally pale ochraceous on the lower surfaces, and mostly
ochraceous to yellow on the rest of the thorax apart from the scutellum, which has medial dark brown hairs, and the mesoscutum, which has a dark
brown posteromedial patch that reaches the medial portion of the tegulae. The apical tergal areas are shiny and glabrous and the galeae are somewhat
shiny (comparatively so against M. metenuus and M. plumosus). The hairs on the inner surfaces of the hind basitarsi are red to yellow and the fifth
and sixth terga have golden to pale brown medial hairs and large white lateral tufts of hairs. The mesoscutal punctures are coarse, and the ones
positioned medially from the parapsidal line are mostly larger than of the mesepisternal or scutellar punctures; mostly separated by less than, or
equal to, one-half of a puncture diameter.
Similar Species
As a result of its subtle and at times minute diagnostic characters, M. compositus can resemble a number of Oregonian Callimelissodes. The females are strongly allied to M. lustrus and M. glenwoodensis due to the shared sculpturing on the first tergum, and males are allied to these species as well due to the sculpturing of the second and third terga as well as the antennal structure. M. compositus can also resemble a few Eumelissodes due to shared vestiture coloration, but can easily be separated from these by the subgeneric characteristics. These being the convex apical margin on the fourth sternum in males along with the flagellar segment depressions, and the punctures not surpassing the basal one-third on the first tergum in the females; note that this female character is shared amongst many Callimelissodews but not all.
♂ Due to the apical areas of the second and third terga having simple, erect to suberect hairs, the galeae often being shiny without shagreening or delicately reticularly shagreened, and the third flagellar segment usually having a well defined ventrolateral depression, males of M. compositus are strongly allied with those of M. lustrus. M. compositus differs from M. lustrus in that the interband zones of the second and third terga have round, deep, abundant punctures that are mostly separated by noticeably less than a single puncture diameter, and the eyes are often greenish blue to blue. Whereas in M. lustrus, the interband zone of the third tergum, and usually the second as well, have punctures that are mostly separated by one puncture diameter, and several punctures that are separated by 2 to 3 puncture diameters, and the eyes are often yellowish green to green. Males can also resemble M. metenuus and M. plumosus as well due to the pale vestiture (shared by almost all males in the genus) and the length of the penultimate flagellar segment being longer than twice its width. However, M. compositus differs from these by the comparably shiny galeae, the noticeably glabrous and shiny apical tergal areas, and the coarser and more well-defined scutellar and mesoscutal punctures.
♀ Females are interesting as they are allied with M. lustrus just as the males, but also M. glenwoodensis to an extent. This is due to the shared characteristics of the first tergum’s crowded basal punctures do not surpass the basal third or less medially (or if the medial one-third of the first tergum has punctures, then they are mostly separated by more than two puncture diameters), and the galeae are moderately shiny above, but delicately reticularly shagreened, with no dense tessellation. However, M. compositus can be quickly separated from those of M. glenwoodensis by the yellow to red hairs on the inner surfaces of the hind basitarsi (M. glenwoodensis has black to dark reddish hairs), and the pale brown medial to long white lateral hairs on the fifth and sixth sterna (M. glenwoodensis has black to dark brown hairs with no lateral tufts). Less diagnostic, but still of importance, M. glenwoodensis tends to be much larger than M. compositus, measuring 11-14 mm. in length, whereas M. compositus measures 9.5-11.0 mm. in length. However, due to the characteristics M. compositus shares with M. glenwoodensis plus the hairs on the inner surfaces of the hind basitarsi being yellow to red, similar sizes, and the pale brown medial to long white lateral hairs on the fifth and sixth sterna, M. lustrus bears a striking resemblance, and is more closely allied to, M. compositus. M. compositus can be separated from M. lustrus by the mesoscutal punctures positioned medially from the parapsidal line, which are mostly larger than the scutellar or mesepisternal punctures, and are mostly separated by less than, or equal to, one puncture diameter. In M. lustrus, these punctures are of irregular size, usually smaller than the majority of the mesepisternal or scutellar punctures, and mostly separated by more than one-half of a puncture diameter (several separated by more than, or equal to, two puncture diameters). Females can resemble M. plumosus because of the same vestiture coloration, but largely differ in the first tergum punctation (M. plumosus has punctures that exceed the basal one-half) and the galeal sculpting (M. plumosus has densely tessellate galeae whereas M. compositus has no tessellation).
Fig. ??. Map showing an estimation for the known distribution of M. (Callimelissodes) compositus. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Johnson, 2020; Carril et al., 2023; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; Gibbs, 2025; Bentley & Osborn, 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; Museum of Southwestern Biology, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026; University of Arizona Insect Collection, 2026a). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0. as applicable (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Callimelissodes) compositus. The x value is the month, and the y value is the number of documented observations occurring in that month. Data compiled from (Ikerd, 2019; Johnson, 2020; Carril et al., 2023; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; Gibbs, 2025; Bentley & Osborn, 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; Museum of Southwestern Biology, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026; University of Arizona Insect Collection, 2026a). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0. as applicable (see "Dataset Licenses").
Flower preference
Due to the few available flower records, a strict statement of oligolecty seems unfounded. However, with the current known data, it seems as though M. compositus may have a preference for Asteraceae (or historically known as Compositae) flowers (Laberge, 1961), but more reports are needed to be certain. All flower records included in this list are from reports in the literature or datasets. Each flower has a parenthesized reference listed after it, corresponding to the literary work or dataset in which it was recorded. Currently, M. compositus has been collected atop Aster sp. (Laberge, 1961), Gossypium thurberi (Laberge, 1961; old synonym), Grindelia sp. (Laberge, 1961), Grindelia squarrosa (Laberge, 1961), Haplopappus gracilis (Laberge, 1961).
Synonyms
Melissodes lupina var composita Tucker, 1909; Melissodes composita Tucker, 1909.
Melissodes dagosus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes dagosus
Melissodes dagosus is one of, if not the most, distinctive Melissodes in Oregon. This is due to the subgenus in which it resides (Apomelissodes, originally Tachymelissodes) has only one Oregonian representative (M. dagosus). So not only can species-specific characters be used to identify M. dagosus in Oregon, but entire subgeneric traits are viable as well. As the subgenus M. Tachymelissodes was absorbed into M. Apomelissodes (Freitas et al., 2023), and M. dagosus specifically was found to be more closely allied with M. Apomelissodes instead of M. Tachymelissodes (Wright, et al., 2020), subgeneric characters for the combination of the two subgenera don’t completely overlap anymore. Herein, when referring to the subgeneric characters of M. dagosus, these will instead be referencing those of M. Tachymelissodes rather than M. Apomelissodes for ease of use. Although this species is the most common in its original subgenus (M. Tachymelissodes), in general, M. dagosus seems as if it may be a rarer bee, especially compared to other M. Eumelissodes. Unexpectedly, M. dagosus actually ranks as the ninth most common species amongst the rest of the genus in Oregon, with a total of 32 records (Fig. 13). This species has been found across the South to Central Eastern areas in Oregon (Fig. ??), and given its inland presence in California and Arizona, it seems as though this may be the most westward of its range in Oregon. M. dagosus was originally thought to be active between the months of May and August with peak activity occurring in June and July (Laberge, 1956b). However, more recent collections suggest this same sentiment, except peak activity seems to more prominently occur in June (Fig. ??).
Field Markers
♂ A pale-colored bee, somewhat white to silvery in color, measuring to be 8-11 mm. in length. The underside of flagellar segments 2-11 and often the
apex of 1 are orange to yellow and the minimum length of the first flagellar segment is approximately more than, or equal to, one-half of the second
second segment's maximum length. The width of the penultimate flagellar segment is around two thirds its width, and in repose, the antennae reach the
basal area of the first tergum. The clypeal punctures then to be somewhat elongate (noticeably so basally), but smaller than those of the female.
The pubescent bands on terga 2-5 are positioned apically, approximately the same width across each respective terga (not widening or narrowing), and
approximately the same width as the other tergal bands. The clypeus is mostly pale yellow apart from the darkened spots denoting the tentorial
depressions and the labrum is mostly cream colored, but becomes dark brown marginally. The mandibular bases usually have triangular macula that are
often somewhat small (very rarely absent) and the wing veins are black to dark reddish brown. The head hairs are almost completely pale with only
a few brown hairs on the vertex occasionally and the thoracic hairs are mostly pale and only have a few brown hairs on the posteromedial mesoscutal
area and the medial scutellar area occasionally. The tergal vestiture description is as follows: The basal three-fifths of the first tergum bear
long white basal hairs. The basal pale pubescent band of the second tergum is white to silver-colored as is the distal band, and the distal band
is complete and usually medially reaches the apical margin. The interband zone of the second tergum bears short, subappressed, brown hairs. The
third and fourth terga are similar to that of the second, differing at the dark brown basal tomentum. The fifth tergum’s pale band is similar to
that of the fourth tergum. The sixth and seventh terga are covered in long pale hairs.
♀ Generally a paler bee (around the same color as those of M. plumosus), medium in size measuring 10-12 mm. in length. The underside of flagellar
segments 3-10 and often the apex of 2 are dark red to yellow and the eyes are greenish blue to blue. The clypeal punctures then to be somewhat
elongate (noticeably so basally) and the distal bands on terga 2-4 are approximately the same width across each terga (not narrowing or widening),
reach the apex across the entire tergum, and around as narrow, or narrower, than the basal dark pubescence. The scopal hairs have 3-6 branches on
either side of each rachis and the pygidial plate is somewhat V-shaped. The head hairs are mostly white to silver-colored with some brown hairs on
the vertex and labrum (in the darkest individuals, the dark hairs are dark brown; in the lightest individuals, these hairs are more ochraceous on
the labrum and lighter brown on the vertex). However, in dark specimens, the dark brown head hairs can also be found on the clypeus and mandibles
as well. The thoracic hairs are mostly pale yellow to white (entirely white on the propodeum) apart from the large dark brown posteromedial
mesoscutal patch of sparse hairs that reaches the medial areas of the tegulae, the medial often dark scutellar hairs, and usually the dark anterior,
ventral, and lateral mesepisternal hairs (these can be entirely pale). The tergal vestiture description is as follows: The basal three-fifths of
the first tergum bear long white basal hairs. The basal pale pubescent band of the second tergum is white to silver-colored as is the distal band,
and the distal band is often narrowly medially interrupted. The distal pale band is also separated from the apical margin, maximally in the medial
one-third and often less, by a narrowed area of simple brown short hairs when not worn. The interband zone of the second tergum bears short,
subappressed, brown hairs. The third and fourth terga are similar to that of the second, differing at the dark brown basal tomentum and the apically
positioned, uninterrupted distal pale pubescent bands (not separated from the apical margin by simple brown short hairs). The fifth and sixth terga
are mostly covered in long, dark brown, appressed hairs, except for paler individuals which have lateral ochraceous to white pale tufts of hairs.
Similar Species
As stated above, only a single species of M. Apomelissodes resides in Oregon, that being M. dagosus. Given how incredibly distinctive it is, unnecessary comparisons between species will be omitted and instead replaced by diagnostic characters for each sex. These traits can also be seen in both the descriptions and dichotomous keys, but are reiterated here for ease of use.
♂ The first flagellar segment is approximately more than, or equal to, one-half of the second second segment's maximum length and the bands on terga 2-5 are positioned apically. The only species that can somewhat resemble M. dagosus is M. rivalis due to a similarly lengthy F1, however, M. rivalis does not have tergal bands that reach the apex across the entire tergum. Furthermore, M. rivalis has a largely protuberant clypeus that protrudes beyond the eye by three-fourths of an eye's width in profile or more, whereas M. dagosus’ clypeus protrudes beyond the eye, but by less than three-fourths of an eye's width in profile.
♀ The second, third, and fourth terga have distal pale bands that reach the apex across the entire tergum, are approximately the same width across each terga (not narrowing or widening), are the same width to one another, and around as narrow, or narrower, than the basal dark pubescence on each tergum. The hairs of these bands do not arise from noticeable punctures.
Fig. ??. Map showing an estimation for the known distribution of M. (Apomelissodes) dagosus in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from ( Ikerd, 2019; Ikerd & Engler, 2023; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; The International Barcode of Life Consortium, 2026). Data licensed under CC-BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Apomelissodes) dagosus in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from ( Ikerd, 2019; Ikerd & Engler, 2023; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; The International Barcode of Life Consortium, 2026) and range-wide compiled from (Ikerd, 2019; Johnson, 2020; Carril et al., 2023; Ikerd & Engler, 2023; Gibbs, 2025; Bentley & Osborn, 2026; Brigham Young University, Arthropod Collection, 2026; Fägerström, 2026; Grinter et al., 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
A stark change from the rest of the genus, M. dagosus seems to be a highly polylectic species (Laberge, 1956b). This species has been found collecting both pollen and nectar from multiple families, including Asteraceae, but only on the genus Cirsium (thistles). More data is required to make a definitive statement on flower preference, but with the current known records, M. dagosus may be a generalist. All flower records included in this list are from reports in the literature or datasets. Each flower has a parenthesized reference listed after it, corresponding to the literary work or dataset in which it was recorded. Currently, M. dagosus has been collected atop Asclepias sp. (Laberge, 1956b), Cirsium sp. (Laberge, 1956b), Medicago sativa (Laberge, 1956b), Melilotus sp. (Laberge, 1956b), Melilotus officinalis (Laberge, 1956b; old synonym), Sisymbrium altissimum (Laberge, 1956b; old synonym), Sphaeralcea sp. (Laberge, 1956b), Thelypodium brachycarpum (Laberge, 1956b).
Synonyms
Melissodes dagosa Cockerell, 1909; Melissodes calloleuca Cockerell, 1924.
Melissodes druriellus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes druriellus
Melissodes druriellus, Drury's long-horned bee, is closely allied to M. pallidisignatus and one of the most variable Melissodes in the female sex. The males are most similar to M. pallidisignatus, but are distinctive in the opaque to deeply infumate apical tergal margins. Females are interesting given their variability across their entire range, and as none have yet to be found in Oregon but likely occur in this state, it's unknown what the general coloration in Oregon is. The rule presented by Laberge (1956a) states that Melissodes typically bear a paler vestiture in the Western and Southwestern areas of North America and a darker vestiture in the east. However, many of the examples used to support this were derived from subgenera that no longer reside within the genus (notably Epimelissodes), which calls into question the viability of this general rule for the modern-day genus. Also, many of the species-specific exceptions made to this statement belong to valid current-day subgenera (e.g. Eumelissodes and Melissodes s.s.) with species such as M. tepidus, an Oregonian resident, showing no real melanistic changes in its southern subspecies in comparison to its northern and eastern counterparts (Laberge, 1956a). With all of this being said, it’s currently unknown what color variations of the females of M. druriellus exist in Oregon, and as shown in Laberge (1961), many may exist within a similar region. Therefore, a general description of females will be provided below based mostly on sculptural characters with an emphasis that color variability is often common and isn’t necessary diognostic. Females are also most closely allied with M. pallidisignatus in Oregon due to the same shared dark mesoscutal hair patch, the impunctate shiny apical areas on the second tergum, the dull interband zone of the second tergum (highly contrasts with the shiny apical areas), and the abundant dark hairs on the tegulae and vertex. Females of M. druriellus are separated from those of M. pallidisignatus by the distal band on the third tergum, the rusty-reddish mesoscutal hairs on the areas anterior to the dark patch, and the sculpture of the first tergum. As stated above, M. druriellus has yet to be collected in Oregon, however, its current known range suggests that it may have a presence in this region (Fig. ??), and is therefore included in this guide. This species was historically believed to be active from May to November with peak activity occurring in August (Laberge, 1961) , and newer data seems to reflect this apart from a recent singular collection in April (Gibbs, 2025) implying that yearly activity commencement may start earlier than originally recognized.
Field Markers
♂ Generally a pale ochraceous to yellow bee with a yellow clypeus (marginally darkened) and gray, greenish yellow, or greenish blue eyes. The wing veins are reddish brown and the labrum is black with a mediobasal pale spot. The underside of flagellar segments 2-11 tend to be orange-colored and the apical areas of the terga are always brownish red. The length of the penultimate segment is usually less than, or equal to, one-third of its length and when in repose, but in some individuals, the penultimate flagellar segment’s length is greater than one-third of its width. When in repose, the flagella usually reach, or barely surpass the pterostigma, but in some individuals, this is not the case; widely variable. The first flagellar segment’s minimum length is approximately one-fourth to one-sixth of the second segment’s maximum length. The basal areas of terga 4-6 are covered in reddish brown pubescence and the mandibular bases do not have yellow maculations. Similarly to the females, the males also vary in color, but to a lesser extent. As stated by Laberge (1961), a similar format addressing variation in males is omitted due to many morphs occupying similar areas. Instead, a generic description, pertaining to all males, was given and will herein be what this setal description references. The hairs on the head are mostly yellow to pale ochraceous apart from the vertex, which usually has a few brown hairs. The thoracic hairs are mostly yellow to pale ochraceous as well, but the scutellar, tegular, and mesoscutal hairs are often brown. The tergal vestiture is as follows: the first tergum has long basal white to ochraceous hairs and long appressed to subappressed apical pale hairs that reach the apex across the tergum but don’t obscure the medial portion of the apical area (sometimes these hairs don’t obscure the apical margin more than medially); the second tergum has white basal pubescence and partially to entirely dark suberect, bristle-like hairs on the interband zone; the distal pubescent band of the second tergum is posteriorly separated from the apex of the tergum by about one-half to one times the length of the medial area of the pale band and is not interrupted medially; terga 3-5 are similar to that of the second tergum except for the interband zones, which in addition to the bristle-like hairs, they often bear delicate, sparse, white, appressed pubescence; the distal bands of terga 3-4 become closer to the apical margin with each tergum and the basal tomentum resembles a dark chocolate color; the fourth tergum’s distal pale band also tends to be medially interrupted and the fifth tergum’s distal pale band is usually absent altogether; the sixth and seventh terga are covered in dark brown hairs apart from the lateral pale tufts.
♀ Highly variable in vestiture coloration, most often a somewhat pale bee with rusty red hairs anterior to the dark mesoscutal patch. The eyes are greenish blue to gray and the wing veins are black to dark brown. The underside of flagellar segments 3-10 are rufescent and the apical areas of terga 1-3 are black to dark brownish red. The terga do not have their pubescent bands complete but instead they are lateral fasciae, or sometimes entirely absent. If one of the tergal bands is complete, then it’s the second tergum’s band and the hairs on the lower lateral mesepisternal surfaces are dark brown; or the terga have their pubescent bands complete on more than just one tergum. If however there is just one complete band on a single tergum, then it’s either not on the first tergum, or the hairs on the lower lateral mesepisternal surfaces aren’t dark. This is dependent on the morph. The distal band on the third tergum is separated from the tergum’s apical margin and The first tergum is densely reticularly shagreened and the punctures are incredibly shallow, large, and crowded. These punctures are usually obscured by the shagreening. The apical areas of the second tergum tend to be shiny and the interband zone tends to be dulled, highly contrasting to the apical area. The clypeus tends to be non-protuberant, in profile, only protruding beyond the eye by usually less than one-half of an eye’s width.
Due to its variability, an entire setal description along with its modifiers for melanism seems appropriate. The description below is derived and expanded from that of Laberge (1961) and represents the palest morph; the starting point for further melanistic additions. The head hairs are mostly ochraceous to white apart from the vertex, which bears brown hairs. The thoracic hairs are ochraceous to white, usually a pale rusty-red color anterior of the dark mesoscutal patch, apart from the large mesoscutal dark patch that’s often larger, or at least as large as, the scutellar dark patch. The first tergum’s basal three-fifths or more bear ochraceous to yellow hairs. The basal pubescent band on the second tergum is pale ochraceous to white and the interband zone has mostly black, suberect hairs. The distal pale pubescent band of the second tergum is often narrowly medially interrupted, does not reach the apical margin laterally, and takes on a yellow to ochraceous hue. The apical areas of the second tergum located near the distal pale band tend to bear short, sparse, subappressed hairs laterally. The third tergum is similar to that of the second, but the basal tomentum is dark brown and dense, and the distal pale pubescent band is uninterrupted. The fourth tergum’s distal pale pubescent band reaches the apex across the entire tergum, is narrower than the basal dark tomentum, and is complete. The fifth and sixth terga are mostly covered in dark hairs, but laterally, small tufts of pale hairs are present. The leg hairs are mostly pale ochraceous, differing at the fore and middle tarsi, which are dark brown; the scopal hairs, which are often yellow; the inner surfaces of the hind basitarsi and hind tibiae, which are dark brown; the basitibial plate, which is brown; the outer surfaces of the fore and middle tibiae, which are dark brown; and the tibiae as well as the upper areas of the hind femora sometimes, which are also dark brown. In general, the darkest females tend to be almost entirely black to dark brown with a few pale hairs on the periphery of the mesoscutum, the wing bases, and the pronotal lobes. Along with this, the scopae also tend to be yellow.
Many levels of melanism seem to exist within females of M. druriellus, and these differences are gradual rather than stark. As the geographical variation in melanism is unknown for M. druriellus females in Oregon, all possible morphs will be accounted for. In his treatment, Laberge (1961) categorized three (presented as a, b, and c) “levels” of melanism across 8 morphological features (e.g. tergal band color, leg hair color, ect.). This resulted in a list allowing for variation accountability rather than writing singular descriptions for each theoretical variety (6, 561 or 3^8 possible morphs). A similar structure derived from Laberge’s (1961) findings will be presented here as a table. The table below lists eight characters along with three melanistic levles per character that can vary from the palest description (table 3). These traits are meant to be applied to, and override portions of, the pale descriptions above. Note that each value per character (light, intermediate, or dark) is independent from one another. Therefore, a female M. druriellus may have no brown pubescence on the fourth tergum (palest choice for the first character), and also have all dark hairs on the legs except for the scopal hairs and sometimes the anterior femoral surfaces (darkest choice for the eighth character).
Table 3. Eight characters pertaining to the vestiture of the females of M. druriellus, each with three different melanistic variations. This table accounts for all possible morphs of the females of M. druriellus when the characters are either added to, or replace, portions of the base-line pale description.
| Character | Light | Intermediate | Dark |
|---|---|---|---|
| The distal pubescent band on the fourth tergum | There is no medial brown pubescence. | There is brown pubescence present apicomedially or up to 50% of the band is brown. | More than 50% of the entire band is brown, or the entire band is completely brown. |
| The distal pubescent band on the third tergum | The band is complete without a medial interruption. | The band is medially interrupted by less than one-third of the tergum’s width. | The band ranges from being medially interrupted by one third of the tergum’s width, to being completely dark brown. |
| The distal pubescent band on the second tergum | The band is medially interrupted, but only narrowly so. Medial interruption less than one-fifth of the tergum’s width. | The band is widely medially interrupted by approximately one-fifth to one-half of the tergum’s width. | The band is either widely medially interrupted by approximately one-half of the tergum’s width, or the band is completely dark brown. |
| The hairs on the anterior surface of the first tergum | The hairs are completely pale. | The hairs are pale and range from having a few dark brown hairs to 50% of the hairs being dark brown. | Over 50% of the hairs are dark brown, or the hairs are completely dark brown. |
| The dark patch of hairs on the mesoscutum | The patch doesn’t reach the anterior tegular margins, the hairs positioned medially of the tegulae are mostly pale, and the pronotal hairs are mostly pale. | The patch reaches or surpasses the anterior tegular margins, the hairs positioned medially of the tegulae are mostly pale, but there may be a few dark hairs present, and the pronotal hairs are entirely, or almost entirely pale. | The patch reaches, or almost reaches, the pronotum medially, the hairs positioned medially of the tegulae are entirely dark, and the pronotal hairs are entirely, or almost entirely, dark. |
| The hairs on the mesepisterna. | The hairs are mostly pale with a few pale brown ventral hairs. | The hairs are brown anteriorly, ventrally, and on the lower lateral surfaces, but less than 50% of the lateral surfaces. | The hairs are dark brown on more than, or equal to, 50% of the lateral surfaces. |
| The hairs on the head | The hairs are entirely pale apart from the vertex, which has many dark hairs. | The hairs are pale apart from the vertex, clypeus, and labrum, which have dark brown hairs. However, the hairs on the clypeus are only partially dark brown, but often mostly pale. | The hairs are almost completely dark brown (a few pale hairs may exist), including the vertex, clypeus, and labrum. |
| The hairs on the legs | The hairs are almost entirely pale on the trochanters, femora, coxae, and outer surfaces of the tibiae. | The hairs on the trochanters, coxae, tibiae, and inner surfaces of the femora, as well as portions of the outer surfaces of the femora, are brown. | The hairs are entirely black to dark brown apart from the scopal hairs and sometimes the anterior femoral surfaces. |
Similar Species
Males and females of Melissodes druriellus are highly variable if not apparent from their descriptions. Females can resemble a number of different species because of how wide their setal coloration ranges. Males are interesting in that their structure as well as their vestiture coloration varies, but even with this taken into consideration, they are still highly distinctive. As can be seen in the key above (couplet 18), males of M. druriellus are split into two separate groups: 1) the males with the length of their penultimate flagellar segment equal to, or less than, three times its width and the antennae does not surpass the pterostigma in repose; 2) males with the length of their penultimate flagellar segment more than three times its length and/or the antennae surpass the pterostigma in repose. For this reason, two male treatments will be given. Given how the coloration and structure of the female vestiture is rather gradual than abrupt, two treatments for dark and light morphs (as seen in the M. bimatris treatment) wouldn’t suffice. Instead, a single comparison highlighting diagnostic characteristics and a few species of high resemblance based on a given morph will be presented.
♂ (1) This grouping of M. druriellus males are most similar to those of M. pallidisignatus. Similar to the males of M. druriellus, M. pallidisignatus have two separate groups as well, however these fall under the same umbrella as group one of M. druriellus males. That is, that the length of the penultimate flagellar segment is less than three times its width (maximum length and narrowest width) and the antennae does not surpass the pterostigma in repose. The groups of M. pallidisignatus are as follows: 1) the mandibular bases have yellow macula; 2) the mandibular bases are entirely black. Group one of M. druriellus males differs from group one of M. pallidisignatus males in that the mandibular bases are entirely black, and differs from both groups of M. pallidisignatus males by the dark apical tergal areas (M. pallidisignatus’ tergal areas are colorless to testaceous or brownish yellow hyaline).
♂ (2) This grouping of M. druriellus males aren’t specifically similar to any single species and are highly distinc in Oregon. This is because M. druriellus males are the only Oregonian resident within Eumelissodes to have opaque brown-red apical tergal margins.
♀ Superizingly, females often only resemble one or two other species in Oregon, but seem to be most closely allied with those of M. pallidisignatus. M. druriellus can be separated by the distal band on the third tergum, which does not reach the apex across the entire tergum and is instead well-separated from it (on M. pallidisignatus, this band usually reaches the apex of the tergum laterally); the pale hairs on the mesoscutum positioned anteriorly to the dark patch of hairs, which are usually rusty-reddish (often pale ochraceous to white in M. pallidisignatus); the first tergum, which is densely reticularly shagreened and the punctures are incredibly shallow, large, and crowded (the punctures are distinct in M. pallidisignatus). In its darkest form, M. druriellus can also resemble M. pullatellus. The two differ significantly in their scopal hair coloration. That being in M. druriellus, the scopal hairs are often yellow to golden, whereas in M. pullatellus, these hairs are dark brown to black.
Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) druriellus. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Paiero & Paiero, 2017; Ikerd, 2019; Johnson, 2019; Telfer, 2019; Johnson, 2020; Archbold Biological Station, 2023; Carril et al., 2023; Hardy & McFarland, 2023; Hardy et al., 2023; Ikerd & Engler, 2023; Purdue Entomological Research Collection, 2023; South Dakota State University, Severin-McDaniel Insect Research Collection, 2023; Texas A&M University Insect Collection, 2023; Jacobson et al., 2024; López-Uribe Lab, 2024; Mazurowski & Hardy, 2024; North Carolina State University Insect Collection, 2024; Ricketts et al., 2024; Veit & McFarland, 2024; Droege & Maffei, 2025; European Bioinformatics Institute (EMBL-EBI), GBIF Helpdesk, 2025; Gibbs, 2025; Grant et al., 2025; Hardy, 2025; Hardy et al., 2025; Holm et al., 2025; Hughes et al., 2025; McKenna & Zitani, 2025; New Brunswick Museum, 2025; Richardson & Hardy, 2025; Roberts, 2025; Scott, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Clemson University Arthropod Collection, 2026; Connecticut Agricultural Experiment Station, 2026; Frost Entomological Museum, 2026; Gross & Oboyski, 2026; Hardy, 2026; Harvard & Morris, 2026; Illinois Natural History Survey, 2026; István, 2026; Johnson, 2026; Kelly et al., 2026; Luther College, 2026; Mertz et al., 2026; Mississippi Entomological Museum, 2026; Motz, 2026; MT James Entomological Collection, Washington State University, 2026; Natural History Museum, 2026; Northern Arizona University, 2026a; Oram, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026; UMMZ & LSA, 2026; University of Minnesota Insect Collection, 2026; Vermont Agricultural and Environmental Laboratory, 2026; Virginia Tech Insect Collection, 2026; Wild, 2026; York University, n.d.a; York University n.d.b; York University, n.d.c). Data licensed under CC0 1.0, CC BY 4.0, and CC BY-NC 4.0 as applicable (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Eumelissodes) druriellus. The x value is the month, and the y value is the number of documented observations occurring in that month. Data compiled from (Paiero & Paiero, 2017; Ikerd, 2019; Johnson, 2019; Telfer, 2019; Johnson, 2020; Archbold Biological Station, 2023; Carril et al., 2023; Hardy & McFarland, 2023; Hardy et al., 2023; Ikerd & Engler, 2023; Purdue Entomological Research Collection, 2023; South Dakota State University, Severin-McDaniel Insect Research Collection, 2023; Texas A&M University Insect Collection, 2023; Jacobson et al., 2024; López-Uribe Lab, 2024; Mazurowski & Hardy, 2024; North Carolina State University Insect Collection, 2024; Ricketts et al., 2024; Veit & McFarland, 2024; Droege & Maffei, 2025; European Bioinformatics Institute (EMBL-EBI), GBIF Helpdesk, 2025; Gibbs, 2025; Grant et al., 2025; Hardy, 2025; Hardy et al., 2025; Holm et al., 2025; Hughes et al., 2025; McKenna & Zitani, 2025; New Brunswick Museum, 2025; Richardson & Hardy, 2025; Roberts, 2025; Scott, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Clemson University Arthropod Collection, 2026; Connecticut Agricultural Experiment Station, 2026; Frost Entomological Museum, 2026; Gross & Oboyski, 2026; Hardy, 2026; Harvard & Morris, 2026; Illinois Natural History Survey, 2026; István, 2026; Johnson, 2026; Kelly et al., 2026; Luther College, 2026; Mertz et al., 2026; Mississippi Entomological Museum, 2026; Motz, 2026; MT James Entomological Collection, Washington State University, 2026; Natural History Museum, 2026; Northern Arizona University, 2026a; Oram, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026; UMMZ & LSA, 2026; University of Minnesota Insect Collection, 2026; Vermont Agricultural and Environmental Laboratory, 2026; Virginia Tech Insect Collection, 2026; Wild, 2026; York University, n.d.a; York University n.d.b; York University, n.d.c). Data licensed under CC0 1.0, CC BY 4.0, and CC BY-NC 4.0 as applicable (see "Dataset Licenses").
Flower preference
M. druriellus is a known oligolege of Asteraceae, and according to the flower records presented by Laberge (1961) M. druriellus seems to have a strong preference toward the genera Aster, Solidago, Gindeliae, and Aplopappus. However, in Western areas (likely including Oregon), M. druriellus seems to prefer Solidago (Laberge, 1961). According to Laberge (1961), of the 420 M. druriellus collected with floral accounts, 130 were found atop Solidago, 109 were found atop aster, 51 were found atop Grindelia, and 28 were found atop Aplopappus; the rest were collected across scattered genera with no substantial values. This is in agreement to the statement made above, but interestingly, Aster seems to be a very close second behind Solidago. Given how close Solidago and Aster are in preference, two percentages will be given. This data renders Solidago to be favored 30.95% of the time, and Aster to be favored 25.95% of the time by M. druriellus.
Synonyms
Apis druriella Kirby, 1802; Macrocera rustica Say, 1837; Melissodes rustica Say, 1837; Melissodes assimilis Smith, 1879; Melissodes ambigua Smith, 1879; Melissodes festonata Provancher, 1888; Melissodes simillima Robertson, 1897; Melissodes asteris Robertson, 1914; Melissodes druriella Kirby, 1802.
Melissodes glenwoodensis

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes glenwoodensis
Melissodes glenwoodensis is a quite distinctive bee, in part due to it being the largest of its genus in Oregon. Interestingly, both males and females are allied with M. lustrus, and to an extent M. composistus, but males share a more similar antennal structure to that of M. lupinus and M. nigracauda than the foregoing species. The males are distinctive in that flagellar segments 5-9 (sometimes on then, never on 11) have shiny depressions, the apical areas of the second and third terga are glabrous as well as impunctate, and the galeae are either shiny or dull due to shagreening. Females are distinctive in that the basal crowded punctures on the first tergum does not surpass the basal one-third, the hairs on the inner surfaces of the hind basitarsi are black to dark reddish brown and the scopal hairs are usually yellow, and the fifth and sixth terga have black to dark brown hairs or no white lateral tufts. Although, as stated above, males are similar to M. lupinus, females seem to not partake in this allyship. Rather, the two species in the female sex differ greatly from one in other by the ultimate flagellar segment structure. M. glenwoodensis has been found in the Northeastern and Southwestern areas of Oregon and given how widespread these collections are, likely occurs across the entire state (Fig. ??). A total of five records comprise its Oregonian range, placing this species as the fifth rarest within the genus for this region. M. glenwoodensis was historically presumed to be active between the months of July and November, but most chiefly so in August and September (Laberge, 1961). New found data currently suggests that this species is active between the months of May and November, with September noticeably out-performing August in terms of abundance (Fig. ??).
Field Markers
♂ A paler bee with darker wing veins (darker than those of M. agilis) and somewhat similar to M. lupinus and M. compositus. The clypeus, mandibular bases,
and labrum are yellow, and the labrum isn’t marginally darkened. The eyes are often grayish green to green and the underside flagellar segments 2-11 are red
to yellow (these segments are dark brown on top). The wing veins are black to brown and terga 2-5 are sometimes yellow hyaline apically, but usually opaque
brown. The first flagellar segment’s minimum length is approximately equal to one-fourth of the second segment’s maximum length, and segments 5-10 (sometimes
also on the apex of 4) have shiny, shallow, longitudinal dorsolateral depressions. The length of the penultimate segments is more than twice, but less than
three times, its width and the ultimate segment’s length is approximately three times its width. The apical areas of the second and third terga are glabrous
as well as impunctate and the galeae are either shiny or dull due to shagreening. The fourth sternum is marginally convex, and the fifth and third sterna are
very feebly convex marginally, almost transverse. Males tend not to vary in setal coloration, the description of which follows: the head hairs are mostly
dark ochraceous to pale ochraceous apart from the vertex, which often has some darker hairs; the thoracic hairs are also mostly dark ochraceous to pale
ochraceous apart from the dorsal areas, which tend to bear darker hairs; the tergal hairs are mostly white, with the second, third, and fourth terga having
glabrous apical areas, the fifth tergum, which is separated from the apex across the entire tergum, and the sixth and seventh terga, which have their surfaces
largely covered in dark brown hairs; the sternal hairs are usually medially brown.
♀ Generally a large bee (largest individuales in Oregon are of this species) covered in pale hairs with very striking banding. Two color variations are
present in the female sex (light and dark), but the differences between the two are somewhat minute. Herein, this description accounts for all variability.
The eyes are greenish blue to gray and the wing membranes are faintly infumate. The wing veins are black to dark brown and the underside of flagellar
segments 3-10 are rufescent. The apical one-half of the mandibles are rufescent and the apical areas of terga 1-3 are black to dark brownish red. The first
tergum’s crowded basal punctures are restricted to, and do not surpass, the basal third or less medially. If the medial one-third of the first tergum has
punctures, then they are mostly separated by more than two puncture diameters. The galeae are moderately shiny above, and delicately reticularly shagreened,
but not densely tessellated. The hairs on the inner surfaces of the hind basitarsi are black to dark reddish brown and the scopal hairs are usually yellow.
The tergal vestiture is as follows: the basal and extreme lateral areas of the first tergum bear pale ochraceous to white hairs; the basal pale pubescent band
on the second tergum is white and complete and the distal pale band on is instead is reduced to white, narrow, oblique, lateral, fasciae that are laterally
spaced from the apex of the tergum, minimally by the length of the fascia at that area; in width, the fasciae of the second tergum are somewhat wider than the
apubescent medial portion of the tergum; the basal tomentum of the third tergum is dark brown, the interband zone bears scattered white to brown pubescence
(these hairs often become browner near the basal area), and the distal pale pubescent band does not reach the apical margin across the entire tergum; the fourth
tergum is similar to that of the third, differing at the distal pubescent band, which is complete and reaches the apex across the entire tergum; the fifth and
sixth terga are mostly covered in black to dark brown hairs apart from the lateral long pale ochraceous to white lateral tufts of hair; sometimes these tufts
are absent on the sixth tergum.
Similar Species
As specified above, M. glenwoodensis is most similar to M. compositus and M. lustrus in the female sex due to the shared sculpturing on the first tergum. The males on the other hand seem to be related to the foregoing species, but most closely allied to M. lupinus males due to the shared antennal structure. As with most Melissodes, the males of this species can superficially resemble a number of others due to the shared vestiture coloration. However, as they are within the subgenus Callimelissodes, males can quickly be separated from any other subgenus in that the fourth sternum is marginally convex rather than transverse. Females are distinctive in the sculpture of the first tergum (this trait is only shared amongst 3 females in Oregon) and the setal coloration on the inner surfaces of the hind basitarsi.
♂ Similarly to the females, M. glenwoodensis males are similar to those of M. lustrus and M. compositus. However, in the male sex, this species vastly differs from both M. lustrus and M. compositus due to the lack of flagellar depressions on F4 and F11 (see couplet 3 in the male key). For this reason, M. glenwoodensis males more strongly resemble those of M. lupinus. Males can be separated from M. lupinus by the apical areas of the second and third terga, which are glabrous as well as impunctate; and the galeae, which are either shiny or dull due to shagreening. In contrast, the apical areas of T2 and T3 on M. lupinus bear suberect hairs and the galeae are often dull due to dense shagreening. Due to the distinctive flagellar segmentation and subgeneric characteristics (the structure of S4), M. glenwoodensis does not directly resemble any other Melissodes in Oregon.
♀ Females of M. glenwoodensis are most similar to M. compositus and M. lustrus, but most likely the latter more so than the former. These species form somewhat of a “morphological clade” in that no other Melissodes share their characteristic of the crowded basal punctures on T1 no surpassing the basal one-third or less medially. Interestingly, within this grouping, M. lustrus seems to be more closely allied to M. compositus than M. glenwoodensis and vice versa. Hence, M. glenwoodensis is separated from both these species due to the hairs on the inner surfaces of the hind basitarsi being black to dark reddish brown (highly contrasting with the scopal hairs) and the scopal hairs being usually yellow (see couplet 14 of the female key). In contrast, M. compositus and M. lustrus have red to yellow hairs on the inner surfaces of the hind basitarsi (not highly contrasting with the scopal hairs) and the scopae are often more pale ochraceous to white. On top of this, females further differ from both these species due to their size. M. glenwoodensis ranges between 11-14 mm. in length, whereas M. compositus and M. lustrus range between 9-12 mm. in length; M. compositus often does not surpass 11 mm.
Fig. ??. Map showing an estimation for the known distribution of M. (Callimelissodes) glenwoodensis in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; The International Barcode of Life Consortium, 2026). Data licensed under CC-BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Callimelissodes) glenwoodensis in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Ikerd, 2019; Johnson, 2020; Carril et al., 2023; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; Droege & Maffei, 2025; Scott, 2025; Zhuang, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Brigham Young University, Arthropod Collection, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; New Mexico State Collection of Arthropods, 2026; Northern Arizona University, 2026a; Orrell & Informatics, 2026a; Orrell & Informatics, 2026b; The International Barcode of Life Consortium, 2026; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
M. glenwoodensis is apparently an oligolect of Asteraceae, and according to the flower records presented by Laberge (1961), This seems to have a strong preference toward the genus Chrysothamnus. According to Laberge (1961), a total of 146 M. glenwoodensis collections with floral data available have been accounted for. These bees were collected across 29 dedicated searches, 20 of which all bees were found atop Chrysothamnus (Laberge, 1961). Assuming all collections resulted in a similar number of specimens, this renders Chrysothamnus to be favored 68.96% of the time by M. glenwoodensis. However, for this to be true, a possibly inaccurate assumption must be made given the numerical data is unpublished, so this is just a speculatory estimation.
Synonyms
None.
Melissodes grindeliae

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes grindeliae
Melissodes grindeliae is closely allied to M. druriellus and M. pallidisignatus, but females differ largely in the tergal and scopal hair coloration, and males differ in the wider tergal bands and shorter F1. Similarly to those of M. druriellus and M. pallidisignatus, males of M. grindeliae have two seperate groups that it can be classified into. This is interesting as all species within the druriellus group (M. druriellus, M. pallidisignatus, and M. grindeliae) both occur or likely occur in Oregon, and share this same structural and integumental binomial variation in the male sex. Although just speculatory, this is possibly a result of a shared similar ancestor that originally resided in Oregon, from which these species diverged. Males of M. grindeliae are distinctive in two manners: group 1) The length of the penultimate flagellar segment is less than three times its width (maximum length and narrowest width) and the antennae reach but do not surpass the pterostigma in repose; the labrum and the mandibles are black without yellow maculations. Group 2) The first flagellar egment’s maximum length is more than one-third of the third segment’s minimum length; the mesoscutal and scutellar hairs are abundantly dark medially. Females are distinctive in that the tergal bands are often yellow to ochraceous, predominantly so on T3 and T4; the scopal hairs are often ochraceous; the distal band on T3 reaches the apex laterally; and the second flagellar segment’s length is less than or equal to its width. Counter intuitively, M. grindeliae (although an Asteraceae oligolege) does not specialize on, and isn’t restricted to, flowers from the genus Gindelia. In fact, this genera seems to make up a very small portion of its floral data (see more in “Flower Preferance”). This species has yet to be found in Oregon, but given the estimated distribution map from Laberge (1961), and more recent distribution data (Fig. ??), M. grindeliae likely occurs in Oregon. Furthermore, given its coastal presence in California coupled with abundant inland documentations (as well as the Washington record from Laberge, 1961; omitted in Fig. ?? due to unknown coordinates), it's probable that M. grindeliae is present across the entirety of Oregon. This species seems to be active between the months of June and September, with peak activity occurring in July, an observation presented by Laberge (1961) and further supported in Fig. ??.
Field Markers
♂ A pale bee with darker hairs on the dorsal thoracic areas and hyaline tergal margins. The clypeus is mostly yellow apart from the piceous to rufescent apical
margin and the eyes are yellowish brown to green. The wing membranes are colorless hyaline to faintly infumate and the wing veins are dark brown to reddish brown.
The minimum length of the first flagellar segment is approximately one-third of the second segment’s maximum length, and the length of the penultimate flagellar
segment is less than, or equal to, three times its width. When in repose, the flagella reach, or almost reach, the pterostigma. The labrum and the mandibles are
black and have no yellow maculations. Two variations of M. grindeliae males are present, some where the length of the penultimate flagellar segment is less than
three times its width (maximum length and narrowest width) and the antennae does not surpass the pterostigma in repose, and others the opposite (see couplet 18
in the male key). However, if an individual presents the characteristics of the latter, the hairs on the mesoscutum always have a dark patch. In the case of the
former, the mesoscutal hairs can be entirely pale or have a dark patch. The tergal vestiture is as follows: the hairs on the first tergum are entirely ochraceous;
the basal area of the second tergum bears pale ochraceous hairs and the interband zone is covered with erect and partially pale hairs; the second tergum’s distal
pale pubescent band is faintly yellow to ochraceous, laterally connected to the basal pale band by pale hairs, often uninterrupted and becoming medially narrowed,
and usually reaches the apical margin at extreme lateral areas; when unworn, the apical area of the second tergum bears many suberect, often brown, and long hairs
(longer than that of the female). The third and fourth terga are similar to that of the second apart from the interband zones, which have entirely to partially
brown hairs; and the distal pale pubescent bands, which reach the apical margin, minimally laterally and sometimes more; the fourth tergum’s distal pubescent
band also usually reaches the apex across the entire tergum. The fifth tergum is similar to that of the fourth, but the apical area is entirely obscured by the
distal band; the sixth and seventh terga are basally covered in brown hairs, but apically, these hairs become yellow.
♀ Generally a pale bee with a distinct large dark patch of hairs on the mesoscutum and scutellum (somewhat reminiscent of M. druriellus). The eyes are greenish
gray to blue and the underside of flagellar segments 3-10 are rufescent (very noticeably so on the distal most three or four segments). The wing veins are black
to brown and the apical margin of the first tergum is very narrowly hyaline. The interband zone of the second tergum is impuncate or essentially so and has erect
to suberect dark hairs. The hairs on the inner surfaces of the hind basitarsi are black to dark reddish brown. The apical areas of the second, third, and fourth
terga are piceous to opaque and dark brown. The length of the second flagellar segment is less than, or equal to, its width. The head hairs are mostly pale
ochraceous to white apart from the vertex, which has abundant dark brown hairs. The thoracic hairs are pale ochraceous to somewhat yellowish except as follows:
the lateral thoracic areas are pale ochraceous to white, but the mesepisternal hairs are sometimes brown on the lower lateral, ventral, and anterior surfaces;
the scutellar hairs are dark brown apart from the pale hairs on its periphery; the mesoscutum has a posteromedal dark patch of hairs that reaches the anterior
tegular margins and often surpasses them. The tergal vestiture is generally yellow to ochraceous (notably so on T3 and T4) and the descriptions are as follows:
the basal area of the first tergum bears yellowish to ochraceous hairs; the basal area of the second tergum bears pale ochraceous hairs and the interband zone is
covered with dark brown pubescence; the second tergum’s distal pale pubescent band is faintly yellow to ochraceous, laterally connected to the basal pale band by
pale hairs, often uninterrupted and becoming medially narrowed, and usually reaches the apical margin at extreme lateral areas; when unworn, the apical area of the
second tergum bears many brown to ochraceous, comparably simple, suberect, long hairs; the third tergum is similar to that of the second tergum apart from the
distal band, which reaches the apical margin laterally and is faintly yellow-colored, and the basal tomentum, which is dark brown; the fourth tergum’s distal pale
band reaches the apex across the entire tergum, is complete, often vibrant yellowish to near orange, and doesn't noticeably widen or narrow; the fifth and sixth
tergum are mostly covered in dark hairs apart from the lateral tufts, which are white.
Similar Species
As shown in the group name and the overview, M. grindeliae are similar to M. druriellus and M. pallidisignatus in both sexes. In males, two groups of M. grindeliae (described above) exist, each one more closely allied with one species than another. In group 1, the males of M. grindeliae are most similar to both M. pallidisignatus and the first grouping of M. druriellus (see grouping in respective species description) due to the shared characteristics of the length of the penultimate flagellar segment being less than three times its width (maximum length and narrowest width) and the antennae not surpassing the pterostigma in repose. However, in group two of M. grindeliae, these individuals seem to be more closely allied with group two of M. druriellus. This is as a result of the shared characteristic pertaining to the length of the penultimate flagellar segment being equal to or more than, three times its width (maximum length and narrowest width). Taking into account these two groups, two treatments will be listed for the males of M. grindeliae in this similar species section. Females, unlike males, have only one distinct form and most strongly resemble M. pallidisignatus, and lesser so M. microstictus and M. druriellus.
♂ (1) Males of this group are distinctive from both M. druriellus and M. pallidisignatus in that the labrum is entirely black with no basomedial yellow macula. Also, M. grindeliae can be separated from the foregoing species in that the antennae reach the pterostigma in repose instead of falling short. Males of M. grindeliae also have their tergal margins hyaline rather than opaque, largely separating them from any individuals of M. druriellus.
♂ (2) Males of this group are related to M. druriellus, but probably more closely so to M. menuachus and M. robustior. M. grindeliae, just like in group one, are easily separated from those of M. druriellus by the hyaline tergal margins and the longer first flagellar segment with the minimum length measure one-third of the second segment’s maximum length. In contrast, M. druriellus has opaque brown to reddish brown tergal margins and the first flagellar segment’s minimum length is approximately one-fourth to one-sixth of the second segment’s maximum length. M. grindelae males are similar to those of M. robustior and M. menuachus in that the first flagellar segment’s maximum length is more than one-third of the third segment’s minimum length, and the first tergum doesn't have an obvious apical band of white, dense, hairs that obscure the apical margin medially. However, M. grindeliae can be separated from M. robustior in that the galeae are shiny and not shagreened as opposed to being dull and densely shagreened. Males can also be separated from M. menuachus in that the labrum and the bases of the mandibles have no pale maculations and the mesoscutal and scutellar hairs are abundantly dark medially. In contrast, the labrum and bases of the mandibles are yellow in M. menuachus and the mesoscutal and scutellar hairs aren’t dark medially.
♀ The females of M. grindeliae can resemble both M. druriellus and M. pallidisignatus, but more so the latter due to the pale hairs anterior to the dark mesoscutal patch. Females can also, to an extent, somewhat resemble those of M. microstictus although less strongly. M. grindeliae can be separated from those of M. druriellus by the distal pale band of the third tergum that reaches the apex of the tergum laterally as opposed to being largely separated from the apical margin and by the pale hairs anterior to the mesoscutal dark patch. Females can be separated from those of M. pallidisignatus by the impunctate, or essentially so, interband zone of T2 that bears erect to suberect dark hairs and the hairs on the inner surfaces of the hind basitarsi that are black to dark reddish brown. As stated above, although not strongly related to this species, M. grindeliae can bear a resemblance to M. microstictus due to shared vesistural coloration, but differ as follows: the underside of flagellar segments 3-10 are rufescent, and very noticeably so on the distal most three or four segments; the distal pale band of the second tergum usually isn’t medially interrupted; and the hairs on the inner surfaces of the hind basitarsi are dark brown to reddish brown.
-Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) grindeliae. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Johnson, 2020; Carril et al., 2023; South Dakota State University, Severin-McDaniel Insect Research Collection, 2023; Droege & Maffei, 2025; Scott, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026; University of Arizona Insect Collection, 2026a; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
-Fig. ??. A graph representing the phenology of M. (Eumelissodes) grindeliae. The x value is the month, and the y value is the number of documented observations occurring in that month. Data compiled from (Ikerd, 2019; Johnson, 2020; Carril et al., 2023; South Dakota State University, Severin-McDaniel Insect Research Collection, 2023; Droege & Maffei, 2025; Scott, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026; University of Arizona Insect Collection, 2026a; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
Although its name suggests oligolecty on the genus of flowers Grindelia, M. grindeliae seems to instead favor Chrysopsis and Ratibida, the two combined accounting for over half of the current floral data records. Out of the 271 M. grindeliae records with annotations of which flower it was collected atop, Chrysopsis ranked number one with a total of 86 collections and Ratibida ranked number two with a total of 62 collections. This renders Chrysopsis to be favored 31.73% of the time and Ratibida 22.87% of the time by M. grindeliae. The remainder of the floral associations seem to show no other notable abundances, suggesting that these two genera are of high importance to M. grindeliae.
Synonyms
None.
Melissodes lupinus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes lupinus
Melissodes lupinus is an extremely common Oregonian species widespread across the entire state. Males seem to be most closely allied with those of M. glenwoodensis due to the shared antennal structure, but are distinctive in the sculpturing of the galeae, and the apical areas of the second and third terga bearing suberect hairs. Females on the other hand aren’t directly allied with any single other species and are distinct in the short truncated penultimate flagellar segment. The characters listed for both sexes, render M. lupinus as one of the most distinctive and easily identified Melissodes in Oregon under close examination. Given its range-wide abundance, it's no surprise that this species ranks high within the Oregonian commonality chart. Even with this taken into account, Eumelissodes in general tend to be more common than Callimelissodes, but unexpectedly, it seems that M. lupinus ranks number 1 with a total of 374 publicly available records, surpassing M. microstictus (the most common Oregonian Eumelissodes) by over 170 records. This, paired with its morphology, render M. lupinus one of the most notable, and somewhat outlying species in Oregon. Although the name suggests otherwise, this species has shown an overwhelming preference towards plants in the family Asteraceae rather than Fabaceae (the family in which lupin resides). M. lupinus seems to be active from March to October with peak activity occurring in June, July, and August.
Field Markers
♂ Paler vestiture with a few darker hairs on the dorsum of the thorax and vertex of the head. The mandibular bases, clypeus, and labrum, are yellow
without a darkened labral margin. The eyes are bluish gray to gray, and on rare occasions, greenish blue and the underside flagellar segments 2-11 are
red to yellow (these segments are dark brown on top). The wing veins are black to brown and terga 2-5 are sometimes yellow hyaline apically, but
usually opaque brown. The first flagellar segment’s minimum length is less than one-fifth of the second segment’s maximum length, and segments 5-9
(sometimes also on 10, never on 11) have shiny, shallow, longitudinal dorsolateral depressions. The length of the penultimate segments is more than
twice its width and the ultimate segment’s length is more than three times its width. The fourth sternum has a wide hyaline flap that is medially
shallowly emarginate. The fifth and third sterna are noticeably apically convex and the galeae are coarsely shagreened above. The apical areas of the
second and third terga have suberect hairs. The first tergum’s basal fourth-fifths bears long pale ochraceous hairs, and the apical area of this same
tergum has suberect, simple, usually pale brown (sometimes ochraceous), short hairs that almost reach the apical margin. The overall vestiture of the
second tergum is pale ochraceous to white. The basal pubescent band of the second tergum is white, as is the distal pale band. However, the distal
pale band is medially interrupted. The apical area of the second tergum bears simple, suberect, often pale ochraceous (sometimes brown) hairs. The
third and fourth terga are similar to that of the second, apart from the distal bands being uninterrupted, the basal tomentum being brown, and the
apical hairs being largely brown (minimally medially and sometimes more). The fifth tergum is similar to that of the fourth, except the distal band
often reaches the apex across the entire tergum and the sixth and seventh terga are entirely covered in pale brown to ochraceous hairs.
♀ Generally a pale bee with a dark patch of mesoscutal hairs and dark hairs on the vertex. The eyes are often gray to bluish-gray and the wing veins
are dark brown. The underside of F3-F10, and usually the apical area of F2 are yellow to reddish beneath and the ultimate flagellar segment is
short, around as long as it is wide and approximately equal to the length of the penultimate segment, as well as truncated (almost creating a
square edged appearance). The scopal hairs are highly branched, having more than three branches on either side of each rachis, mostly ochraceous
to white, and, on the posterior area of the tibial apices, a few to several scopal hairs are somewhat sinuous and appressed. The first tergum has
long basal and lateral hairs that are pale ochraceous. The basal area of the second tergum has long pale ochraceous hairs while the interband
zone has suberect comparably simple, short, dark hairs that are dark ochraceous to brown. The distal pale pubescent band of the second tergum is
wide, often becomes medially interrupted, arched, and laterally reaches, or almost reaches, the apes of the tergum. The apical area of the second
tergum is narrowly glabrous marginally, but anterior to this margin, bears suberect to appressed, simple, ochraceous to brown hairs. The third tergum,
is similar to that of the second, apart from the wider uninterrupted distal band that laterally reaches that apex of the tergum, and the interband
zone, which bears appressed, sparse, pale pubescence. The pale ochraceous band on the fourth tergum is wide, positioned apically, and, at times, has
some pale simple hairs near the medial area of the apical margin. The fifth and sixth terga are covered in dark brown hairs apart from the lateral
tufts, which are pale ochraceous.
Similar Species
Given thet the structure of the ultimate segment of female M. lupinus is shared by no other species within the genus, a comparison is unnecessary. However, males can strongly resemble those of M. glenwoodensis due to both species having longitudinal depressions on the outer surfaces of F5-F9 (and sometimes F10, but never F11) and the first flagellar segment’s minimum length less than, or equal to, one-fourth of the second segment’s maximum length.
♂ Males of M. lupinus can be separated from those of M. glenwoodensis by the apical areas of the second and third terga, which have suberect hairs; and by the galeae, which are often dull due to dense shagreening. In contrast, the apical areas of the second and third terga of M. glenwoodensis are glabrous as well as impunctate and the galeae are either shiny or dull due to shagreening.
Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) lupinus in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Johnson, 2020; Graham et al., 2021; Best et al., 2022a; Ikerd & Engler, 2023; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026; The International Barcode of Life Consortium, 2026). Data licensed under CC-BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Callimelissodes) lupinus in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Ikerd, 2019; Johnson, 2020; Graham et al., 2021; Best et al., 2022a; Ikerd & Engler, 2023; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026; The International Barcode of Life Consortium, 2026) and range-wide data compiled from (Ikerd, 2019; Johnson, 2020; Graham et al., 2021; Best et al., 2022a; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; Droege & Maffei, 2025; Gibbs, 2025; Scott, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Best, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Fägerström, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Kelly et al., 2026; Kenneth S. Norris Center for Natural History, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Oram, 2026; Orrell & Informatics, 2026a; San Diego Natural History Museum, 2026; Santa Barbara Museum of Natural History, 2026; The International Barcode of Life Consortium, 2026; University of Arizona Insect Collection, 2026; University of California, Davis, 2026; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
While its name seems to suggest that it specializes on lupin (Lupinus), M. lupinus seems to be an oligolege of Asteraceae instead. Although it has been found atop 16 other families, the numbers of specimens collected atop these are minute in comparison to those collected atop Asteraceae, and are therefore likely collecting only nectar instead of pollen, supporting the assumption of oligolecty of Asteraceae (Laberge, 1961). According to the records from Laberge (1961), out of the 787 M. lupinus with floral accounts, 659 of them were collected from Asteraceae. This renders Asteraceae to be favored 83.73% of the time by M. lupinus.
Synonyms
Melissodes lupina, Cresson, 1878; Melissodes intermediella catalinensis, Cockerell, 1905; Melissodes humilior catalinensis, Cockerell, 1923; Melissodes catalinensis vanduzeei, Cockerell, 1923
Melissodes lustrus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes lustrus
Melissodes lustrus is by far most closely allied to M. compositus, but the females are also somewhat similar to M. glenwoodensis on some accounts. Males are distinct in that F4-F10 have longitudinal depressions (if a depression is present on the third segment, it’s not well defined); the length of the penultimate flagellar segment is longer than two times its width; the apical areas of the second and third terga are glabrous and the galeae are often shagreened dorsally; the eyes are often yellow-green to green; and the interband zone of the third tergum (usually the second one as well) have their punctures that are mostly separated by one puncture diameter, with many that are separated by two to three puncture diameters. Females are distinctive in that the first tergum’s crowded basal punctures are restricted to, and do not surpass the, basal third or less medially; punctures past the medial one-third of the first tergum are mostly separated by more than two puncture diameters (not crowded); the galeae are moderately shiny above, delicately reticularly shagreened, but not densely tessellate; the hairs on the inner surfaces of the hind basitarsi are red to yellow and the scopal hairs are pale ochraceous to white; the fifth and sixth terga have golden to pale brown medial hairs and large white lateral tufts of hairs; the punctures on the mesoscutum medial to the parapsidal lines are irregularly sized (mostly smaller than those of the mesepisterna and scutellum), and separated by more than one-half, with many by more than two, puncture diameters. In both sexes, the apical areas of the terga (apart from the first and last two) are glabrous and very shiny, likely the character from which the species name “lustrus” (originally lustra) was derived. A similar pattern to that of the M. druriellus group (M. druriellus, M. pallidisignatus, and M. grindeliae) is mirrored within the M. compositus group (the group in which M. lustrus resides). That is, all species range-wide within these groups all occur, or likely occur, in Oregon and share very similar characteristics. This suggests that a common ancestor may be shared amongst the M. compositus group, from which the current-day species diverged. M. lustrus has been found across most of Oregon, and undoubtedly occurs across the entire state (Fig. ??). This species was originally thought to be active between the months of August and November (Laberge, 1961), but newer data suggests that activity starts around July (a few observations have been made in May and March, but these may not reflect the true phenological cycle) with peak activity occurring in August and September.
Field Markers
♂ Like most males, generally a pale bee with yellow-brown hyaline apical tergal margins. The mandibular bases, clypeus, and labrum are yellow, with the
labrum’s apical margin not darkened. The eyes are greenish yellow and the wing veins are black to brown. The underside flagellar segments 2-11are red to
yellow (these segments are dark brown on top), the first flagellar segment’s minimum length is one-third to one-fourth of the second segment’s maximum
length, and segments 4-10 (sometimes also on 11 but poorly defined on 3 if at all present) have shiny, shallow, longitudinal dorsolateral depressions. The
length of the penultimate segments is more than twice, but less than three times, its width and the ultimate segment’s length is approximately three times
its width. The fourth sternum is medially broad and shallowly emarginate, or evenly convex across the sterna, and the fifth and third sterna are noticeably
apically convex. The apical areas of the second and third terga are glabrous and the galeae are often shagreened dorsally. The interband zone of the third
tergum, and usually the second as well, have their punctures mostly separated by one puncture diameter, and several that are separated by 2 to 3 puncture
diameters. The head hairs are most ochraceous to cinereous, apart from the vertex, which bear brighter (sometimes faintly rufescent) hairs. The thoracic
hairs are also mostly ochraceous to cinereous apart from the dorsal areas, which bear brighter (sometimes faintly rufescent) hairs. The tergal hairs are
mostly pale ochraceous to white, with the second, third, and fourth terga having glabrous apical areas; the fifth tergum, which reaches the apex across
the entire tergum; and the sixth and seventh terga, which have some pale brown to dark ochraceous hairs, predominantly medially and basally.
♀ Genearlly a pale ochraceous bee with entirely opaque tergal margins. The eyes are green to greenish yellow and the wing veins are dark brown. The
underside of F3-F10, and usually the apical area of F2 are yellow to reddish beneath and the galeae are moderately shiny to shiny, reticularly shagreened
in the apical half or more, and have largely spaced punctures with straight hairs arising from them (no dense tessellation) The scopal hairs are highly
branched, having more than three branches on either side of each rachis and white to pale ochraceous. The first tergum’s crowded basal punctures are
restricted to, and do not surpass, the basal third or less medially. If areas past the medial one-third of the first tergum have punctures, then they
are mostly separated by more than two puncture diameters. The hairs on the inner surfaces of the hind basitarsi are red to yellow and the fifth and sixth
terga have golden to pale brown medial hairs as well as large white lateral tufts of hairs. The punctures on the mesoscutum medial to the parapsidal
lines are irregularly sized (mostly smaller than those of the mesepisterna and scutellum), and separated by more than one-half, with many by more than two,
puncture diameters. The first tergum has ochraceous basal hairs, but the apical area is glabrous. The second tergum’s basal area has white to pale
ochraceous pubescence, and the distal band is instead reduced to lateral short, oblique, pale fasciae that reaches, or almost reaches, the apical margin
at extreme lateral areas. The basal band is connected to this lateral fasciae by pale pubescence. The apical area of the second tergum is glabrous, but
the interband zone has suberect to appressed, simple, pale brown, tiny, scattered hairs. The third tergum is similar to that of the second, except the
basal hairs are brown, the distal band is usually complete with no medial interruption, the apical area is shorter (approximately one-half of the medial
area of the distal band), and the interband zone is instead covered in pale, diffused, appressed, hairs. The fourth tergum is similar to that of the
third, except the distal band reaches the apex across the entire tergum and is always complete.
Similar Species
M. lustrus is interesting in that the females are similar to two species, those being M. compositus and M. glenwoodensis, but males are only related to one, M. compositus. M. lustrus females are allied with these species in that the crowded basal punctures of T1 do not surpass the basal one-third medially, but seem to be most closely related to the former. Males on the other hand only resemble those of M. compositus due to F4-F10 have depressions, the length of the penultimate flagellar segment is longer than two times its width, and the apical areas of T2 and T3 are glabrous. Surprisingly given the female clade that is formed, males are highly distinct from those of M. glenwoodensis in the matter of their flagellar depressions.
♂ Males can be separated from those of M. compositus by the interband zone of the third tergum, and usually the second as well, which have punctures that are mostly separated by one puncture diameter (several that are separated by 2 to 3 puncture diameters) and the eyes, which are often yellowish green to green. Although females are similar to M. glenwoodensis, males differ in that flagellar segments 4-11 have longitudinal depressions, whereas in M. glenwoodensis, only F5-F9 (occasionally F10, but never F4 or F11) have depressions. For this reason, M. glenwoodensis is strangely more allied with M. lupinus due to the antennal structure.
♀ Females of M. lustrus can be separated from those of M. glenwoodensis by the red to yellow hairs on the inner surfaces of the hind basitarsi, and the fifth and sixth terga having golden to pale brown medial hairs as well as large white lateral tufts of hairs. In contrast, M. glenwoodensis have black to dark reddish brown hairs on the inner surfaces of the hind bastitarsi and the fifth and sixth terga have black to dark brown hairs or no white lateral tufts. However, M. compositus shares these same traits with M. lustrus, and is instead separated by tergal sculpturing. M. lustrus is separated from those of M. compositus in that the mesoscutal punctures medial to the parapsidal lines are irregularly sized (mostly smaller than those of the mesepisterna and scutellum), and separated by more than one-half, with many by more than two, puncture diameters. In contrast, M. compositus has its mesoscutal punctures medial to the parapsidal lines often and/or mostly larger than those of the mesepisterna or scutellum, and mostly separated by less than, or equal to, one-half of a puncture diameter.
Fig. ??. Map showing an estimation for the known distribution of M. (Callimelissodes) lustrus in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Johnson, 2020; Best et al., 2022a; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026). Data licensed under CC-BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Callimelissodes) lustrus in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Ikerd, 2019; Johnson, 2020; Best et al., 2022a; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026) and range-wide data compiled from (Ikerd, 2019; Johnson, 2020; Graham et al., 2021; Best et al., 2022a; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; Droege & Maffei, 2025; Gibbs, 2025; Scott, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Best, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026a; Cheadle Center for Biodiversity and Ecological Restoration, 2026b; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Fägerström, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Kelly et al., 2026; Kenneth S. Norris Center for Natural History, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Oram, 2026; Orrell & Informatics, 2026a; San Diego Natural History Museum, 2026; Santa Barbara Museum of Natural History, 2026; The International Barcode of Life Consortium, 2026; University of Arizona Insect Collection, 2026a; University of California, Davis, 2026; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
According to Laberge (1961) M. lustrus is a known oligolege of Asteraceae, depending on its plants for pollen, but especially so for the genera of Gutierrezia (snakeweed). Along with this, M. lustrus also seems to partially rely on plants from the genera Chrysothamnus (rabbitbrush) and Isocoma (goldenbrush), but to a lesser extent. From a total of 63 specimens with floral data attached, 61 were collected atop Asteraceae, hence the oligolecty. However, it seems as though these 61 specimens were collected across 44 collection events, 17 events of which all specimens were collected atop Gutierrezia. Assuming a similar amount of individuals were collected across all collection events, approximately 24 specimens were found atop species of Gutierrezia, rendering this genus to be favored by M. lustrus 39.34% of the time. For this estimation to be somewhat accurate, an assumption of individuals per collection events must be made, and as this isn’t specified in Laberge (1961), the percentage is therefore speculatory.
Synonyms
Melissodes lustra, Laberge, 1961.
Melissodes lutulentus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes lutulentus
Melissodes lutulentus, the Muddy longhorn bee, is a rarer Melissodes in Oregon, with a total of 9 publicly available state records, placing this species as the eighth rarest Oregonian Melissodes. M. lutulentus seems to be allied with M. subagilis as well as M. pullatellus, but more so the former than the latter. Males are distinctive in that the first flagellar segment’s minimum length is usually equivalent to one-fifth to one-eighth the second segment’s maximum length; the clypeus is entirely light excluding the dark spots denoting the tentorial depressions or the anterior notches and margin; the labrum usually has a mediobasal cream-colored macula, although not always; the mandibular bases have no yellow macula; the punctures on the interband zone of the second tergum are large and mostly separated by two puncture diameters or less (each puncture is notably wider than the bases of the hairs that arise from them); the interband zones of the third and fourth terga have large, deep, round punctures that are mostly separated by 1 puncture diameter; the first tergum’s apical hairs are appressed, white, dense, plumose, and short creating a distinct band that reaches the apex across the entire tergum (only fully obscuring the apical areas laterally); the second tergum’s distal pale pubescent band is at least as wide, and usually wider, than the apical area (sometimes obscures the entire apical area). Females are distinctive in that the apical area of the third tergum is narrower than the second tergum’s distal pale band (and one-third of the width of the third tergum’s pale band or less); the apical tergal areas are opaque, or translucent dark brown (not hyaline); the distal pale band of the second tergum usually isn’t medially interrupted; the underside of most of the flagellum is dark red; the interband zone of the second tergum bears pale subappressed hairs and usually dark brown, appressed to subappressed, spine-like hairs. This species has been found across the majority of Oregon, and although its westward most Oregonian observation occurred near Klamath Falls, it seems as though M. lutulentus inhabits somewhat coastal/bay areas in California, such as Antioch, suggesting it may be present throughout the entirety of the state. This species was historically believed to be active from May to October with peak activity occurring in July and August, and newer data suggest a similar sentiment, but peak activity seems to occur more so in August than July.
Field Markers
♂ Very pale in vestiture, cinereous to almost white. The eyes are often blue to greenish blue, and sometimes they can be gray to yellowish gray.
The clypeus is yellow besides for the darkened tentorial depressions and the testaceous apical margin and the labrum usually has a mediobasal cream-colored
macula, although not always. The wing veins are dark brown or black to red and the underside of the flagellar segments 2-11, which are red to yellow
(F1 entirely dark brown). The terga are colorless to faintly yellow hyaline in the apical areas and the clypeus is flat without a large bulge or
concavity. The first flagellar segment’s minimum length is usually equivalent to one-fifth to one-eighth the second segment’s maximum length. The
minimum length of the first flagellar segment (smaller side of F1) is often more than half the maximum length (longer side of F1). The length of the
penultimate flagellar segment is longer than three times its width. The flagellum reaches the pterostigma when in repose. The punctures on the interband
zone of the second tergum are larger and mostly separated by two puncture diameters or less; each puncture is notably wider than the bases of the hairs
that arise from them. The interband zones of the third and fourth terga have large, deep, round punctures that are mostly separated by 1 puncture diameter.
The head hairs are often entirely cinereous to white. The thoracic hairs are mostly cinereous to white apart from the mesoscutum, which has its
posteromedial area abundantly covered in long brown hairs, and the scutellum, which has long brown medial hairs. The setal description is as follows:
the first tergum’s apical hairs are appressed, white, dense, plumose, and short. This creates a distinct band that reaches the apex across the entire
tergum, but only obscures the apical area laterally. The second tergum’s distal pale pubescent band is at least as wide, and usually wider, than the
apical area (often reaching the apical margin laterally and sometimes across the entire tergum). The third, fourth, and fifth terga are similar to that
of the second, except the pubescent bands become closer to the apical margin with each subsequent tergum until the band reaches the apex across the apex
across the entire tergum on T5. The sixth and seventh terga are completely covered in white to dark ochraceous hairs.
♀ Generally pale bee with dark hairs on the dorsum of the thorax, much less ochraceous than those of M. agilis. The eyes are often blue, but sometimes they
are gray or greenish blue and the mandibles are usually rufescent. The wing veins are usually reddish brown to black, and often dark red. The underside of
flagellar segments 3-10, and usually the second in the apical one-third are red to yellow (dark brown on top) and the apical one-third of first tergum,
which is usually rufescent. The interband zone of the second tergum has dark brown, appressed to subappressed, spine-like hairs. The galeae are dull due
to shagreening and the hairs on the inner surfaces of the hind basitarsi are dark. The apical areas of the second, third, and fourth terga are piceous
(mostly opaque, or if slightly hyaline, then dark brown hyaline). The apical area of the third tergum is narrower than the second tergum’s distal pale
band (and one-third of the width of the third tergum’s pale band or less). The first tergum’s basal area bears pale ochraceous hairs, but in the apical
areas, the tergum is glabrous. The basal pale pubescent band of the second tergum is white. The interband zone of the second tergum bears short,
completely to primarily ochraceous, suberect to subappressed hairs. The distal pale pubescent band of the second tergum is white to pale ochraceous,
often uninterrupted medially, medially wider than that of the apical area, often reaches the apex of the tergum laterally, and composed of short, closely
spaced, appressed, plumose hairs (these hairs present across the apical area of the band as well). The third tergum is similar to that of the second,
except the distal pale pubescent band is around three times the width of the medial portion of the apical area or more, and the basal tomentum is brown.
The fourth tergum’s distal pale pubescent band is pale ochraceous to white, complete, and reaches the apex across the entire tergum. The fifth tergum
either bears, or does not bear long wide lateral tufts of pale hairs and the sixth tergum is medially orange-ish brown to brown either with or without
pale tufts.
Similar Species
Both sexes of M. lutulentus are most similar to M. subagilis, but females are also very weakly allied with M. pullatellus. This is due to a similar sculpturing shared between the species, but females do not closely resemble those of M. pullatellus due to the differing vestiture (M. pullatellus is almost entirely black), and therefore a subsequent comparison seems unnecessary. Females can also, to an extent, resemble those of the M. druriellus group (M. druriellus, M. pallidisignatus, and M. grindeliae) as well as M. microstictus as the vestiture is similar, but to a lesser extent than M. subagilis. Males are somewhat similar to the M. semilupinus group (M. bimatris and M. semilupinus) due to the apical band of hairs on T1, but slightly differ in this band’s structure.
♂ Males of M. lutulentus can be separated from those of M. subagilis by the first tergum’s apical hairs, which are appressed, white, dense, plumose, and short creating a distinct band that reaches the apex across the entire tergum (only obscures the lateral areas); the mesoscutum and scutellum usually have dark hairs (not always); and the second tergum’s distal pale pubescent band, which is at least as wide, and usually wider, than the apical area. In contrast, the first tergum’s apical hairs of M. subagilis are medially sparse and/or minutely barbed (only dense and plumose laterally), the dorsal thoracic hairs are entirely light, and the second tergum’s distal pale pubescent band is usually narrower than the apical area. The males can also somewhat resemble those of M. bimatris and M. semilupinus, but can be easily separated by the apical band on the first tergum not medially obscuring the apical margin.
♀ Given their strong aliance, M. lutulentus females will initially be compared to those of M. subagilis, then subsequent comparisons of weaker resemblance will follow. M. lutulentus can be identified from those of M. subagilis by the large dark mesoscutal patch that's minimally larger than the scutellar dark patch, and the overall pale hairs taking on a white color. In contrast, the mesoscutum of M. subagilis usually doesn’t have a dark patch of hairs, but if it does, then it’s smaller than that of the scutellum’s dark patch, and the pale hairs are mostly ochraceous to yellow (prominently so on the mesoscutum). M. lutulentus can also resemble those of M. grindeliae due to similar vestiture, but differ in the pale appressed to subappressed hairs of the interband zone of T2 and the more abundant punctures on the interband zone of T2. In contrast, the interband zone of the second tergum of M. grindeliae is impuncate or essentially so and has erect to suberect dark hairs. However, M. lutulentus often has dark brown appressed to subappressed, spine-like hairs on the interband zone mixed with the pale hairs, but M. grindeliae has no pale hairs. Females can also resemble M. pallidisignatus, but differ in the smaller size (the length of the forewing including the tegulae is less than 9 mm.). Lastly, M. lutulentus can bear a resemblance, though not very strongly, to M. druriellus, but can quickly be separated by the non-contrasting sculpturing of the second tergum’s interband zone and apical area.
Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) lutulentus in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from ( Ikerd, 2019; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026). Data Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Eumelissodes) lutulentus in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from ( Ikerd, 2019; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026) and range-wide data compiled from (Ikerd, 2019; Johnson, 2020; Carril et al., 2023; Droege & Maffei, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Fägerström, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026; University of Arizona Insect Collection, 2026a; University of Arizona Insect Collection, 2026b; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
Very little is known in regards to the flower records of M. lutulentus. Laberge (1961) states the M. lutulentus is an Asteraceae oligolege, preferring Chrysothamnus, Solidago, and Grindelia, but that seems to be the extent of what is currently known.
Synonyms
Melissodes lutulenta, Laberge, 1961.
Melissodes menuachus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes menuachus
Melissodes menuachus, the Goldenrod longhorn bee, is interesting as the females are very closely allied with M. semilupinus, and males seem to be allied with those of M. semilupinus as well, but lack the distinctive character of the apical band on the first tergum. Males fall into two separate groups based on their morphology, one where the first flagellar segment’s maximum length is more than one-third of the third segment’s minimum length, and the other where the first flagellar segment’s maximum length is less than, or equal to, one-third of the third segment’s minimum length. These will be known as group one (more than one-third), and group two (equal to, or less than, one-third); this can be seen at couplet 23 of the male key. Group one of the males are distinctive in that the first flagellar segment’s maximum length is more than one-third of the third segment’s minimum length; the first tergum has a band across its apical margin, but does not obscure the apical area medially; the galeae are shiny with no shagreening; the labrum and mandibular bases are yellow and the mesoscutal and scutellar hairs are not medially dark. Group two of the males are distinctive in that the first flagellar segment’s maximum length is less than, or equal to, one-third of the third segment’s minimum length; the clypeus is entirely light excluding the dark spots denoting the tentorial depressions or the anterior notches and margin; the mandibular bases have a yellow macula and the labrum has a large mediobasal pale macula (at least equal to, and usually larger than, one-third of the labrum); the galeae are shiny above with no shagreening, except for the tips; the first flagellar segment’s maximum length is equal to, or almost equal to, one-third of the third segment’s maximum length, and the minimum length of the first flagellar segment is notably longer than the pedicel’s length on the same side. Females do not share the same variation as the males, and are distinct in that the length of the second flagellar segment is faintly, but notably longer than its width; the hairs on the inner surfaces of the hind basitarsi are dark brown to brown; the fifth and sixth terga have pale ochraceous to white lateral tufts of hairs; the mesepisterna have no dark hairs. This species has a total of six state-wide records, placing it the seventh rarest in Oregon, that mostly occur in the Eastern areas of this region (Fig. ??). However, even though its westward most Oregonian observation was recorded near Pendleton, range-wide data show that M. menuachus has a presence in coastal Californian areas, implying that it may occur across the entire state. This species has been historically known to be active between July and October with peak activity occurring in August and September (Laberge, 1961), and new data seem to reflect this (Fig. ??).
Field Markers
♂ Generally a vey pale bee, often with yellowish hairs on the distal two most terga. The eyes are greenish gray to yellowish gray and the clypeus is
yellow. The labrum is mostly white except for the apical margin, which is narrowly brown and the bases of the mandibles are yellow. The wing veins are
pale red to yellow and flagellar segments 2-10 are red to yellow on the underside and dark red to brown on top (first segment usually completely dark).
The apices of the terga are colorless to yellow hyaline and the clypeus protrudes beyond the eye by about half the width of an eye in profile view.
The first flagellar segment’s minimum length is about two-thirds of its maximum length, and noticeably longer than the pedicle on the same side. The
penultimate segment is about three times as long (maximum) as it is wide (minimum). None of the flagellar segments have longitudinal lateral depressions.
The first flagellar segment’s maximum length can be more than, less than, or equal to one-third of the third segment’s minimum length, and the first flagellar
segment’s minimum length is about one-fifth or somewhat less than the maximum length of the second segment. The galeae are shiny with no shagreening and the
mesoscutal and scutellar hairs are not dark medially. The hairs on both the head and the thorax are white to pale ochraceous, but on the vertex of the head
and the dorsal area of the thorax, the hairs become a brighter ochraceous. The first tergum has long basal white to ochraceous hairs and long appressed to
subappressed apical pale hairs that reach the apex across the tergum but don’t obscure the apical portion medially; sometimes not obscuring the apical
margin more than medially. The second tergum has white basal pubescence and pale suberect, bristle-like hairs on the interband zone. The distal pubescent
band of the second tergum is anteriorly separated from the apex of the tergum by about one-half to one times the length of the medial area of the pale
band and is not interrupted medially. Terga 3-5 are similar to that of the second tergum except for the interband zones, which in addition to the bristle-like
hairs there are also delicate, sparse, white, appressed pubescence. Also, the distal bands of terga 3-5 become closer to the apical margin with each tergum.
♀ Very pale bee, especially for those of female Melissodes, with incredibly long, often pale yellow, sometimes pale ochraceous scopae. The eyes are greenish
blue to bluish gray and the apical margin of the clypeus is often rufescent. The wing veins are reddish brown to red and the underside of flagellar segments
3-10 are rufescent. The apical areas of terga 2-4 are sometimes rufescent and the galeae are shiny. The fifth and sixth terga have pale ochraceous to white
lateral tufts of hairs. The mesepisterna have no dark hairs. The length of the second flagellar segment is faintly, but notably longer than its width and the
hairs on the inner surfaces of the hind basitarsi are are dark brown to brown. The first tergum’s basal area has long, pale, ochraceous hairs that reach the
apical margin at extreme lateral areas but not medially. The apical area of the first tergum is glabrous. The second tergum’s white basal pubescence laterally
connects to the distal pale band. The interband zone of the second tergum has rather simple, subappressed, sparse pale hairs. The distal pale pubescent band
is two times the length of the apical glabrous area laterally, and medially, the band is narrowed and notched measuring only half the length of the apical
glabrous area. The third tergum is similar to that of the second, except the basal tomentum is dark brown and the distal pale band is separated from the apical
margin due to a narrow line/zone of rather simple, suberect, pale hairs. The fourth tergum is similar to that of the third, but lacks the narrow line/zone of
simple, suberect, pale hairs. The fifth and sixth terga are entirely covered in brown hairs excluding the white lateral tufts.
Similar Species
Females of M. menuachus are very similar to those of M. semilupinus in the length of the second flagellar segment. Males are similar to M. semilupinus as well, but starkly differs in the vestiture of the first tergum. Other than M. semilupinus, M. menuachus does not seem to directly resemble any other species in Oregon (M. menuachus is related to M. submenuachus, but this species doesn't reside in Oregon) apart from superficial vestiture similarities, which will be treated below.
♂ Males are somewhat allied with M. semilupinus, be it less than the females, and differ largely in that the first tergum’s apical margin isn’t obscured by a dense band of pubescence. Due to similar vestiture and integumental coloration, M. menuachus can also resemble M. agilis in that they both have hyaline tergal margins, mandibular macula, pale labrums, very similar to almost identical vestiture (when compared to a lighter male of M. agilis), and similar antennal coloration. However, the two differ greatly in the length of the first flagellar segment and the galeal sculpture. M. menuachus has shiny, unshagreened to very little shagreened galeae and the first flagellar segment’s minimum length is about one-fifth or somewhat less than the maximum length of the second segment. Whereas in M. agilis, the galeae are densely shagreened and the minimum length of the first flagellar segments is about one-sixth to one-fifth of the second segment’s maximum length.
♀ Females are really only similar to one species, that is M. semilupinus, due to the length of the second flagellar segment being faintly, but notably longer than its width (these are the only two species in Oregon with this characteristic). M. menuachus can be separated from those of M. semilupinus by the fifth and sixth terga, which have pale ochraceous to white lateral tufts of hairs; and the mesepisterna, which have no dark hairs. In contrast, \the fifth and sixth terga of M. semilupinus do not have pale ochraceous to white lateral tufts of hairs; and the mesepisternal hairs are black to dark brown on the lower-lateral areas. As no other species share the antennal structure in Oregon, this renders M. menuachus quite distinct, and further comparisons based on similar vestiture seem unnecessary.
Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) menuachus in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Illinois Natural History Survey, 2026). Data licensed under CC-BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Eumelissodes) menuachus in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Ikerd, 2019; Illinois Natural History Survey, 2026) and range-wide data compiled from (Ikerd, 2019; Johnson, 2020; Carril et al., 2023; Ikerd & Engler, 2023; South Dakota State University, Severin-McDaniel Insect Research Collection, 2023; Texas A&M University Insect Collection, 2023; Droege & Maffei, 2025; Scott, 2025; Sánchez & Comisión, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Gibbs, 2026; Grinter et al., 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Mertz et al., 2026; Mississippi Entomological Museum, 2026; MT James Entomological Collection, Washington State University, 2026; New Mexico State Collection of Arthropods, 2026; Oram, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026; University of Arizona Insect Collection, 2026a; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable. (see "Dataset Licenses").
Flower preference
M. menuachus is a known oligolege of plants from the family Asteraceae, and seem to specifically prefer Grindeliae and Solidago (Laberge, 1961). Of the 153 M. menuachus collected with floral data attached, 69 have been collected atop species from the genus Grindelia, and 23 have been collected from Solidago; the two combined comprising over 60% of the total flower records. This renders Grindeliae to be favored 45.09% of the time, and Solidago 15.03% of the time, by M. menuachus. The remaining flowers do not show any specific outlying values, suggesting that the foregoing genera are of great importance to M. menuachus, hence the high percentages.
Synonyms
Melissodes mennacus, Uhler, 1877; Melissodes pallida, Robertson, 1895; Melissodes mizeae, Cockerll, 1905; Melissodes blakei, Cockerell, 1905; Melissodes lavata, Cockerell, 1924; Melissodes octobris, Cockerell, 1934.
Melissodes metenuus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes metenuus
Melissodes metenuus seems to be most closely allied with M. plumosus in both sexes, but the males seem to resemble one another much more than the females. Similarly to those of M. druriellus, M. metenuus females range widely in setal colorations, but no specific geographical variations have been discovered in regards to their vestiture. As it's unknown what morphs occur in Oregon, general descriptions of the two extremes (darkest and lightest) will be given, along with a list of the features and how they gradient between the extremes. Males are distinctive in that the length of the penultimate flagellar segment is longer than two times its width; the apical areas of the second and third terga have simple, erect to suberect hairs; F4-F10 have lateral depressions, and the third flagellar segment also usually has a well defined ventrolateral depression; the galeae are dorsally shiny and unshagreened, or if shagreened, then delicately so on less than, or equal to, the apical half; sterna 2-4 are medially shiny and delicately reticularly shagreened; the clypeus is coarsely punctate basally. Females are distinctive in that the scopae are mostly white to pale yellow; the lateral areas of the mesepisterna bear dark hairs; the crowded basal punctures of the first tergum do not surpass the basal one-half; the head hairs are entirely, or almost entirely, dark brown to black; the hairs on the inner surfaces of the hind bastitarsi and often the tibiae are dark reddish brown to black; the galeae are shiny to moderately shiny above with no, or very little, faint shagreening. This species has a total of 84 state records, placing it the third most common Oregonian Melissodes, outpacing M. robustior by 9 observations. M. metenuus has been found across the entirety of Oregon, with most observations occurring West of bend, but these may be simply due to sampling biases. In the literature, this species has been reported to show activity between June and September, with peak activity occurring in July and August, and current data suggest the same pattern (Fig. ??).
Field Markers
♂ Generally a paler bee with its tergal rims somtimes yellow hyaline apically, but usually opaque brown. The mandibular bases and labrum
(the labrum having its margin not darkened are yellow). The clypeaus is a light yellowish orange and the eyes are greenish blue to greenish gray.
The underside flagellar segments 2-11 are red to yellow (these segments are dark brown on top) and the wing veins are black to brown. The first
flagellar segment’s minimum length is less than, or equal to, one-fifth of the second segment’s maximum length, and segments 3-10 (often on the
basal portion of 11 as well) have shiny, shallow, longitudinal dorsolateral depressions. TThe length of the penultimate flagellar segment is
longer than two times its width and the ultimate segment’s length is more than three times its width. The fourth sternum has a wide hyaline flap
that is not medially shallowly emarginate (or if emarginate, then incredibly shallowly), and the fifth and third sterna are noticeably apically
convex. The galeae are dorsally shiny and unshagreened, or if shagreened, then delicately so and only on less than, or equal to, the apical half.
Sterna 2-4 are medially shiny and are delicately reticularly shagreened. The clypeus is coarsely punctate basally. The apical areas of the second
and third terga have simple, erect to suberect hairs. The first tergum’s basal fourth-fifths bears long pale ochraceous hairs, and the apical
area of the tergum has suberect, simple, usually dark brown, short, sparse hairs that almost reach the apical margin. The overall vestiture of
the second tergum is pale ochraceous to white. The basal pubescent band of the second tergum is white, as is the distal pale band. However, the
distal pale band is medially interrupted. The apical area of the second tergum bears simple, sparse, suberect, often pale dark brown hairs. The
third and fourth terga are similar to that of the second, apart from the distal bands being uninterrupted and the basal tomentum being brown. The
fifth tergum is similar to that of the fourth, except the distal band often reaches the apex across the entire tergum. The sixth and seventh
terga are entirely covered in pale brown to ochraceous hairs.
♀ Generally a darker bee, but vestiture coloration can vary widely. However, the head hairs are always dark, an important feature in
identification of M. metenuus. The eyes are bluish green to greenish gray and the wing veins are black to dark brown. The underside of F3-F10,
and usually the apical area of F2 are yellow to reddish. The galeae are shiny to moderately shiny above with no, or very little, faint
shagreening. The hairs on the inner surfaces of the hind bastitarsi and often the tibiae are dark reddish brown to black. The lateral areas
of the mesepisterna bear dark hairs and the crowded basal punctures of the first tergum do not surpass the basal one-half. Given that the females
are highly variable in vestiture (not binomial as in M. bimatris having a single light and dark form with no intergrades), full descriptions
regarding their setae will be given; one for the lightest form, the darkest form, and the steps of integration. The darkest individuals have
entirely black hairs, differing at the scopae, which are whitish to pale yellow, the tibiae, which are also whitish to pale yellow; the basitarsi,
which are brown to pale yellow; and the outer surfaces of the tibiae, which are apically white. The description for the palest individuals is
as follows. The head hairs are entirely black. The thoracic hairs are entirely dark brown to black, differing at the scutellum, which has some
pale hairs on its periphery; the mesoscutum, which has some pale ochraceous hairs on the lateral areas and anterior most one-third; the
metanotum, which has some pale hair dorsally; the propodeum, which also has some pale hair dorsally; and the top one-third of the lateral
mesepisternal surfaces, which have ochraceous hairs. The basal one-half of the first tergum bears mostly dark hairs with a few pale hairs mixed.
The second tergum has cinereous, oblique, lateral, fasciae on either side of the tergum rather than a complete band. The fasciae are medially
separated from one another minimally by the length of one fascia, and sometimes more. The third tergum has a distal pale band of cinereous pubescence
that is separated from the apical margin across the entire tergum. The fourth tergum is similar to that of the third. The fifth and sixth terga
are covered entirely in dark hairs with no lateral pale tufts. The scopae are white to pale yellow with some brown hairs near the basitibial
plate, as well as on the lateral and apical areas of the basitarsi. The outer surfaces of the fore tibiae and middle tibiae usually bear white
hairs near the apex.
M. menuachus females range between the two extremes presented. Starting from the darkest form, light hairs appear in these steps: 1) pale fasciae
on the third tergum is developed and the dorsal surfaces of the metanotum and propodeum bear their pale hairs; 2) pale hairs develop laterally on
the fourth tergum, on the anterior areas of the mesoscutum, and on the periphery of the scutellum; 3) pale hairs develop on the second tergum,
the basal areas of the first tergum, and on the upper lateral areas of the mesepisterna.
Similar Species
M. metenuus is perhaps most closely allied to those of M. plumosus, but females can resemble a few other Oregonian Melissodes, especially so in the darkest morph. Males are similar to M. plumosus as they both share the characters of the apical areas of the second and third terga having simple, erect to suberect hairs; the galeae are often being shiny without shagreening or delicately reticularly shagreened; The third flagellar segment usually having a well defined ventrolateral depression; the length of the penultimate flagellar segment being longer than two times its width; and F4-F10 (along with the well defined depression on F3) have longitudinal depressions on the outer surface that are shallow, narrow, and shiny. Females aren’t necessarily directly related to any other Callimelissodes in Oregon, but rather can resemble a few darker Eumelissodes such as M. pullatellus, M. bimatris, and sometimes even M. rivalis.
♂ Males of M. metenuus can be separated from those of M. plumosus by the galeae, which are dorsally shiny and unshagreened, or if shagreened, then delicately so on less than, or equal to, the apical half; sterna 2-4 are medially shiny and are delicately reticularly shagreened; and the clypeus is coarsely punctate basally. In contrast, males of M. plumosus have galeae that are somewhat dull dorsally due to delicate distinct shagreening; sterna 2-4, which are only moderately shiny medially due to shagreening; the clypeus, which has small round basal punctures. Separation of the two species is quite difficult as they are incredibly similar, only distinctly differing in the sculpture of sterna 2-4.
♀ Whithin their sungenus, females of M. metenuus are somewhat distinct in Oregon given the dark hairs on their heads. However, outside Callimelissodes, darker forms of M. metenuus can resemble M. pullatellus, and lighter forms can resemble those of M. rivalis. Females can be separated from all other Oregonian subgenera by the dark hairs on the side of the thorax, and the crowded basal punctures on the first tergum not surpassing the basal one-half, and often less. Females can be separated from those of M. rivalis in that the inner eye margins noticeably converge near the mandibles, and that the clypeus protrudes beyond the eye by less than one-half of an eye's width in profile view. In contrast, M. rivalis’ inner margins of the eyes are parallel to one another, and the clypeus protrudes beyond the eye by one-half to two-thirds of an eye’s width in profile view. Due to the combination of these two characters, females of M. metenuus are somewhat distinctive in Oregon, much more so than their male counterparts.
Fig. ??. Map showing an estimation for the known distribution of M. (Callimelissodes) metenuus in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Johnson, 2020; Graham et al., 2021; Best et al., 2022a; Ikerd & Engler, 2023; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026). Data liscenced under CC BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Callimelissodes) metenuus in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Ikerd, 2019; Johnson, 2020; Graham et al., 2021; Best et al., 2022a; Ikerd & Engler, 2023; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026) and range-wide data compiled from (Ikerd, 2019; Johnson, 2020; Graham et al., 2021; Best et al., 2022a; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; Droege & Maffei, 2025; Bentley & Osborn, 2026; Best, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026a; Grinter et al., 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
A total of 3 flower records are known of M. metenuus, only one of a female visitor. All flower records included in this list are from reports in the literature or datasets. Each flower has a parenthesized reference listed after it, corresponding to the literary work or dataset in which it was recorded. Currently, M. metenuus has been collected atop Cosmos sp. (Laberge, 1961), Eriogonum sp. (Laberge, 1961; listed as “Erigonum,” a possible misspelling), and Melilotus officinalis (Laberge, 1961; listed as the historical synonym “Melilotus alba”). With only a single female record (Melilotus officinalis), a justifiable statement of preference and/or oligolecty seems unfounded.
Synonyms
Melissodes metenua, Cockerell, 1924.
Melissodes microstictus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes microstictus
Melissodes microstictus, the small longhorn bee, is, as its vernacular name suggests, a small bee that seems to be very distinctive in Oregon. This species seems to be the most closely related to M. menuachus out of all the other Oregonian Melissodes (be it weakly), but most closely resembles those of M. paululus. Males are distinctive in that the underside of the flagellae are almost completely black, apart from a small ventral pale macula, and have no depressions; the terga are apically hyaline; the first flagellar segment’s maximum length is less than, or equal to, one-third of the third segment’s minimum length; and the clypeus is partially black. The females are distinctive in that the flagellum is almost entirely black to dark reddish brown apart from a few dark red small ventral maculations of F3-F10; the distal pubescent band of the second tergum is narrowly interrupted; the hairs on the inner surfaces of the hind basitarsi are dark; the length of the forewing including the tegulae measures to be 7.0 to 8.5 mm. and the pygidial plate is V-shaped acutely; The apical areas of the second, third, and fourth terga are piceous; the apical area of the third tergum is often mostly obscured due to the distal band which reaches the apex, minimally laterally and often more (apical area always narrower than the second tergum’s distal band); the hairs on the anterior area of the mesoscutum are erect and blunt-tipped, appearing as if they have been cut. This species is the second most common of its genus in Oregon with a total of 202 records widespread across the entire state, falling second only to M. lupinus. This high commonality paired with the distinctive morphological traits renders this species quite notable within Oregon (similar to M. lupinus), making it easily one of the most recognizable species state-wide. M. microstictus has been historically recorded to be active between the months of June and October, with peak activity occurring in July and August (Laberge, 1961), but it seems a few collections suggest that activity can possibly commence as early as April (Fig. ??). However, these records seem to be outliers, and June is suggestive of the most representative month of activity commencement.
Field Markers
♂ Somewhat pale bee, but there is a noticeable black to very dark brown patch of hairs on the posteromedial area of the mesoscutum, and
most of the scutellum. The eyes range between grayish green, dark gray, and bluish and the underside of flagellar segments 3-11 are black
to dark reddish brown with a small rufescent ventral spot on each individual segment. The apical one-half of the mandibles are rufescent
(bases black without any macula) and the clypeus is mostly yellow, differing at its posterior most one-third to two-thirds, which are
black, and the apical margin, which is brown. The wing veins are black to dark brownish red and the apical areas of the terga are colorless
to brownish yellow hyaline. The first flagellar segment’s minimum length is approximately one-fifth to one-seventh of the second segment’s
maximum length, and when in repose, the flagella reach the pterostigma. The clypeus is flat without a large bulge or concavity and the
length of the penultimate flagellar segment is less than three times its width. The sixth sternum’s subapical oblique carina is either
absent or reduced, becoming indistinct. The head hairs are mostly pale ochraceous to white with several brown hairs atop the vertex. The
thoracic hairs are white on the lateral areas, white on the mesoscutum with a large posteromedial patch of dark brown hairs that’s often
smaller than twice the size of the scutellar dark patch, dark brown hairs on the tegulae, and dark brown on the scutellum with white hairs on the periphery. The first
tergum has white hairs at its base, but laterally, these hairs reach the apex of the tergum. The apicomedial apubescent area has simple,
dark brown, short hairs that reach the apical margin. The basal area of the second tergum bears white hairs. The interband zone of the
second tergum bears wire-like, erect to suberect, brown to white hairs. The distal pale band of the second tergum is white, connected to
the basal pubescence laterally, reaches the apical margin laterally, and approximately the same width medially as the apical area. The
apical area of the second tergum bears suberect to subappressed, long, dark brown hairs. The third, fourth, and fifth terga are similar
to that of the second, differing at the basal tomentum, which is brown; the apical areas, which are narrower to completely absent; and
the interband zones, which have white hairs that are diffused across the surface. The sixth and seventh terga are covered in pale brown
to yellow hairs.
♀ Generally a pale bee often having the hairs on terga 2-4 ochraceous-yellow. The eyes are grayish green to blue and the wing veins are
black. The underside of flagellar segments 3-10 are black to faintly dark brownish red. The length of the forewing including the tegulae
measures to be 7.0 to 8.5 mm. and the pygidial plate is V-shaped acutely. The distal pubescent band of the second tergum is narrowly
interrupted, and the hairs on the inner surfaces of the hind basitarsi are dark. The third tergum’s distal pale pubescent band reaches
the apical margin minimally laterally and often more, but if the apical area is present, then it’s glabrous, impunctate, and narrower than
that of the second tergum’s distal band. The apical area of the first tergum is often opaque and the length of the second flagellar
segment is less than, or equal to, its width. The head hairs are mostly yellow to pale ochraceous with many brown hairs on the vertex.
The thoracic hairs are pale ochraceous to white on the lateral areas, ochraceous to yellow with a large posteromedial patch of dark brown
hairs that reaches the anterior margins of the tegulae on the mesoscutum, dark brown hairs on the tegulae, and dark brown with ochraceous hairs on the periphery of the
scutellum. The first tergum bears ochracous hairs basally, and has short, dark brown, subappressed hairs on the anteriolateral rounded
projections on the apical area. The basal area of the second tergum bears pale ochraceous hairs. The interband zone of the second tergum
bears wire-like, dark brown, suberect to erect hairs. The distal pale band of the second tergum is ochraceous, connected to the basal
pubescence laterally, and narrowly medially interrupted due to the presence of brown hairs. The apical area of the second tergum bears
simple, long, suberect to subappressed, dark brown hairs apart from the apicomedial area, which is narrowly glabrous. The third tergum is
similar to that of the second, differing at the basal tomentum, which is brown; the distal pubescent band, which is complete and reaches
the apical margin of the tergum, minimally so in the lateral areas, and sometimes across more; and the apical area, which is much narrower
to entirely absent. The fourth tergum is similar to the third, but the distal pale pubescent band reaches the apex across the entire tergum,
and sometimes that are a few dark brown hairs at the apical margin in less than, or equal to, the medial one-fourth of the tergum. The fifth
and sixth terga are covered in dark brown hairs, but the fifth tergum sometimes have some lateral tufts of ochraceous hairs.
Similar Species
M. microstictus is quite a distinctive bee in Oregon due to the flagellar coloration. As stated above, this species is weakly allied with M. menuachus and M. paululus, but both sexes greatly differ.
♂ Males are highly distinctive in Oregon due to their near entirely black antennae and the posterior one-third to two-thirds of the clypeaus being darkened. M. paululus can have a similar darkened clypeal coloration, but the terga are opaque and the antennae have flagellar depressions, separating it further from M. microstictus. Due to this, no other Oregonian Melissodes resemble males and further comparisons are unnecessary.
♀ Females can be similar to a few Oregonian Melissodes, but seems to most resemble those of M. paululus due to the same blunt-ended appearance of the anterior mesoscutal hairs. M. microstictus distinctly differs from this species in that the underside of the flagellar segments are much darker, nearing full black antennae with just a few dark brownish red colored maculae on a few segments. In contrast, M. paululus and M. menuachus have the underside of flagellar segments 3-10 rufescent. The vestiture of M. microstictus can look similar to those of the druriellus group (M. druriellus, M. pallidisignatus, and M. grindeliae), but females can be separated from this group by the distal pubescent band of the second tergum being narrowly interrupted, the darkened flagella, and the hairs on the inner surfaces of the hind basitarsi being dark. In contrast, all females of the druriellus group have rufescent colorations on the underside of their flagella, often lighter hairs on the inner surfaces of the hind basitarsi (not always), and the second tergum’s distal pale pubescent band is complete.
Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) microstictus in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Johnson, 2020; Best et al., 2022a; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026; MT James Entomological Collection, Washington State University, 2026; The International Barcode of Life Consortium, 2026). Data liscenced under CC BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Eumelissodes) microstictus in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Johnson, 2020; Best et al., 2022a; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026; MT James Entomological Collection, Washington State University, 2026; The International Barcode of Life Consortium, 2026) and range-wide data compiled from (Ikerd, 2019; Johnson, 2020; Best et al., 2022a; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; Droege & Maffei, 2025; Grant et al., 2025; Scott, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Best, 2026; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Gibbs, 2026; Illinois Natural History Survey, 2026; Kenneth S. Norris Center for Natural History, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Native, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
M. microstictus is a likely oligolege of Asteraceae, and according to the flower records presented by Laberge (1961) this species seems to be frequently found atop Aster, Solidago, Helianthus, Grindeliae, Erigeron, Guterrezia, and Chysothamnus. According to Laberge (1961), of the 104 M. microstictus collected with floral accounts, 89 were found atop plants of the family Asteraceae, but no single genera seemed to be of preference.
Synonyms
Melissodes microsticta, Cockerell, 1905.
Melissodes minusculus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes minusculus
Melissodes minusculus is very closely related to M. ablusus, and as stated by Laberge (1961), may in fact be a different morph of this species. The males are the most similar, having near identical flagellar structure, sternal structure, and very similar sculpturing, separated only by vestiture color. Females share this same allyship as well, both having very similar sculptures (especially the galeae) and structures, differing mostly in the vestiture. Males are distinctive in that F4-F10 have depressions; the length of the penultimate flagellar segment is two times its width or less; the galeae are matte and dull dorsally due to coarse, dense, and regular tessellation; the first flagellar segment’s minimum length is less than one-fifth of the second segment’s maximum length; the hairs on the sixth and seventh terga are white to gold-colored and the hairs on the basal half of terga 3-5 are pale brown to ochraceous. Females are distinctive in that the first tergum’s crowded basal punctures usually extend to the basal half medially. The medial one-third of the first tergum has punctures that are mostly separated by one puncture width or less, minimally basally and sometimes more; the galeae are matte and dull above due to coarse, dense, regular tessellation; the hairs on the inner surfaces of the hind basitarsi are dark red to dark brown; the hairs on the head are entirely white apart from the brown vertex hairs (no dark hairs below the vertex). Although this species has yet to be found in Oregon, its range may have a possibility of overlap, and is herein accounted for in the guide. Given how closely related M. ablusus and M. minusculus are, and the singular M. ablusus Oregonian observation straying quite far from its historically recognized range, there is a possibility that the specimen collected from Oregon is M. minusulus rather than M. ablusus, although this is just speculation. M. minusculus currently has a total of 65 publicly available records, which seem to indicate that this species is active between the months of June and August, with peak activity occurring in July (Fig. ??).
Field Markers
♂ A pale bee, paler than those of M. ablusus, with bluish gray to gray, and on rare occasions, greenish blue eyes. The mandibular bases
and clypeus are cream to pale yellow and the entire labrum (apical margin not darkened) is yellow. The underside flagellar segments 2-11
are red to yellow (these segments are dark brown to dark red on top) and the wing veins are reddish brown to red. Terga 2-5 are sometimes
yellow hyaline apically, but usually opaque brown. The first flagellar segment’s minimum length is less than one-fifth of the second
segment’s maximum length, and segments 3-10 (sometimes also on 11 near the base) have shiny, shallow, longitudinal dorsolateral depressions.
The length of the penultimate segments is equal to, or less than, to times its width and the ultimate segment’s length is less than three
times its width. The fourth sternum has a wide hyaline flap that isn’t medially shallowly emarginate (or if so then very shallowly and
indistinctly so), and the fifth and third sterna are noticeably apically convex. The galeae are matte and dull dorsally due to coarse,
dense, and regular tessellation. The overall vestiture is mostly pale ochraceous, differing at the vertex of the head, which is ochraceous
to yellowish; the mesoscutum and scutellum, which are also ochraceous to yellowish; the basal tomentum terga three, four, and five, which
is dark brown in the basal most areas; the sixth and seventh terga, which are golden brown to ochraceous; the inner surfaces of the basitarsi
and distitarsi, which are red to yellow (predominantly red on the middle basitarsi and hind basitarsi). The distal pale band of the second
tergum is complete and reaches the apex of the tergum minimally laterally. The distal pale bands of the third and fourth tergum reach the
apex of the tergum minimally laterally, and the interband zones bear pale pubescence that diffuses across the integument.
♀ Very pale and small bee measuring around 9-10 mm. in length. The eyes are often gray to bluish-gray and the wing veins are dark brown.
The underside of F3-F10, and usually the apical area of F2 are yellow to reddish. The hairs on the inner surfaces of the hind basitarsi are
dark red to dark brown. The hairs on the head are entirely white apart from the brown vertex hairs; there are no dark hairs below the vertex.
The galeae are matte and dull above due to coarse, dense, regular tessellation. The first tergum’s crowded basal punctures usually extend to
the basal half medially. The medial one-third of the first tergum has punctures that are mostly separated by one puncture width or less,
minimally basally and sometimes more. The head hairs are mostly white, apart from the vertex, which has several dark brown hairs. The thoracic
hairs are mostly white as well, differing at the mesoscutum, which has pale ochraceous hairs and a large patch of dark hairs that sometimes
reaches the median of the tegulae, but never reaches past; and the scutellum, which has a few scarce medial dark brown hairs. The basal one-half
of the first tergum bears ochraceous hairs and the apical area bears simple, minute, appressed, sparse, dark brown hairs. The basal pubescence
of the second tergum is white, and the interband zone minimally bears suberect brown hairs in the medial areas, and sometimes more. The distal
pale pubescent band of the second tergum is wide, medially interrupted narrowly, and reaches the apex of the tergum laterally. The basal tomentum
of the third tergum is dark brown and the interband zone bears white hairs that diffuse across the integument. The distal pale band of the third
tergum reaches the apex across the entire, or most of, the tergum, and is complete. The fourth tergum also has dark brown basal tomentum and an
uninterrupted pale band. The fifth and the sixth terga are covered in orange to pale brown hairs, often paler on the sixth terga than the fifth,
apart from the lateral pale tufts that are approximately equal to one-fourth to one-third of the tergum’s width.
Similar Species
As can be determined above, M. minusculus is very closely related to M. ablusus in both sexes. Females are similar in that the galeae are matte and dull above due to coarse, dense, regular tessellation, and the crowded punctures on the first tergum surpass the basal one-third medially. Males are similar in that F4-F10 have longitudinal depressions, the length of the penultimate flagellar segment is two times its width or less, and that the galeae are matte and dull.
♂ Males can be separated from those of M. ablusus only with some difficulty. The two differ in that the hairs on the sixth and seventh terga are white to gold-colored in M. minusculus, and the hairs on the basal half of terga 3-5 are pale brown to ochraceous. In M. ablusus, the hairs on the sixth and seventh terga are mostly brown and the hairs on the basal half terga 3-5 are mostly brown as well. These species are very challenging to separate and even more difficult without a reference to compare. Males of M. minusculus are also similar to those of M. clarkiae given the shared antennal structure, but can be separated by the densely tesselated and dulled galeae. In contrast, the galeae of M. clarkiae are also somewhat dulled, but only by delicate reticular shagreening rather than dense tessellation.
♀ Females differ from those of M. ablusus in that the hairs on the inner surfaces of the hind basitarsi are dark red to dark brown, and the hairs on the head are entirely white apart from the brown vertex hairs; there are no dark hairs below the vertex. In contrast, for minusculus the hairs on the inner surfaces of the hind basitarsi are black to dark reddish brown, and the hairs on the head are entirely, or almost entirely, dark brown; minimally there are dark and pale hairs mixed on the face below the vertex. To an extent, females also resemble those of M. plumosus, but can be quickly separated by the mate and dull galeal sculpture. Due to the specific characters listed above, females of M. minusculus don’t directly resemble any other species.
-Fig. ??. Map showing an estimation for the known distribution of M. (Callimelissodes) minusculus. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Johnson, 2020; Bentley & Osborn, 2026; Grinter et al., 2026; Illinois Natural History Survey, 2026; Kelly et al., 2026; Mertz et al., 2026). Data licensed under CC BY 4.0 and CC0 1.0 as applicable (see "Dataset Licenses").
-Fig. ??. A graph representing the phenology of M. (Callimelissodes) minusculus. The x value is the month, and the y value is the number of documented observations occurring in that month. Data compiled from (Ikerd, 2019; Johnson, 2020; Bentley & Osborn, 2026; Grinter et al., 2026; Illinois Natural History Survey, 2026; Kelly et al., 2026; Mertz et al., 2026). Data licensed under CC BY 4.0 and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
Very little is known in regards to the flower records, non-the-less flower preference of M. minusculus. Due to the lack of sufficient data, the current known flower records will be listed. All flower records included in this list are from reports in the literature or datasets. Each flower has a parenthesized reference listed after it, corresponding to the literary work or dataset in which it was recorded. Currently, M. minusculus has been found atop Cressa cretica (Laberge, 1961) Heliotropium curassavicum (Laberge, 1961), Lythrum sp. (Illinois Natural History Survey, 2026; GBIF record 3801380879).
Synonyms
Melissodes minuscula, Laberge, 1961.
Melissodes nigracauda

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes nigracauda
Melissodes nigracauda is highly distinctive in both sexes. Males are interesting as they are the only Callimelissodes in Oregon to have no flagellar depressions, and females are also unusual as they are the only Callimelissodes in Oregon to have dark scopae. Males are distinctive in that the first flagellar segment’s minimum length is more than one-fourth of the second segment’s maximum length; the flagella have no shallow narrow depressions laterally, at most, some segments might have some flattened areas; the galeae are matte and dulled dorsally due to regular, dense tessellation; the fourth sternum is widely convex apically. Females are distinctive in that the majority of the scopal hairs are dark brown, sometimes paler brown medially; the metasomal hairs are mostly dark brown to black (sometimes there is a small amount of paler pubescence on the second tergum); the galeae have dense tessellation. This species isn’t related to a single other Callimelissodes, but females can resemble those of M. bicoloratus and dark forms of M. bimatris due to similar vestiture. This is interesting as the only three Melissodes in the entire genus that exhibit this darkened vestiture with ochraceous highlights (M. nirgracuda, M. bicoloratus, and dark forms of M. bimatrus) also occur, or likely occur, in Oregon, suggesting these vestiture colorations possibly play a role in keeping these species safe. Furthermore, both M. bicoloratus and M. bimatris seem to prefer the genera Ericameria and Chrysothamnus (both similar plants in structure), which are similarly colored to the ochraceous dorsal pubescence, potentially allowing for better camouflage against predators (M. nigracauda has too few records to justify a flower preference). Given the lack of antennal depressions males can resemble a few other Eumelissodes superficially, although to a lesser extent than the females due to the subgeneric characters. Although it has yet to be found in Oregon, a 2024 survey from the Washington State Department of Agriculture yielded results of M. nigracauda occurring in Washington. When pairing this with its Southern Californian observations, it would be surprising if this species doesn’t occur in Oregon (Fig. ??). M. nigracauda has been recorded to be active between the months of June to September, with peak activity occurring in September (Fig. ??).
Field Markers
♂ A very pale bee with few dark hairs. The eyes are grayish blue to gray and the wing veins are black to dark reddish brown. The mandibular bases
and clypeus are cream colored to pale yellow and the labrum is white apart from the narrow brown apical margin. The underside of flagellar
segments 2-11 are red to yellow. The first flagellar segment's minimum length is longer than, or equal to, one-fourth of the second segment's
maximum length. The penultimate and ultimate flagellar segments are minimally three times their width. The flagella do not have dorsolateral
longitudinal depressions like many other Callimelissodes, but rather flagellar segments 5-11, and often the basal areas of the fourth segment,
are noticeably compressed and have shiny flattened areas. The fourth sternum is apically widely convex, and usually very shallowly medially emarginate.
The apical margins of sterna 3 and five are only very slightly convex, and can almost appear as being transverse if not viewed from afar. The hairs on
the head are entirely ochraceous. The thoracic hairs are also entirely ochraceous, but the hairs on the mesoscutum and scutellum tend to be faintly
darker. The metasomal hairs are ochraceous, differing at the apical area of the first tergum, which has short, appressed, dark brown hairs; the apical
areas of terga two and four, and usually the medial areas of the fifth tergum as well, which have similar apical hairs to that of the second; the basal
areas of the third, fourth, and fifth terga, which have dark brown tomenum; the sternal hairs, which are yellow to brownish in the medial areas,
becoming pale ochraceous laterally. The leg hairs are mostly ochraceous apart from the orange to yellow hairs on the inner surfaces of the tarsi.
♀ A very dark bee, not the darkest in Oregon (M. pullatellus), but nearing so. Minimally, flagellar segments 4-10, and often 3 as well, are rufescent
and although not explicitly stated by Laberge (1961), some M. nigracauda females, when alive, may have very dark blue eyes. The majority of the scopal
hairs are dark brown, sometimes paler brown medially, and the metasomal hairs are mostly dark brown to black (sometimes there is a small amount of
paler pubescence on the second tergum). The galeae have dense tessellation and the scopal hairs are highly branched, having more than three branches
on either side of each rachis. The surface of the first tergum is somewhat shiny, but dulled due to sense reticulotransverse shagreening and the basal
one-half has indistinct, sparse, and incredibly shallow punctures. The apical one-half of the first tergum is entirely impunctate. The punctures in the
basal area of the second tergum are minute, mostly separated by three to four puncture diameters, and the surface of this area is shiny. The interband
zone of the second tergum bears indistinct punctures sparsely located around the integument and the apical area is entirely impunctate; the microsculpture
of these surfaces are similar to the shagreening of the first tergum. The third and fourth terga are similar to that of the second. The pygidial plate is
shaped like a triangle with a wide base, the tip becoming pointed rather than apically rounded as in most Melissodes. The head hairs are almost entirely
black to dark brown, apart from the vertex and the areas around the antennal fossae, which may sometimes have a few pale hairs. The thoracic hairs are
black to dark brown, differing at the tegulae, mesoscutal, scutellar, and dorsal propodeal surfaces, which bear ochraceous to faintly rufescent hairs.
The metasomal hairs are black to dark brown, differing at the basal one-half of the first tergum, which usually has some long pale hairs mixed with the
dark pubescence; the distal pale band of the third tergum, which usually has a few ochraceous hairs laterally, but sometimes entirely dark. The apical
hairs of the second and third terga are dark brown, simple, sparse, appressed and minute. The leg hairs are mostly black to dark brown with the hind
tibial, and sometimes basitarsal scopal hairs airing more towards a dark brownish hue rather than black (scopae dark).
Similar Species
M. nigracauda females only resemble two other Melissodes, M. bicoloratus and dark forms of M. bimatris, both of which occur or likely occur in Oregon. Males on the other hand are quite distinctive due to the structure of the fourth sternum (a Callimelissodes subgeneric characteristic), and the lack of antennal depressions. For this reason, only a female comparison will be given, as no other Oregonian Melissodes exhibit the traits of these males.
♀ Given the darkend vestiture with prominent ochraceous pubescence on the dorsal areas of the thorax and somtimes head, M. nigracauda resembles the foregoing species. Females can be separated from those of M. bimatris much easier than from those of M. bicoloratus in the color of the scopae. M. nigracauda has dark, near-black scopal hairs whereas M. bimatris has bright ochraceous to pale fluvo-ochraceous scopae, often lighter than their dorsal thoracic pubescence. On the other hand, M. bicoloratus shares this same scopal hair coloration, and the two are instead most easily separated by galeal sculpture. M. nigracauda has distinctly shagreened galeae whereas M. bicoloratus has shiny unshagreened galeae. Furthermore, the two can be separated by subgeneric characteristics as M. bicoloratus resides in Eumelissodes and M. nigracauda resides in Callimelissodes.
Fig. ??. Map showing an estimation for the known distribution of M. (Callimelissodes) nigracauda. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Texas A&M University Insect Collection, 2023; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; MT James Entomological Collection, Washington State University, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Callimelissodes) nigracauda. The x value is the month, and the y value is the number of documented observations occurring in that month. Data compiled from (Ikerd, 2019; Texas A&M University Insect Collection, 2023; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; MT James Entomological Collection, Washington State University, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
Although the lack of sufficient flower data aren’t indicative of certain diet preferences, the flowers from which M. nigracauda has been seen will be listed. All flower records included in this list are from reports in the literature or datasets. Each flower has a parenthesized reference listed after it, corresponding to the literary work or dataset in which it was recorded. Currently, M. nigracauda has been found atop Erigeron sp. (Laberge, 1961), Senecio flaccidus var. douglasii (Laberge, 1961; historical synonym), Stephanomeria sp. (Laberge, 1961), Stephanomeria exigua (Laberge, 1961), Stephanomeria virgata (Laberge, 1961). Counterintuitively, the flower records show no indications of M. nigracauda even visiting Ericameria or Chrysothamnus, suggesting that this species is further separated from both M. bimatris and M. bicoloratus not only in morphology, but in diet as well.
Synonyms
None.
Melissodes pallidisignatus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes pallidisignatus
Melissodes pallidisignatus, the pale striped long-horn bee, is highly variable in both sexes and closely related to M. druriellus. Males are distinctive in that the length of the penultimate flagellar segment is always less than three times its width (maximum length and narrowest width); the labrum has a yellow macula, and the mandibles can have maculae (often a small basal pale macula in this region if present), but are often entirely black; when in repose, the antennae usually do not reach the pterostigma; the apical areas of terga 2-5 are colorless to testaceous or brownish yellow hyaline; the basal areas of terga 4-6 are covered in pale pubescence and hairs. Females are distinctive in two manners due to their variation, that being apical suberect pale hairs near the distal pubescent band of the third tergum are either present or absent, separating females into two groups. Group 1 of females are distinctive in that the distal band on the third tergum often reaches the apex of the tergum at extreme lateral areas; the pale hairs on the mesoscutum positioned anteriorly to the dark patch of hairs are pale ochraceous to white; the scopal hairs are ochraceous; the first tergum is reticularly shagreened and the punctures are small, distinct, and approximately separated by one-half of a puncture width; the interband zone of the second tergum is shiny, not dull; the mesoscutum clearly has a dark brown to black patch of posteromedial hairs; the clypeus rarely protrudes beyond the eye by one-half of an eye’s width in profile, almost always by less; the apical area of the third tergum either bears some erect pale somewhat simple hairs, or is glabrous, impunctate, and narrower than that of the second tergum’s distal band. Group two of females are distinctive in that the length of the forewing including the tegulae measures to be 9 to 10 mm; the lateral raised areas of the interband zone of the second tergum have distinct punctures that are of two sizes, some large and rather posteriorly directed, and the other that is tiny; the hairs on the inner surfaces of the hind basitarsi are dark brown; the underside of most of the flagella is rufescent; and the apical area of the third tergum is either glabrous, impunctate, and widely triangularly shaped, although no wider than around one-third of the tergum’s width, or. However, many females found in Oregon seem to be much darker (the darkest M. pallidisignatus ever found was in Oregon), often having its distal pale band on the third tergum composed partially of dark hairs. This species has been found across the majority of Oregon, with a few notable records in Crater Lake-Klamath Marsh, where the darkest known females reside. M. pallidisignatus was originally known to be active between the months of June to October with peak activity occurring in August (Laberge, 1961), but newer data show that this species has been observed during May as well (Fig. ??).
Field Markers
♂ Often a pale bee, hence the vernacular name, but setal coloration varies, be it not substantially. The eyes are grayish
blue to green and the wing veins are dark brown to red. The mandibular bases can be one of three varieties, entirely black,
have a small round pale macula, or have a large triangular macula. However, the majority of males collected in Oregon (and in
most western regions as well) often have no macula to very small basal pale macula. No males have currently been found that bear
large triangular yellow maculae. The clypeus is yellow and the underside of flagellar segments 2-11 are red to yellow. The apical
areas of the terga are dark brownish red to colorless hyaline and the labrum bears a mediobasal pale macula that ranges from
small to large in size. The minimum length of the first flagellar segment is approximately one-fourth to one-third of the second
segment’s maximum length, and the length of the penultimate flagellar segment is less than three times its width. When in repose,
the flagella do not reach the prestigma. Similar to the females, males can vary in setal coloration, although to a much less extent
and more variation within a given population occurs. Males vary in the sculptures of the galeae, base of the first tergum, and the
interband zones of the second and third terga. Across most of Oregon, males (dark and pale alike, no correlation seems to be present
between vestiture and sculpting; see setal variation below) seem to present a ratio of approximately 50:50 of shiny terga and galeae
to dulled terga and galeae. Note that in this region, there seems to be more of an emphasis on shagreening, therefore “shiny” isn’t
completely unshagreened, rather less dense than the dulled variant. Although these are common in Oregon, interpopulational variation
is present. As no documentations of variation in Oregon have been published since 1961, a general statement will also be given to
account for any unfound sculptural variation using range-wide extremes. The tergal and galeal sculpture of males ranges from shiny
with no to very faint shagreening, to densely shagreened and dull. The setal description given below will be broadened to include
the different morphs of males that occur range wide, with parenthesised annotations for characters that are often Oregon specific
in accordance to Laberge (1961). Due to this, integumental and structural features should be used when identifying M. pallidisignatus.
The hairs on the head are white to pale ochraceous, but on the vertex, the hairs may be brown (often brown in Oregon). The thoracic
hairs are mostly white to pale ochraceous as well, but the scutellum usually has some dark medial hairs (varying amounts per individual,
but also often present in Oregon), the tegulae rarely has brown hairs (darkest Oregonian specimens may), and the mesoscutal hairs tend to
be abundantly dark brown (predominantly in western individuals, including those of this range). The hairs on the dorsum of the thorax range
from being long and somewhat erect, to erect and short (often short to medium length hairs that are truncated/blunt-ended in Oregon, but
not always). The first tergum has long basal white to ochraceous hairs and long appressed to subappressed apical pale hairs that reach the
apex across the tergum but don’t obscure the apical portion medially; sometimes not obscuring the apical margin laterally as well. The
second tergum has white to rich brown basal pubescence (often darker in males from Oregon) and pale suberect, bristle-like hairs on the
interband zone. The distal pubescent band of the second tergum is anteriorly separated from the apex of the tergum by about one-half to
one times the length of the medial area of the pale band and is not interrupted medially. Terga 3-5 are similar to that of the second
tergum except for the interband zones, which in addition to the bristle-like hairs there are also delicate, sparse, white, appressed
pubescence. Also, the distal bands of terga 3-5 become closer to the apical margin with each tergum. The sixth and seventh terga are
covered in yellowish to white pubescence. The sternal hairs are medially reddish to pale ochraceous, and laterally white. The legs are
white to ochraceous excluding the pale rufescent to golden yellow inner surfaces of the tarsi.
♀ Females, like their male counterparts, are widely variable, but to a much higher extreme. In general females are moderately pale (similar in
color to M. druriellus) to distinctly dark (nearing levels of M. metenuus or M. rivalis). The eyes are bluish gray to gray and the wing veins
are brownish red to black ( often becoming paler in eastern individuals, but light wing veins may be present in Oregon as well). The underside
of flagellar segments 3-10 are rufescent and in the paler individuals, the apical tergal one-halves are rufescent. Females have two variations
of the apical area of the third tergum bears its hairs, one in which the apical area of the third tergum is glabrous, apart from a few suberect
hairs that are sometimes present near the distal most area of the pubescent band with the apical area approximately as wide as the band medially,
and one in which the apical area is glabrous and triangularly shaped with the distal pale band reaching the apex laterally. The apical area of
the second tergum is glabrous (a few dark appressed short hairs may be present near the band, but often not), impunctate, and shiny with incredibly
faint reticulotransverse shagreening. The interband zone of the second tergum ranges from being dull due to dense reticular shagreening (highly
contrasts with the apical area sculpturing; common characteristic of Oregon females) to less densely shagreened but still contrasting with the
apical area. The mesoscutum clearly has a dark brown to black patch of posteromedial hairs (sometimes when dark enough, the entire mesoscutum is
black). The clypeus rarely protrudes beyond the eye by one-half of an eye’s width in profile, almost always by less. The interband zone of the
second tergum is distinctly punctate and the length of the forewing including the tegulae measures to be 9 to 10 mm. The lateral raised areas of
the interband zone of the second tergum have distinct punctures. These punctures are of two sizes, some large and rather posteriorly directed,
nd the other that is tiny. The hairs on the inner surfaces of the hind basitarsi are dark brown. Although a few descriptions of the setae have
been given, it is apparent from these that females are highly variable in this manner. Laberge (1961) noted that most females from Oregon (especially
in the Crater Lake-Klamath marsh area) are distinctly dark, some of the darkest in the entire range (see table 4). However, no studies since have
been conducted to find if this holds true in current times. Therefore, a similar structure to that of Table 3 in the M. druriellus description
will be given below. This table was derived from characteristics listed in the geographical variation section in the M. pallidisignatus
description from Laberge (1961). Preceding this, a general pale description will be given to which the characters in table 4 modify.
The head hairs are mostly pale ochraceous to white, apart from the vertex, which bears many dark hairs. The thoracic hairs are
also mostly pale ochraceous to white, except as follows: the scutellar hairs, which are mostly dark brown; the mesoscutum, which
often has a large patch of dark posteromedial hairs that’s minimally equal to the size of the scutellar dark patch, and often
twice the size (the dark mesoscutal patch usually has a few pale hairs mixed with the dark hairs in the anterior areas). The
first tergum’s basal area has long, pale ochraceous hairs that reach the apical margin at extreme lateral areas but not medially.
The apical area of the first tergum is glabrous. The second tergum’s pale ochraceous basal pubescence laterally connects to the
distal pale band. The interband zone of the second tergum has rather simple, subappressed, sparse pale ochraceous hairs along
with a few wire-like erect hairs. The distal pale ochraceous pubescent band is often separated from the apical margin laterally
(apart from rare occurrences), and medially, the band is narrowed and notched. The apical area is glabrous. The third tergum is
similar to that of the second, except the basal tomentum is dark brown and the distal pale band usually reaches the apical
margin laterally, but not medially. The apical area of the third tergum is glabrous, apart from a few suberect hairs that are
sometimes present near the distal most area of the pubescent band, and approximately as wide at the distal pale band medially.
The fourth tergum is similar to that of the third, but the distal pale band reaches the apex across the entire tergum. The fifth
and sixth terga are entirely covered in brown hairs excluding the white lateral tufts. The sternal hairs are almost entirely
brownish red apart from the apical areas of the fifth sternum. The leg hairs are mostly pale ochraceous, differing at the
bastitibial plates, which are dark brown; the fore tibiae and middle tibiae, which are often pale brown on the outer apical
areas; the middle and hind basitarsi, which are dark brown; the fore basitari and often the distitarsi, which are also dark
brown.
Many levels of melanism seem to exist within females of M. pallidisignatus across its range. 8 characteristics each having three
different levels of melanism derived from descriptions of Laberge (1961) to account for these variations rather than writing
singular descriptions for each theoretical variety (6, 561 or 3^8 possible morphs). These are listed in the table below (table 4).
Traits of differing levels of melanism are meant to be applied to, and override portions of, the pale description above. Note that
each value per character (light, intermediate, or dark) is treated as being independent from one another (although, many are highly
correlated). Therefore, a female M. pallidisignatus may have all of its head hairs dark (darkest choice for the first character),
and also have a pale band across the fourth tergum (palest choice for the sixth character).
Table 4. Eight characters pertaining to the vestiture of the females of M. pallidisignatus each with three different melanistic variations. This table accounts for all possible morphs of the females of M. pallidisignatus when the characters are either added to, or replace, portions of the base-line pale description.
| Character | Light | Intermediate | Dark |
|---|---|---|---|
| Head hairs | Ochraceous with a few to many brown hairs on the vertex, less than 50% of the head hairs are dark. | Many more dark hairs, more than 50%, but the head still bears pale hairs. | Head hairs entity dark. |
| Dorsal thoracic hairs | The dorsal thoracic hairs range from having dark hairs only on the scutellum with no posteromedial dark patch, to having a smaller dark patch that doesn’t reach across the entire mesoscutum; the hairs on the pronotal lobes are pale . | Much larger dark patch of the mesoscutum, but pale pubescence is still present anteriorly and laterally, and the scutellar hairs are almost entirely dark; the hairs on the pronotal lobes are pale. | The entirety of the mesoscutum is dark apart from a narrow anterior area; the hairs on the pronotal lobes are dark. |
| Mespisternal hairs | Pale ochraceous to white. | A few dark hairs present ventrally, anteriorly and laterally on the mespisterna. | Over 50% to the entire lateral area of the mesepisterna is dark (entier surface may be dark). |
| Basal pubescence on the first tergum | Pale ochraceous. | Mostly pale ochraceous with very sparse dark hairs (overall, basal tergal vestiture mostly pale) | Noticable amounts of dark pubescence is mixed with pale hairs. |
| The distal band of the second tergum | Pale ochraceous with a narrow notch medially (often no dark hairs). | Few dark medial hairs, interrupted only narrowly. | Many dark hairs medially, widely interrupted. |
| The distal band of the third tergum | Pale ochraceous with a narrow notch medially (often no dark hairs). | Few dark medial hairs, interrupted only narrowly. | Many dark hairs medially, widely interrupted. |
| The apical distal band of the fourth tergum | Pale ochraceous. | A few to many dark hairs present. | Entirely dark. |
| Lateral tufts of the fifth and sixth terga | Present. | Reduced (often still noticeably present). | Absent. |
Similar Species
M. pallidisignatus is very similar to M. druriellus, both in vestiture variation and superficial resemblance. Males of M. pallidisignatus are much more similar to those of the first group of M. druriellus (see M. druriellus description) due to the same antennal structure, but differ in 2 or more characters. Females on the other hand, although closely allied with M. druriellus, can resemble a number of other Eumelissodes in multiple morphs (although, the existence of the palest morph in Oregon is unknown).
♂ Males can be seperated from those of the first group of druriellus in two ways. The first is that certain individuals of M. pallidisignatus may have mandibles that bear a basal yellow macula (likely a small pale yellow macula is present as no males with large triangular maculae have been found in Oregon). However, as stated above, this is a very rare occurrence from this state and the males often have entirely black mandibular bases; similar in integumental color to those of M. druriellus. The most reliable way to separate the two species is by the tergal rims. M. druriellus has its terga almost completely opaque to very deeply infumate dark brown, whereas M. pallidisignatus has its tergal rims hyaline yellow to brown. Males are also somewhat similar to group one of M. grindeliae males (see M. grindeliae description) as they share similar antennal structures as well. The two can be separated in that M. pallidisignatus have mediobasal cream to yellow colored maculae on their labrum whereas group one of M. grindelae have their labrums entirely black.
♀ Females are most similar to M. druriellus and M. grindeliae, but probably more so the former than the latter. M. pallidisignatus can be separated from those of m. druriellus by the distal pale band on the third tergum, which reaches the apex laterally; the hairs anterior to the mesoscutal dark patch, which are ochraceous to white; the scopae, which are ochraceous; ant the pictures on the first tergum, which are small, distinct, and approximately separated by one-half of a puncture width. In contrast, M. druriellus’ distal pale band on the third tergum does not reach the apex across the entire tergum and is instead well-separated from it; the pale hairs on the mesoscutum anterior to the dark patch of hairs are usually rusty-reddish; the scopae are usually a golden-yellow color; and the punctures on the first tergum are incredibly shallow, large, and crowded (these punctures are usually obscured by shagreening). Females can be separated from those of M. grindeliae by the distinctly punctate interband zone of the second tergum. In contrast, the interband zone is impuncate or essentially so. In its darkest morph, females look somewhat similar to those of M. rivalis due to the presence of white metasomal hairs (M. druriellus’ dark form is entirely black), but can be easily separated by the inner eye portions, which converge towards the mandibles in facial view rather than being parallel to one another. Although other Melissodes may bear a resemblance to the many forms of females of M. pallidisignatus, the foregoing species are the ones which seem to hold the highest similarities.
Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) pallidisignatus in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Johnson, 2020; Graham et al., 2021; Best et al., 2022a; Texas A&M University Insect Collection, 2023; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026). Data liscenced under CC BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Eumelissodes) pallidisignatus in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Ikerd, 2019; Johnson, 2020; Graham et al., 2021; Best et al., 2022a; Texas A&M University Insect Collection, 2023; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026) and range-wide data compiled from (Ikerd, 2019; Johnson, 2020; Graham et al., 2021; Best et al., 2022a; Carril et al., 2023; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Best, 2026; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Gibbs, 2026; Illinois Natural History Survey, 2026; Kenneth S. Norris Center for Natural History, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Native, 2026; Northern Arizona University, 2026a; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026; University of Arizona Insect Collection, 2026a; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
M. pallidisignatus is an oligolege of Asteraceae, and according to the flower records presented by Laberge (1961) this species seems to be frequently found atop Isocoma, Chrysothamnus, Grendeliae, and Solidago. According to Laberge (1961), of the 333 M. pallidisignatus collected with floral accounts, 279 were found atop plants of the family Asteraceae, but no single genera from the list above seems to be of significant preference. However, newer data also suggest that the genera Heterotheca and Ericameria are of importance to this species (Best et al., 2022a; Carril et al., 2023; Bently & Osborn, 2025; Illinois Natural History Survey, 2025; Kenneth S. Norris Center for Natural History, 2025; MT James Entomological Collection, Washington State University, 2026).
Synonyms
Melissodes nigrosignata pallidisignata, Cockerell, 1905; Melissodes menuacha vernonensis, Viereck, 1905; Melissodes praeluata, Cockerell, 1905.
Melissodes paululus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes paululus
Melissodes paululus is incredibly distinctive in the male sex and one of the most obscure Melissodes in Oregon. This is due to the structure of the flagella, that is, most male Eumelissodes have rounded non-depressed flagellar segments, but M. paululus males do (a characteristic typically associated with Callimelissodes males). Also interestingly, M. paululus males have their apical margins entirely opaque rather than hyaline as in most other Melissodes (apart from M. druriellus) in its subgenus, another characteristic that’s common within Callimelissodes males. This species seems to be the only Eumelissodes range wide in which the males have depressed flagellar segments. This coupled with the transverse margin of the fourth sternum (a characteristic that separates Eumelissodes from Callimelissodes) and the opaque tergal rimes renders M. paululus to be highly distinct. Females can be identified by the interband zone of the second tergum, which has punctures that are distinct, regular, and round across the entire tergum (the punctures might be more abundant laterally than medially); the apical one-half of the second tergum, which bears punctures and dark hairs; the mesoscutum, which clearly has a dark brown to black patch of posteromedial hairs; the clypeus, which rarely protrudes beyond the eye by one-half of an eye’s width in profile, almost always by less; and the apical area of the third tergum has dark hairs on the medial one-third. Currently, the only known records of this species in Oregon are from Umatilla, but likely occur slightly further eastward based on coastal Californian collections. M. paululus has been recorded to be active between the months of July and November with peak activity occurring in September and October (Laberge, 1961). Newer data seem to suggest a similar pattern (Fig. ??).
Field Markers
♂ Somewhat of a pale bee, but a noticeable dark patch on the dorsum of the thorax is present. The eyes range between grayish green, dark gray, and bluish and the
underside of flagellar segments 2-11 are yellow to red. The clypeus is mostly yellow, differing at its posterior most one-third to two-thirds, which is black and the
apical margin, which is brown. The labrum is entirely black. The wing veins are black to dark brownish red and the apical areas of the terga are piceous. The first
flagellar segment’s minimum length is approximately one-sixth of the second segment’s maximum length, and when in repose, the flagella surpasses the pterostigma.
The length of the penultimate flagellar segment is more than three times its width. F4-F10 bear shiny longitudinal depressions or flattened areas where the bright
yellow-red underside and the brown dorsal colorations meet (much as in most males from Callimelissodes). The sixth sternum’s subapical oblique carina is prominent
and apically converges, but doesn’t meet. The head hairs are mostly pale ochraceous to white with several brown hairs atop the vertex. The thoracic hairs are white
on the lateral areas, white on the mesoscutum with a large posteromedial patch of dark brown hairs that’s often smaller than twice the size of the scutellar dark
patch, and dark brown on the scutellum with white hairs on the periphery (the tegulae usually have no brown hairs, but sometimes, a few may be present). The first
tergum has white hairs at its base, but laterally, these hairs reach the apex of the tergum. The apicomedial apubescent area has simple, dark brown, short hairs that
reach the apical margin. The basal area of the second tergum bears white hairs. The interband zone of the second tergum bears wire-like, erect to suberect, dark brown
hairs. The distal pale band of the second tergum is white, connected to the basal pubescence laterally, reaches the apical margin laterally, and approximately the same
width medially, or shorter than, the apical area. The apical area of the second tergum bears suberect to subappressed, long, dark brown hairs. The third, fourth, and
fifth terga are similar to that of the second, differing at the basal tomentum, which is brown; the apical areas, which are narrower to completely absent; and the interband
zones, which have white hairs that are diffused across the surface. The sixth and seventh terga are covered in pale to dark brown hairs, often becoming paler in the lateral
areas. The sternal hairs are entirely white to yellow. The leg hairs are mostly white, apart from the inner surfaces of the tarsi, which are yellow.
♀ An intermediate colored bee, much lighter than those of M. bicoloratus and dark morphs of M. pallidisignatus and M. druriellus, but darker than M.
plumosus or M. agilis due to the dark dorsal thoracic patch (similar to that of the male). The eyes are grayish green to blue and the wing veins are black.
The underside of flagellar segments 3-10 are black to faintly dark brownish red and the apical area of the first tergum is often rufescent. The interband
zone of the second tergum bears punctures that are distinct, regular, and round across the entire tergum that may become more abundant laterally than medially.
The apical area of the second tergum has many dark appressed hairs arising from several punctures in the basal portion of this apical area. The apical area of
the third tergum has dark hairs in the medial one third and the pale distal band of this tergum reaches the apex laterally. The head hairs are mostly yellow to
pale ochraceous with many brown hairs on the vertex. The thoracic hairs are blunt-ended/truncated, pale ochraceous to white on the lateral areas, dark ochraceous
to vibrant rusty-red on the mesoscutum with a large posteromedial patch of dark brown hairs that falls short of reaching the anterior margins of the tegulae, and
isn’t larger than twice the size of the scutellar dark patch; and dark brown with ochraceous hairs on the periphery of the scutellum. The first tergum bears pale
ochracous hairs basally, and has short, dark brown, subappressed hairs on the anteriolateral rounded projections on the apical area. The basal area of the second
tergum bears pale ochraceous hairs. The interband zone of the second tergum bears dark brown, appressed to suberect hairs. The distal pale band of the second
tergum is pale ochraceous, medially interrupted with the lateral pubescence narrowing medially creating fasciae, and connected to the basal pubescence laterally.
The apical area of the second tergum bears simple, long, appressed, dark brown hairs that are close to one another apart from the apicomedial area, which is narrowly
glabrous. The third tergum is similar to that of the second, differing at the basal tomentum, which is brown; the distal pubescent band, which is complete and reaches
the apical margin of the tergum laterally; and the interband zone, which is reduced to entirely absent. The fourth tergum is similar to the third, but the distal pale
pubescent band reaches the apex across the entire tergum with no brown apicomedial hairs. The fifth and sixth terga are covered in dark brown hairs, apart from the
lateral tufts of ochraceous hairs.
Similar Species
Due to the subgeneric character of the fourth sternum being transverse rather than convex separating males from Callimelissodes, and the presence of shiny lateral depressions or flattened areas of flagellar segments 4-10, males do not resemble any other species in particular. The closest resemblance they may bear is to those of M. microstictus as both have similar vestiture, but M. paululus can easily be separated from all Eumelissodes by the foregoing characters. Therefore, in-depth comparisons seem unnecessary. Females on the other hand can resemble a number of other Eumelissodes such as M. microstictus, M. druriellus and M. pallidisignatus to an extent, and sometimes M. robustior.
♀ Females of M. paululus can be seperated from those of M. microstictus in that the apical area of the third tergum has dark hairs in the medial one-third, but often reaches the apex in the lateral areas. M. microstictus tends to have its distal pale band on the third tergum reach the apical areas laterally as well, but the apical area lacks dark hairs, or is entirely absent all together, but often appears almost triangular to thinner than the second tergum’s distal band. M. microstictus also greatly differs from M. paululus in that the underside of the flagella is very dark with each segment having a dark red to dark brownish red macula, whereas M. paululus has the underside of its flagella (apart from the first and second segments) bright reddish-yellow. M. paululus can also somewhat resemble lighter forms of M. pallidisignatus, M. grindeliae, and M. druriellus (the druriellus group) given the somewhat similar vestiture, but is quickly separated by the apical area of the second tergum, which bears many punctures and appressed dark hairs and is usually shiny. However, the interband zone of this tergum is also shiny, be it faintly shagreened, not contrasting with the apical area. For M. druriellus and M. pallidisignatus, the apical area of the second tergum is often glabrous and shiny, but the interband zone is densely shagreened, highly contrasting to the apical area. M. grindeliae can be separated in a similar way to M. microstictus.
Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) paululus in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019). Data liscenced under CC BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Eumelissodes) paululus in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Ikerd, 2019) and range-wide data compiled from (Ikerd, 2019; Johnson, 2020; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; Museum of Southwestern Biology, 2026; Orrell & Informatics, 2026; The International Barcode of Life Consortium, 2026; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC0 1.0, and CC BY-NC 4.0 as applicable (see "Dataset Licenses").
Flower preference
M. paululus seems to be an oligolege of Asteraceae (Laberge, 1961). According to Laberge (1961), of the 328 total M. paululus records with floral data attached, 324 of them were collected upon Asteraceae, with the bulk of this number falling into three genera: Isocoma (139), Solidago (84), and Gutierrezia (73). This renders Isocoma to be favored 42.37% of the time, Solidago 25.61 % of the time, and Guterrezia 22.26% of the time by M. paululus, these genera accounting for 90.24% of all flower records.
Synonyms
Melissodes paulula, Laberge, 1961.
Melissodes plumosus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes plumosus
Melissodes plumosus seems to be related to M. lupinus and M. metenuus, but more so the latter than the former. This species is quite rare in Oregon with a total of one publicly available state record, although more likely occur within this area. Males are distinctive in that the galeae are somewhat dull dorsally due to delicate distinct shagreening; sterna 2-4 are only moderately shiny medially due to shagreening; the apical areas of the second and third terga have simple, erect to suberect hairs and the galeae are often shiny without shagreening or delicately reticularly shagreened; minimally F4-F10 have longitudinal depressions and the third flagellar segment usually has a well defined ventrolateral depression; the clypeus has small round basal punctures; the length of the penultimate flagellar segment is longer than two times its width. Females are distinctive in that the galeae are moderately shiny above, but somewhat dull due to faint reticular shagreening, notably so in the apical half or less; the first tergum’s crowded basal punctures usually extend to the basal half medially and The medial one-third of the first tergum has punctures that are mostly separated by one puncture width or less, minimally basally and sometimes more; the head hairs are often pale apart from the abundant darker vertex hairs; the scopal hairs are ochraceous to white and usually have more than 6 branches on either side of each rachis. The species name, “plumosus” is likely derived from the structure of the female scopal hairs bearing more branches than most other Melissodes. Due to its singular publicly available Oregonian observation, a species map will be omitted along with the species line in the phenology chart. However, the range-wide phenology will be given below. M. plumosus has been recorded to be active between the months of June and August with peak activity occurring in June and July (Laberge, 1961), but newer data suggest early emergence may commence in May, and activity seems to be present through September (Fig. ??).
Field Markers
♂ Generally a somewhat pale bee with more vibrant hairs on the vertex and dorsal thoracic areas. The clypeus and labrum (the labrum having its margin not darkened) are
yellow and the mandibular bases bear pale yellow macula. The eyes are bluish gray to gray, and on rare occasions, greenish blue and the underside flagellar segments
2-11 are red to yellow (these segments are dark brown on top). The wing veins are brown to brownish red and terga 2-5 are sometimes yellow hyaline apically, but usually
opaque brown. The first flagellar segment’s minimum length is more than, or equal to, one-fifth of the second segment’s maximum length, and segments 3-10 (sometimes also
on 11 basally) have shiny, shallow, longitudinal dorsolateral depressions. The length of the penultimate segments is more than twice its width and the ultimate segment’s
length is more than three times its width. The four maxillary palpal segments decrease from basal to distal in a ratio of approximately 4.5:3.0:3.0:1.0. The fourth sternum
has a wide hyaline flap that is medially often not shallowly emarginate (or if so than very indistinctly), and the fifth and third sterna are noticeably apically convex.
The impunctate areas of the second, third, and fourth sternum are often dull due to dense reticular shagreening. The clypeus has small round basal punctures and the galeae
are somewhat dull dorsally due to delicate distinct shagreening. The length of the penultimate flagellar segment is longer than two times its width. The head hairs are mostly
ochracerous to pale ochraceous, apart from the vertex, which has brown hairs. The thoracic hairs are also mostly ochraceous to pale ochraceous, differing at the long brown
posteromedial mesoscutal hairs, and the scutellar hairs, which tend to be brown as well. The first tergum’s basal fourth-fifths bears long pale ochraceous hairs, and the apical
area of this same tergum has suberect, simple, usually pale brown (sometimes ochraceous), short hairs that almost reach the apical margin. The overall vestiture of the second
tergum is pale ochraceous to white. The basal pubescent band of the second tergum is white, as is the distal pale band. However, the distal pale band is often complete. The
apical area of the second tergum bears simple, suberect, often pale ochraceous (sometimes brown) hairs. The third and fourth terga are similar to that of the second, apart
from the basal tomentum being brown and the apical hairs being largely brown (minimally medially and sometimes more); these bands are often wider than those of M. lupinus.
The fifth tergum is similar to that of the fourth, except the distal band often reaches the apex across the entire tergum. The sixth and seventh terga are entirely covered
in pale brown to ochraceous hairs.
♀ Females are some of the paler to palest Melissodes in Oregon. The eyes are often gray to bluish-gray and the wing veins are dark brown. The underside of F3-F10,
and usually the apical area of F2 are yellow to reddish beneath and the galeae are moderately shiny above, but somewhat dull due to faint reticular shagreening,
notably so in the apical half or less. The first tergum’s crowded basal punctures usually extend to the basal half medially and the medial one-third of the first
tergum has punctures that are mostly separated by one puncture width or less (minimally basally and sometimes more). The scopal hairs are white to pale ochraceous
and tend to bear more than 6 branches on either side of each rachis located at the apical one-half, and the rachis often doesn’t surpass the branched area in length
by much. The head hairs are mostly white to pale ochraceous apart from the darker vertex hairs which tend to take on a more brownish hue. The hairs on the thorax
are mostly pale ochraceous to ochraceous apart from the large dark brown mesoscutal patch of hairs that usually reaches anteriorly to the anterior margins of the
tegulae, and the large dark brown medial patch of hairs on the scutellum. The first tergum has long basal and lateral hairs that are pale ochraceous. The basal area
of the second tergum has long pale ochraceous hairs while the interband zone has suberect comparably simple, short, dark hairs that are dark ochraceous to brown.
The distal pale pubescent band of the second tergum is wide, often becomes medially interrupted, arched, and laterally reaches, or almost reaches, the apes of the
tergum. The apical area of the second tergum is narrowly glabrous marginally, but anterior to this margin, bears suberect to appressed, simple, ochraceous to brown
hairs. The third tergum, is similar to that of the second, apart from the wider uninterrupted distal band that laterally reaches that apex of the tergum, and the
interband zone, which bears appressed, sparse, pale pubescence. The pale ochraceous band on the fourth tergum is wide and positioned apically (never pale hairs near
the medial area of the apical margin). The fifth and sixth terga are covered in dark brown hairs (these hairs are much lighter than those of M. lupinus, the sixth
tergum often having its hairs golden-brown to orange) apart from the lateral tufts, which are pale ochraceous.
Similar Species
M. plumosus is similar to a number of other Melissodes, but particularly closely allied with M. metenuus in both sexes. Males seem to be most similar to those of M. metenuus, but females more so resemble M. minusculus or M. ablusus due to the absence of dark hairs on and below the frons. Although to a lesser extent, both sexes also somewhat resemble those of M. lupinus based on superficial characteristics, but can be separated as described below.
♂ Males are similar to those of M. metenuus in that they share the characteristics of flagellar segments 3-10 (often 11 as well) have depressions, the apical area of the second and third terga bear simple, erect to suberect hairs, and the length of the penultimate flagellar segment is longer than twice its width. However, males of M. plumosus can be separated from those of M. metenuus in that the galeae are somewhat dull dorsally due to delicate distinct shagreening; sterna 2-4 are only moderately shiny medially due to shagreening; and the clypeus has small round basal punctures. In contrast, for M. metenuus, the galeae are dorsally shiny and unshagreened, or if shagreened, then delicately so on less than, or equal to, the apical half; sterna 2-4 are medially shiny and are delicately reticularly shagreened; and the clypeus is coarsely punctate basally. M. plumosus can also faintly resemble M. lupinus superficially, but the two can be easily separated in that M. lupinus never has a depression on F11 or F3, whereas M. plumosus usually has a depression on F11 and always has a well defined one on F3.
♀ Females are fairly similar to M. minusculus and M. ablusus in that the first tergum’s crowded basal punctures usually extend to the basal half medially; the medial one-third of the first tergum has punctures that are mostly separated by one puncture width or less, minimally basally and sometimes more; the head hairs are mostly pale apart from the vertex; the scopae are light; and the galeae are dulled. However, M. plumosus greatly differs from the foregoing species by the degree of dulling on the galeae. This species has moderately shiny galeae above that are somewhat dull due to faint reticular shagreening (prominently son in the apical one-half or less). In contrast, M. minusculus and M. ablusus have distinctly matte and dull galeae that are densely, regularly, and coarsely tessellated. Although the males are similar to M. metenuus, and females share similar sculpturing, the two greatly differ by the facial vestiture. In M. plumosus, the head hairs are mostly light apart from the vertex, whereas in M. metenuus, these hairs are entirely to almost entirely dark brown to black.
Fig. ??. A graph representing the phenology of M. (Callimelissodes) plumosus. The x value is the month, and the y value is the number of documented observations occurring in that month. Data compiled from (Ikerd, 2019; Johnson, 2020; Droege & Maffei, 2025; Bentley & Osborn, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026a; Grinter et al., 2026; Illinois Natural History Survey, 2026; MT James Entomological Collection, Washington State University, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026). Data licensed under CC BY 4.0, CC0 1.0, and CC BY-NC 4.0 as applicable (see "Dataset Licenses").
Flower preference
Very little is known in regards to the flower records, let alone flower preference, of M. plumosus. All flower records included in this list are from reports in the literature or datasets. Each flower has a parenthesized reference listed after it, corresponding to the literary work or dataset in which it was recorded. Currently, M. plumosus has been found atop Encelia californica (Laberge, 1961), Helianthus petiolaris (Laberge, 1961), Iliamna rivularis (MT James Entomological Collection, Washington State University, 2025; GBIF occurrence id 5141240695).
Synonyms
Melissodes plumosa, Laberge, 1961.
Melissodes pullatellus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes pullatellus
Melissodes pullatellus is probably one of the most distinct, and most fascinating bees in this guide. This species has only 14 records, with only four containing any coordinates, all of which occur in Oregon. However, all current known occurrences of M. pullatellus are females with the male still remaining unknown. Although it’s not yet possible to definitively state which species are most closely related to M. pullatellus (due to the male’s absence), it seems that this species shares quite a few similarities to those of M. pullatus of Cuba, both species sharing dark scopae, mostly to entirely black to dark brown thoracic hairs, and similar sizes (both ranging between 9-12 mm.). Although just speculatory, it’s possible that at one point in time M. pullatellus may have been transferred from Oregon to Cuba via anthropomorphic means. Furthermore, given that the only other Cuban Melissodes fall into a tight subgeneric clade apart from Eumelissodes (Melissodes+Ecplectica), the subgenus in which both M. pullatellus and M. pullatus reside, this suggests that M. pullatus may possibly be an island mutation of M. pullatellus from Oregon. Females of this species are distinct in that the scopae are entirely dark with a few faintly paler hairs medially (overall, still quite dark in comparison to most other species), and the thoracic hairs are almost entirely black to dark brown (apart from a minute amount of pale hairs on the dorsal area of the propodeum, the periphery of the scutellum, and near the tegular bases). Of the other Oregonian Melissodes, this species is only allied with one, M. lutulentus, be it very weakly as they only share size and sculpture but greatly differ in vestiture. M. pullatellus has been collected between the months of July and September with the most collections taking place in August. However, a large proportion of these August collections were taken on the same day and in the same location. Looking purely at unique collection events, M. pullatellus has been collected once in July, three times in August, and once in September; too few events to make a definitive statement on phenology (Laberge, 1961).
Field Markers
♀ A very dark bee, seemingly the darkest in Oregon being rivaled only by the darkest forms of M. druriellus. The eyes are often blue, but sometimes gray or greenish blue and the wing veins are usually reddish brown to black, although often dark red. The underside of flagellar segments 3-10, and usually the second in the apical one-third are red to yellow (dark brown on top) and the apical one-third of the first tergum is usually rufescent. The head hairs are entirely dark brown (no light facial hairs or vertex hairs). The thoracic hairs are often completely dark brown except as follow: behind the tegulae, which sometimes (not always) have ochraceous hairs; the metatonum, which sometimes (not always) has ochraceous hairs; and the posterior and dorsal propodeal surfaces, which follow the same pattern of sometimes, be it not always, having ochraceous hairs. The metasomal hairs are almost completly black dark brown except as follows: the long basal hairs of the first tergum, which on rare occasions may be partially ocheracous and dark hairs mixed; and the basal tomentum of the second tergum, which has pale ochraceous hairs mixed. The distal dark band of the second tergum is often separated from the apical margin across the entire tergum and medially interrupted. The distal dark band of the third tergum is similar to that of the second. The distal dark band of the fourth tergum reaches the apex across the entire tergum. The leg hairs are dark brown, differing at the scopae, which are often brown to paler brown with the tibial hairs becoming paler medially (sometimes, the tibial hairs may be pale ochraceous medially and the basitarsal hairs may be pale ochraceous mediobasally). Females are one of the easiest species of Melissodes to identify in Oregon using the marked features of dark scopae and dark thoracic vestiture.
Similar Species
M. pullatellus, as can be discerned from above, is incredibly distinct and doesn't resemble a specific species in particular. However, darker forms of M. druriellus do show some similarities to this species, however, they are easily separated in that the scopae of M. pullatullus is dark and the scopae of M. druriellus is light. As no other Oregonian Melissodes are mostly to entirely black with dark scopae, subsequent comparisons seem unnecessary.
Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) pullatellus. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Bentley & Osborn, 2026; Grinter et al., 2026).
Fig. ??. A graph representing the phenology of M. (Eumelissodes) pullatellus. The x value is the month, and the y value is the number of documented observations occurring in that month. Data compiled from (Laberge, 1961).
Flower preference
Due to the paucity of species records, let alone records that contain floral data, it’s unknown what the dietary preference of M. pullatellus is. The only known floral record of this species is of a species of Grindelia (Laberge, 1961)
Synonyms
Melissodes pullatella, Laberge, 1961.
Melissodes rivalis

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes rivalis
Melissodes rivalis, the western thistle long-horned bee, is a fairly common, highly distinctive, and beautifully marked Oregonian Melissodes. Females are distinctive in that the inner eye margins are parallel to one another rather than converging towards the mandibles and males are distinctive in that the first flagellar segment’s minimum length is 0.4 or more times the second flagellar segment’s maximum length, but the tergal bands are not positioned apically as in M. dagosus. This species has an incredibly rich taxonomic history with five historic synonyms, each seemingly pertaining to different morphs range-wide. However, Scullen (1926) documented a nesting aggregation near the Coos Bay area along the Oregon coast and classified this species as Melissodes mysops, a historical synonym representing darker morph of M. rivalis as outlined in Hogland (2025b). This seems to be in compliance with the general variation statements given by Laberge (1961), although in his paper, the darkest females are historically known as M. desponsiformis, suggesting that while the morph originally described as M. mysops is quite dark in comparison to the more eastward dwellers, it’s not the darkest. Both morphs originally described as M. mysops and M. desponsiformis have been collected in Oregon (Laberge, 1961), implying that darker forms of M. rivalis are common within this state. This species was originally classified in a separate subgenus from Eumelissodes, Heliomelissodes. Species of Heliomelissodes were separated from other subgenera in that the males have their first flagellar segment’s minimum length 0.4 or more times the second flagellar segment’s maximum length and females have parallel inner eye margins (characteristics indicative of M. rivalis in this guide). As only two species existed within the subgenus (M. rivalis and M. desponsus), both occupying vastly different regions of the United States (M. rivalis located westward and M. desponsus located eastward), subgeneric characteristics of Heliomelissodes from Laberge’s (1961) key are sufficient for species level identification of M. rivalis in Oregon. However, Freitas et al (2023) and Wright et al (2020), found that the presence of the subgenus Heliomelissodes renders Eumelissodes paraphyletic (due to Heliomelissodes being nested within Eumelissodes) synonymizing the two subgenera, hence the absence of Heliomelissodes in current-day taxonomy. M. rivalis seems to occur across most of, if not all of Oregon (Fig. ??). This species was historically documented to be active between the months of May and October with peak activity occurring in July and August (Laberge, 1956b). Newer data seem to suggest a similar pattern, though July seems to be of the highest abundance (Fig. ??).
Field Markers
♂ Fairly pale bee with noticeable reddish orange markings on the inner portions of the legs.
The eyes are green to gray and the wing veins are black. The clypeus is paleish yellow (the
posterior margin of the paleish yellow area is angled upward to form an upside down V-shape
in the median one-third), excluding the brownish red apical margin and the black laterobasal
notches denoting the tentorial depressions. The mandibles very rarely (less than 2%) have a
basal yellow macula, but are almost always black. The underside of flagellar segments 2-10
are yellow to red and the sterna are dark reddish brown. The apices of the terga are usually
dark reddish brown. The first flagellar segment’s minimum length is usually no less than one-third
the maximum length of the second segment, if not more. The clypeus is distinctly protuberant,
protruding beyond the eye by three-fourths of an eye's width in profile or more. Because M.
rivalis varies widely and the morphs of which occur in Oregon aren’t definitely known, a full
setal description with its variations will be given below.
The darkest specimens have white to pale ochraceous head hairs, except for the vertex,
which has brown hairs. The mesosoma has white to ochraceous hairs except for the mesoscutum,
which has a patch of dark brown hairs that reaches anteriorly past the middle of the tegulae,
and the scutellum, which has some dark medial hairs. The first tergum has long basal pale hairs,
and short dark apical hairs. The second tergum has a basal pale pubescent band as well as a
distal pale pubescent band. The distal pubescent band is complete, medially narrowed, and separated
from the apical margin. The third tergum’s distal pale pubescent band is medially interrupted
with brown hairs. Terga four, five, six, and seven do not have any pale hairs. The legs mostly
have pale hairs, especially on the outer surfaces of the middle and hind tibiae, except for the
inner surfaces of the hind tibiae, the fore basitarsi, the middle basitarsi, and the hind
basitarsi, which are red to reddish brown (Fig. 6). The palest specimens are similar to that
of the darkest specimens except as follows: the hair on the head is completely white to pale
ochraceous, and has no dark hairs on the vertex. The mesosoma has completely white to pale
ochraceous hairs with no dark patches on the mesoscutum or the scutellum. Terga 2-5 have complete
pale pubescent bands, and tergum 6 has long pale lateral tufts. Sterna 2-5 have red hairs medially,
and pale ochraceous hairs laterally. The sixth sternum has entirely brown hairs. The legs have
white to ochraceous hairs, except for the inner surfaces of the hind tibiae, fore basitarsi,
middle basitarsi, and hind basitarsi, which are red, and the hairs on the outer surfaces of the
fore tarsi and fore tibiae, which are brown.
Male M. rivalis ranges in setal color between these two dark and pale descriptions. As M.
rivalis progressively lightens from the dark extreme to the pale extreme, the pale tergal
hairs start to appear. These pale hairs first develop laterally on the fourth tergum. Then,
lateral pale pubescence develops on the fifth tergum, and the pale band of the third tergum
interrupted with brown hairs becomes complete. Next, the lateral developed pale hairs on the
fourth and fifth terga meet medially creating complete bands, as this happens, the dark hairs
on the head and thorax turn pale. Lastly, on the palest individuals, the dark hairs on the
scutellum turn pale.
♀ Often, at least in Oregon, a dark bee, but females can range in color quite substantially.
The eyes are usually grayish blue and the wing veins are black. The underside of flagellar
segments 2-10 are reddish brown and in profile view, the clypeus protrudes beyond the eye
by one-half to two-thirds of an eye’s width. The inner margins of the eyes are parallel to
one another. Similar to their male counterparts, females can vary largely in vestiture color
as well. Although the vast majority of females that have been found in Oregon are of the
darker varieties (historically M. myops and M. desponsiformis), the extent of variation in
this state is currently unknown. For this reason, two female setal descriptions will be
given, one for the darkest form and one for the lightest, with a subsequent description of
intergrades between these two morphs.
The darkest specimens have black to dark brown head hairs, except for the vertex and face,
which are completely pale ochraceous, and sometimes a mix of ochraceous and dark brown.
Laberge (1956a) states “Metasoma with pale ochraceous hairs except as follows:...” then
proceeds to list mesosomal structures. Later he states “Mesosoma with dark brown to black
hairs except as follows:...” then proceeds to list metasomal structures. This is most likely
a typo and will be herein treated as such. The mesosomal hairs are pale ochraceous except
for the large posteromedian patch of dark brown to black hairs on the mesoscutum that usually
surpasses the anterior margins of the tegulae, but usually isn’t larger than the size of the
dark patch on the scutellum (Fig. 4), and the medial dark brown to black hairs on the scutellum.
The tegulae and pronotal lobes have dark hairs. The general surfaces of the mesepisterna
have dark brown to black hairs and the lateral surfaces have dark brown hairs on the lower
two thirds and sometimes higher. The lateral surfaces of the propodeum also have dark brown
to black hairs. The metasoma has dark brown to black hairs except for the basal area of the
first tergum, which has long dark brown hairs, and the basal band of the second tergum, which
usually has some pale hairs. The leg hairs are dark brown to black other than the ochraceous
to yellow scopa, and the dark red to black hairs on the inner surfaces of the hind tibia and
hind basitarsi. The palest specimens have white to grayish white head hairs, except for the
vertex which has some brown hairs. The surfaces of the mesepisterna have white to pale
ochraceous hairs. The pronotal lobes have white to pale ochraceous hairs. The large
posteromedian patch of dark brown to black hairs on the mesoscutum reaches the middle
portion of the tegula across the mesoscutum, and sometimes surpasses it. The first tergum
has long pale basal hairs. The basal band of the second tergum is white, and the distal
band is pale ochraceous, narrowed medially, usually interrupted medially, and does not reach
the apical margin. The third and fourth tergum each have a distal pale band of pubescence,
and the band of the fourth tergum is usually positioned apically. The fifth tergum has small
white lateral tufts of hairs. The second, third, and fourth sterna have reddish brown hairs
medially, and pale hairs apicolaterally. The inner surfaces of the fore basitarsi have red
to reddish brown hairs, along with the inner surfaces of the middle bastitarsi, hind bastitarsi,
and hind tibiae. The remainder of the leg hairs are black except for the ochraceous to yellow scopa.
M. rivalis ranges in setal color between these two dark and pale descriptions. As M. rivalis
progressively lightens from the dark extreme to the pale extreme, pale tergal hairs start to
appear. These pale hairs first develop on the base of the first and second tergum. Next, lateral
pale pubescence develops on the second tergum, then the third, and subsequently the fourth. Lastly,
lateral pale tufts develop on the fifth tergum. In the palest specimens, the lateral pale pubescence
become complete bands, reaching completion in the same order as the appearance of the lateral pale
pubescence.
Similar Species
Given how drastically different both sexes are due to them being in a unique clade of their own (the Heliomelissodes clade inside Eumelissodes), structurally, these species resemble none other in Oregon. With how distinctive the male's antennal structure is, this character is enough to identify M. rivalis to species in Oregon. Similarly, the inner eye margins of females being parallel to one another coupled with the clypeal protrusion and jet-black inner hind basitarsal hairs are sufficient for species-level identification as well. Due to these stark differences, similar species comparisons of both sexes will be omitted.
Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) rivalis in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Johnson, 2020; Best et al., 2022a; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026; Motz, 2026; The International Barcode of Life Consortium, 2026). Data liscenced under CC BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Eumelissodes) rivalis in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Ikerd, 2019; Johnson, 2020; Best et al., 2022a; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; Bentley & Osborn, 2026; Best, 2026; Illinois Natural History Survey, 2026; Motz, 2026; The International Barcode of Life Consortium, 2026) and range-wide data compiled from (Ikerd, 2019; Johnson, 2020; Best et al., 2022a; Carril et al., 2023; Ikerd & Engler, 2023; Prescott et al., 2023; South Dakota State University, Severin-McDaniel Insect Research Collection, 2023; Texas A&M University Insect Collection, 2023; Droege & Maffei, 2025; Scott, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Best, 2026; Brigham Young University, Arthropod Collection, 2026; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Fägerström, 2026; Gibbs, 2026; Grinter et al., 2026; Harvard & Morris, 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Mertz et al., 2026; Mississippi Entomological Museum, 2026; Motz, 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Native, 2026; Northern Arizona University, 2026a; Oram, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
M. rivalis is most certainly an oligolect of Asteraceae with a strong preference towards thistles (as the vernacular name suggests), specifically in the genus Cirsium (Laberge, 1956b). According to Laberge (1956b), of the total 201 M. rivalis with floral data attached, 171 were found atop Cirsium. This renders Cirsium to be favored by M. rivalis 85.07% of the time by M. rivalis.
Synonyms
Melissodes desponsiformis Cockerell, 1905; Melissodes habilis Cockerell, 1925; Melissodes hexacantha Cockerell, 1905; Melissodes mysops Cockerell, 1905; Melissodes nigrosignata Cockerell, 1905.
Melissodes robustior

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes robustior
Melissodes robustior is a somewhat distinctive species within Oregon, often nearly resembling those of M. agilis in vestiture coloration (ochraceous-yellowish), but having a dark dorsal thoracic patch of hairs. Males are quite distinctive, not resembling one species in particular, but females are similar to M. paululus in a few characters. Interestingly, this species is known to be one of the commonest found Eumelissodes on the Pacific Coastal region (Laberge, 1961), but similar to M. agilis (a prolifically common Melissodes across its entire range), M. robustior has much fewer documentations in Oregon than implied by such statements with a mere total of 72 state records; outpacing M. agilis by 4 observations. The similarities between the two species don’t cease here, M. robustior seems to have a strong preference toward the flowers of the genus Helianthus, the genus on which M. agilis is known for being an oligolege, and both species have distinctly shagreened galeae in both sexes. Males are distinctive in that the mandibular bases are black; the scutellar and mesoscutal hairs are medially black; the galeae are matte and dull due to dense tessellation; the first flagellar segment’s maximum length is more than one-third of the third segment’s minimum length; the labrum is partially dark, bearing a mediobasal pale macula; and the tergal rims are colorless to yellow hyaline. Females are distinctive in that the interband zone of the second tergum is usually impunctate, or if there are punctures, then they are irregularly shaped and sized and indistinct or the punctures only occur on the lateral raised areas; the apical area of the second tergum is dull due to shagreening and bears many appressed to subappressed dark hairs; the mesoscutum clearly has a dark brown to black patch of posteromedial hairs; the clypeus rarely protrudes beyond the eye by one-half of an eye’s width in profile, almost always by less; and the apical of the third tergum has dark hairs on the medial one-third or more with the distal pale band reaching the apex laterally. M. robustior has nearly been found across the entire state with a high probability of it occurring in unsampled areas (Fig. ??). Laberge (1961) noted that this species has been recorded to be active between the months of May through October with peak activity allotted to June, July, and August; a similar pattern as seen in figure ??.
Field Markers
♂ Intermediate in vestiture due to the darkend dorsal thoracic patch, but overall, M. robustior males
tend to be quite pale. The eyes are gray to brown to greenish yellow and the wing veins are brownish
red to dark red. The clypeus is yellow apart from its tentorial pits which are dark, and is apical
margin, which is testaceous and the labrum has a pale mediobasal macula. The mandibles are entirely
black with no basal macula and the underside of flagellar segments 2-11 (and sometimes the apical area
of the first segment) are red to yellow. The apical rims of the terga are colorless to yellow hyaline.
The first flagellar segment’s minimum length is often approximately one-fourth to one-fifth of the
second segment’s maximum length. The galeae are dull, rough, and matte due to dense shagreening. The
first flagellar segment’s maximum length is more than one-third of the third segment’s minimum length.
The head hairs are mostly pale ochraceous to white apart from the vertex, which is sometimes ochraceous
but usually has some brown as well. The thoracic hairs are mostly ochraceous, becoming more pale in the
posterior and lateral areas. The scutellum is ochraceous with many brown medial hairs and the mesoscutum
has a large dark posteromedial patch of hairs that is minimally equal to the size of the scutellar dark
patch, and usually two times its area. The tegulae often bear brown hairs. The basal area of the first
tergum bears long pale ochraceous hairs that, in length, reach the apex of the tergum across the entire
tergum, these hairs becoming appressed near the apical margin. The basal pale band of the second tergum
is pale ochraceous to white. The interband zone of the second tergum bears erect to suberect, long, pale
ochraceous hairs and the distal pale pubescent band is usually medially interrupted and narrow. The
apical area of the second tergum bears many appressed to suberect, pale ochraceous hairs when unworn.
The third, fourth, and fifth terga are similar to that of the second, apart from the distal pale bands,
which are complete and become closer to the apex of the tergum with each subsequent terga until the
apical margin is usually reached on the fifth tergum. The sixth and seventh terga bear gold-colored to
ochraceous hairs.
♀ Often a pale ochraceous to yellowish bee with a distinct dark thoracic patch (similar to the male,
but more distinctly contrasting with the surrounding thoracic hairs). The eyes are faintly green to dark
gray and the underside of flagellar segments 3-10 are brownish red to red. The wing vines are brown to
dark red and the apical rim of the first tergum, which is hyaline to rufescent, but only narrowly so.
The interband zone of the second tergum is usually impunctate, or if there are punctures, then they are
irregularly shaped and sized and indistinct or the punctures only occur on the lateral raised areas.
The apical area of the second tergum either has many suberect to appressed pale to dark hairs and the
surface is usually dull due to shagreening. The apical area of the third tergum is laterally obscured
due to the distal band which reaches the apex laterally, and bears dark hairs in the medial one-third
(and sometimes more). The head hairs are mostly pale ochraceous to white, apart from the vertex, which
bears brown hairs, and the occipital area, which bears ochraceous hairs. The thoracic hairs are white
to pale ochraceous posteriorly and laterally, and ochraceous dorsally except as follows: the scutellum,
which mostly covered in dark brown hairs, with some ochraceous fringe surrounding its periphery; and
the mesoscutum, which bears a large dark posteromedial patch of hairs that can be two times as large
as the dark patch on the scutellum, but often falls short. The first tergum bears pale ochraceous to
ochraceous hairs basally, becoming glabrous apically. The basal band of the second tergum is white.
The interband zone of the second tergum is scattered with abundant erect, dark brown, short hairs.
The distal pale band of the second tergum is often complete but posteriorly notched/narrowed, pale
ochraceous to white, and wide. The apical area of the second tergum bears appressed to suberect,
dark brown, short hairs. The third tergum is similar to that of the second, differing at the basal
tomentum, which is brown, and the distal pale pubescent band, which often reaches the apex of the
tergum laterally. The distal pale band of the second tergum is complete and isn’t margined with dark
hairs. The fifth and sixth terga are mostly covered in dark hairs apart from the lateral tufts,
which are pale. The hairs on the inner surfaces of the hind bastitarsi are reddish brown to dark
red.
Similar Species
Although the females are fairly distinctive in Oregon, they share a few characteristics with those of M. paululus and M. agilis, although more so the former than the latter. Males also aren’t directly related to any other species in Oregon given M. montanus hasn’t been recorded in this state (unless M. montanus and M. robustior have hidden cryptic species within them that are unknown). However, given the dark dorsal thoracic hair patches and overall pale vestiture, males can weakly superficially resemble a few other Eumelissodes, although these comparisons below will be light given the large differences.
♂ In Oregon, male M. robustior is likely most similar to M. grindeliae given both species share the characters of the first flagellar segment’s maximum length being more than one-third of the third segment’s minimum length, entirely black mandibles with no maculations, and the dark dorsal thoracic patches of hairs. However, the two distinctly differ in the galeal sculpturing. That being, M. robustior has very rough, matte, and dull galeae due to dense shagreening, and M. grindeliae has shiny, mostly unshagreened galeae apart from the tips.
♀ Females seem to be only distinctly similar to M. paululus females in vestiture and, in certain areas, sculpture, but as shown above, can share a few characteristics with M. agilis. M. robustior can be separated from those of M. paululus in that the interband zone of the second tergum is usually impunctate, or if there are punctures, then they are irregularly shaped and sized and indistinct or the punctures only occur on the lateral raised areas. In contrast, the interband zone of the second tergum of M. paululus has punctures that are distinct, regular, and round across the entire tergum (the punctures might be more abundant laterally than medially). Although similar to M. agilis in galeal sculpture, vestiture color, and, in areas, integumental color, M. robustior differs in the presence of the dark thoracic patch and the dark hairs on the inner surfaces of the hind basitarsi. M. agilis has the entirety of their thoracic hairs ochraceous to rusty-red and the inner surface of the hind basitarsi are light, not distinctly contrasting with the rest of the scopa.
Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) robustior in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Best et al., 2022a; Bentley & Osborn, 2026; Best, 2026; Johnson, 2026; MT James Entomological Collection, Washington State University, 2026; The International Barcode of Life Consortium, 2026). Data liscenced under CC BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Eumelissodes) robustior in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Ikerd, 2019; Best et al., 2022a; Bentley & Osborn, 2026; Best, 2026; Johnson, 2026; MT James Entomological Collection, Washington State University, 2026; The International Barcode of Life Consortium, 2026) and range-wide data compiled from (Ikerd, 2019; Johnson, 2020; Best et al., 2022a; Carril et al., 2023; Ikerd & Engler, 2023; Prescott et al., 2023; South Dakota State University, Severin-McDaniel Insect Research Collection, 2023; Texas A&M University Insect Collection, 2023; Droege & Maffei, 2025; Scott, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Best, 2026; Brigham Young University, Arthropod Collection, 2026; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Fägerström, 2026; Gibbs, 2026; Grinter et al., 2026; Harvard & Morris, 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Mertz et al., 2026; Mississippi Entomological Museum, 2026; Motz, 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Native, 2026; Northern Arizona University, 2026a; Oram, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
As seen above, M. robustior is certainly an oligolege of Asteracea with a preference for the genus Helianthus (Laberge, 1961). Although this genus was the highest single value for any of the other flower genera, Helianthus was only chosen 41.39% of the time by M. robustior (178 out of the 430 floral records), suggesting that this species, while having a strong preference, may be less specialized than other Oregonian Melissodes. Of the notable flower genera (apart from Helianthus), M. robustior is also fairly commonly found atop Corepsis (Laberge, 1961) and Madia (MT James Entomological Collection, Washington State University, 2025).
Synonyms
None.
Melissodes saponellus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes saponellus
Melissodes saponellus is similar to M. pullatellus, although not in the traditional morphological sense. Both M. saponellus and M. pullatellus are only “known” from the female sex and are highly distinctive with only one other Oregonian species coming close to resembling them. However, the similarities stop here as neither species resembles the other. M. saponellus has fairly little documentation on it given its few historical records (Laberge, 1961 only examined 18 females), but newer data will allow for a more thorough treatment. Although the male isn’t “known” yet (no formal description), it seems as if there is a high possibility that it was recently discovered in Utah via DNA barcoding and is awaiting its morphological treatment (iBOL, 2025l; GBIF record 2248749984). Due to this, only the female field markers will be given below. This species is distinctive in that it’s one of the only two females in Oregon (one of three range-wide) that have translucent tergal rims, much as in most Eumelissodes males. On top of this, the hairs on the mesoscutum are mostly pale with no, or very few, dark hairs (no dark patch present) and the hairs on the apical areas of the second and third terga are white. Laberge (1961) noted that M. saponellus was originally only known from a few specimens collected in Colorado, Utah, Washington, and Oregon, however, newer data show a much larger range outside these states. Given that only three publicly available records of this species are from Oregon (placing it fourth/fifth most common in the state, tying with M. sternsi), the state line in the phenology graph will be omitted with only the range-wide graph given, but an Oregonian distribution map will still be present. This species has been found in Malheur County, Oregon (iBOL, 2025), Harney County, Oregon (iBOL, 2025), Umatilla County, Oregon (iBOL, 2025).This species has been collected from April to September with peak activity occurring in June (however, this may be sampling bias).
Field Markers
♀ Often a very pale bee in vestiture, usually seeming more pale from afar due to the hyaline tergal margins creating light apical “integumental bands” (not the hair). The eyes are often grayish yellow to grayish green and the wing veins are brownish red to dark red. The underside of flagellar segments 3-10, and usually the second in the apical one-third are red to yellow (dark brown on top) and the apical area of the first tergum is widely colorless hyaline, transitioning to red-yellow basally (then opaque). The apical areas of the second, third, and fourth terga are also colorless to faintly yellow hyaline. The apical area of the third tergum is narrower than the distal pale band on the second tergum and the hairs on the inner surfaces of the hind basitarsi are reddish orange to yellow. The head hairs are entirely ochraceous to white. The thoracic hairs are pale ochraceous to white laterally and yellow to ochraceous dorsally. The basal area of the first tergum bears white hairs and the lateral rounded projections of the apical area that reach anteriorly bear white, short, subappressed hairs. However, posterior to these projections, the apical area of the first tergum bears appressed, white pubescence. The basal pale band of the second tergum is white. The interband zone of the second tergum bears white, short, subappressed hairs and the distal pale band is white, laterally connected to the basal band, wider medially than the apical area, and reaches the apex of the tergum laterally. The apical area of the second tergum bears brown to ochraceous, suberect, short hairs. The third tergum is similar to that of the second, differing at the distal pale pubescent band, which often reaches the apex across most of to the entirety of the tergum; and the apical, which is usually absent, but when present, is very narrow, reduced to one-fourth of the tergum’s width longitudinally (or less). The fourth tergum is similar to that of the second, but the distal pale band reaches the apex across the entire tergum. The fifth and sixth terga are mostly covered in ochraceous hairs mediobasally, but laterally, these hairs often become white.
Similar Species
The female character of hyaline tergal rims separates M. saponellus out from 96% of other Oregonian Melissodes. The only other female that shares this trait, and is therefore the only other female that is closely related to M. saponellus in Oregon, is M. vernalis. However, even though M. vernalis is included in this guide due to its range overlapping Oregon (see Melissodes vernalis treatment), it should be known that there has yet to be a state collection of this species.
♀ Females of M. saponellus and M. vernalis, though sharing a very diagnostic character, largely differ in a few traits. The mesoscutum of M. saponellus has no, or very few, dark brown hairs, the hairs on the apical areas of the second and third terga are white, and the hairs across the fifth and sixth terga are ochraceous to white. In contrast, the mesoscutum of M. vernalis has a large brown patch of hairs, the hairs on the apical areas of the second and third terga are dark brown, and the hairs across the fifth and sixth terga are usually dark brown as well. Given no other females in Oregon share the hyaline tergal margins, subsequent comparisons of other species based on shared vestiture seem unnecessary.
Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) saponellus in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (The International Barcode of Life Consortium, 2026). Data liscenced under CC BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Eumelissodes) saponellus. The x value is the month, and the y value is the number of documented observations occurring in that month. Data compiled from (Ikerd, 2019; Sheffiled, 2019; Johnson, 2020; Carril et al., 2023; Ikerd & Engler, 2023; Bentley & Osborn, 2026; Brigham Young University, Arthropod Collection, 2026; Grinter et al., 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Native, 2026; Northern Arizona University, 2026a; The International Barcode of Life Consortium, 2026; University of Arizona Insect Collection, 2026b). Data licensed under CC BY 4.0 and CC BY-NC 4.0 and CC0 1.0 as applicable (see "Dataset liscences").
Flower preference
With the total records of M. saponellus summing to 18 when Laberge (1961) wrote his species treatment, the lack of data resulted in the lack of flower records as well as definitive floral preference. Apart from three collection events noting this species on Platyschkuhria integrifolia (old synonym), Chaenactis stevioides (presented as “Chaenactis stenioides,” a possible misspelling), and Sphaeralcea sp. from Laberge’s (1961) distribution data section, no flower records have been published. Herein will be a list of current known flower records of M. saponellus based on GBIF datasets by extracting flower names from historical labels. With a total of only 31 of the 178 publicly available M. saponellus records containing floral data, a proper analysis on floral preference cannot definitely be given. All flower records included in this list are from reports in the literature or datasets. Each flower has a parenthesized reference listed after it, corresponding to the literary work or dataset in which it was recorded. Currently, M. saponellus has been found atop Encelia farinose (Illinois Natural History Survey, 2025; GBIF record 3801377092), Erigeron compositus (Carril et al., 2023; GBIF record 3801852152), Haplopappus croceus (Illinois Natural History Survey, 2025; GBIF record 3801940083), Helianthus sp. (Carril et al., 2023; GBIF record 3421528374), Helianthus petiolaris (Carril et al., 2023; GBIF record 3421520757), Heterotheca villosa (Carril et al., 2023; GBIF record 3421439862), Hymenopappus filifolius (Carril et al., 2023; GBIF record 3421483446), Machaeranthera sp. (Carril et al., 2023; GBIF record 3421519785), Malacothrix fendleri (Illinois Natural History Survey, 2025; GBIF record 3801375159), Poliomintha incana (Illinois Natural History Survey; GBIF record 3801853108), Platyschkuhria integrifolia (Laberge, 1961; old synonym), Rubus bifrons (MT James Entomological Collection, Washington State University, 2025; GBIF record 5141256798), Scabrethia scabra (Carril, et al., 2023; GBIF record 3421437689), Sphaeralcea sp. (Carril et al., 2023; GBIF record 3421446249), Sphaeralcea coccinea (Carril et al., 2023; GBIF record 3421528387), Stanleya viridiflora (Bently & Osborn, 2025; GBIF record 1913918095), Tetradymia canescens (Bently & Osborn, 2025; GBIF record 657818464), Thelesperma subnudum (Carril et al., 2023; GBIF record 3421489107), Xylorhiza wrightii (Illinois Natural History Survey 2026; old synonym; GBIF record 3801939110). Although many records across several datasets include the same flower record, only one GBIF record and dataset license citation are provided for each to keep this concise.
Synonyms
None.
Melissodes semilupinus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes semilupinus
Melissodes semilupinus, the composite long horn bee, very closely resembles M. bimatris and sometimes M. menuachus in the male sex, but females resemble M. menuachus much more than they do M. bimatris. However, both sexes can be distinguished from all other species in Oregon by only a few characteristics. Males are distinctive in that the mandibular bases have a yellow macula and the labrum is mostly yellow and has a darkened border; the first flagellar segment’s minimum length is usually more than, or equal to, one-third of the third segment’s maximum length; the first tergum has an obvious and well developed distal band consisting of appressed or subappressed, dense, white pubescence that reaches and obscures the apical margin across the entire tergum (sometimes the apical band is worn); the first flagellar segment’s maximum length is more than one-third of the third segment’s minimum length. Often, males can just be identified by the combination of the apical hairs on the first tergum obscuring the apical margin (reducing possible species to two, M. semilupinus and M. bimatris) and the presence of mandibular macula. Females are distinctive in that the fifth and sixth terga do not have pale ochraceous to white lateral tufts of hairs; the mesepisternal hairs are black to dark brown on the lower-lateral areas; the length of the second flagellar segment is faintly, but noticeably longer than its width and the hairs on the inner surfaces of the hind basitarsi are are dark brown to brown. In Oregon, M. semilupinus seems to be similar in rarity to M. glenwoodensis, having a total of 5 publicly available state records mostly scattered around the mid to north eastern areas of the state. Although limited records were available at the time of his revision (112), Laberge (1961) noted that this species was originally collected from August to October, with peak activity occurring in September. Newer data seem to follow a similar pattern, however, M. semilupinus has been recorded in July (Fig. ??).
Field Markers
♂ A very pale bee with no bright dorsal thoracic hairs or bright vertex hairs (mostly a dull white color).
The eyes are green to yellowish brown and the clypeus is yellow. The labrum is mostly white except for
the apical margin, which is narrowly brown and the bases of the mandibles are yellow. The wing veins are
pale brownish red to red and flagellar segments 2-10 are red to yellow on the underside and dark red to
brown on top (first segment usually completely dark). The apices of the terga are colorless to yellow
hyaline. The first flagellar segment’s minimum length is about two-thirds of its maximum length, and
about one-fifth or somewhat more than the maximum length of the second segment. Flagellar segments 9
and 10 (usually 7 and 8 as well) narrow toward their base, and the distal most segments are faintly
crenulate laterally. The penultimate segment is about three times as long (maximum) as it is wide
(minimum). The first flagellar segment’s maximum length is more than one-third of the third segment’s
minimum length. The hairs on both the head and the thorax are dull white without becoming brighter on
the dorsal thoracic areas and vertex. The first tergum has long basal dull white hairs and an obvious
and well developed distal band consisting of appressed or subappressed, dense, white pubescence that
reaches and obscures the apical margin across the entire tergum (sometimes the apical band is worn).
The second tergum has white basal pubescence and pale suberect, bristle-like hairs on the interband
zone. The distal pubescent band of the second tergum is anteriorly separated from the apex of the
tergum by about one-half to one times the length of the medial area of the pale band and is not
interrupted medially. Terga 3-5 are similar to that of the second tergum except for the interband
zones, which in addition to the bristle-like hairs there are also delicate, sparse, white, appressed
pubescence. Also, the distal bands of terga 3-5 become closer to the apical margin with each tergum.
The sixth and seventh terga are covered in yellowish to white pubescence.
♀ Females are fairly pale, similar to M. menuachus or the lighter females of M. bimatris in color.
The eyes are bluish gray and the apical margin of the clypeus is often rufescent. The wing veins
are dark brown and the underside of flagellar segments 4-10 and sometimes 3 as well are rufescent
(F2 and sometimes F3 completely black). The apical areas of terga 2-4 are often black and opaque,
but sometimes they can be a dark brownish red and the length of the second flagellar segment is
faintly, but noticeably longer than its width. The head hairs are white excluding the vertex, which
is usually ochraceous, and the labral and mandibular hairs, which are usually brown. The thoracic
hairs are bright ochraceous to pale ochraceous dorsally, and white to pale ochraceous posteriorly
and laterally apart from the lower lateral and anterior mesepisternal surfaces, which bear dark
brown hairs. The first tergum’s basal area has long, pale, ochraceous hairs that reach the apical
margin at extreme lateral areas but not medially. The apical area of the first tergum is glabrous.
The second tergum’s white basal pubescence laterally connects to the distal pale band. The interband
zone of the second tergum has rather simple, suberect to appressed, sparse, long pale hairs. The
distal pale pubescent band is two times the length of the apical glabrous area laterally, and medially,
the band is narrowed and notched measuring only half the length of the apical glabrous area. The
distal pale band and the basal band of the second tergum often merge both medially and laterally.
The third tergum is similar to that of the second, except the basal tomentum is dark brown and the
distal pale band is separated from the apical by about half the width of the distal band, and the
apical area bears comparably simple, suberect, brown to white hairs. The fourth tergum is similar
to that of the third, but often reaches the apex across the tergum. The fifth and sixth terga are
entirely covered in brown hairs excluding the white lateral tufts. The hairs on the inner surfaces
of the hind basitarsi are are dark brown to brown.
Similar Species
The male characteristic of having an apical band of dense hairs on the first tergum that obscures the apical margin across the entire tergum (even medially) is highly distinctive within Oregonian Melissodes, separating it from all other species apart from one (M. bimatris). Females have a similar distinctive characteristic in that the length of the second flagellar segment is faintly, but noticeably longer than its width. This trait separates female M. semilupinus from all other Oregonian Melissodes as well apart from one, M. menuachus. Taking this into account, comparisons of the two sexes will primarily focus on the species that shares the distinctive trait, then each comparison will also quickly highlight how to separate species with similar vestiture.
♂ Although males can resemble those of M. bimatris due to the shared dense hair band on the first tergum that obscures the apical margin and the distal flagellar segments being crenulate, M. semilupinus can be quickly separated by the presence of mandibular maculations and the dorsal thoracic hairs being dull white rather than brighter ochraceous. For contrast, M. bimatris has its mandibles entirely dark and the dorsal thoracic hairs are often darker ochraceous to brown. Given the overall very pale and somewhat desaturated color of M. semilupinus males, they can superficially resemble many other males within the genus. However, the trait of the apical band on T1 and the transverse apical margin of the fourth sternum (separating males from all other Callimelissodes) narrows down the number of Oregonian species significantly. Something of note, other Eumelissodes males (apart from M. semilupinus and M., bimatris) may have dense apical hairs on T1, but these hairs won’t obscure the apical margin medially.
♀ While as straight forward to seperate as the males, the characteristic to separate females is somewhat less pronounced. Females of M. semilupinus most closely resemble M. menuachus given these are the only two Oregoninan species in which the length of the second flagellar segment is faintly, but noticeably longer than its width. However, although they share similar antennal structures, M. semilupinus can be separate from those of M. menuachus by the lack of pale ochraceous to white lateral tufts of the fifth and sixth terga, and the mesepisternal hairs being black to dark brown on the lower-lateral areas. For contrast, the fifth and sixth terga of M. menuachus have pale ochraceous to white lateral tufts of and the mesepisterna have no dark hairs. Similar to the male, the general vestiture color of female M. semilupinus can superficially resemble a number of Melissodes in multiple subgenera. As stated above, only two female Oregonian Melissodes have the characteristic of the length of the second flagellar segment being faintly, but noticeably longer than its width, and this trait is what separates M. semilupinus from all (apart from one) other Melissodes in Oregon. However, the length of the second flagellar segment is a very subtle feature that, without comparisons, is quite challenging to correctly identify. Given its diagnostic nature, comparisons of the second flagellar segment of females who have “short” vs “long” segments will be provided below to aid in accurate identification.
Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) semilupinus in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Johnson, 2020; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; The International Barcode of Life Consortium, 2026). Data liscenced under CC BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Eumelissodes) semilupinus in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Ikerd, 2019; Johnson, 2020; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; The International Barcode of Life Consortium, 2026) and range-wide data compiled from (Ikerd, 2019; Johnson, 2020; Carril et al., 2023; Texas A&M University Insect Collection, 2023; Scott, 2025; Bentley & Osborn, 2026; Brigham Young University, Arthropod Collection, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Northern Arizona University, 2026a; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
Historically, M. semilupinus was known as an oligolege of Chrysothamnus, similar to M. bimatris, with all known female collections with floral data attached being taken from this genus (Laberge, 1961). The flower records given in Laberge (1961) are as follows: Chrysothamnus sp., Cleome sp., Ericameria nauseosa (old synonym), Euthamia occidentalis (old synonym), and Isocoma sp. However, the only known number of examined individuals was a total of 112, likely many of these having no floral data attached. Newer data highlight 8 new genera that M. semilupinus has been found on, and a slightly different flower preference than that hypothesized in Laberge (1961). All flower records included in this list are from reports in the literature or datasets. Each flower has a parenthesized reference listed after it, corresponding to the literary work or dataset in which it was recorded. Currently, M. semilupinus has been found atop Chrysothamnus sp. (Laberge, 1961), Chrysothamnus greenei (Carril et al., 2023; GBIF record 3421474495), Chrysothamnus viscidiflorus (Carril et al., 2023; GBIF record 3421535830), Cleome sp. (Laberge, 1961), Cleome lutea (Carril et al., 2023; GBIF record 3421535830), Dieteria canescens (MT James Entomological Collection, Washington State University, 2025; GBIF record 5141269694), Ericameria nauseosa (Laberge, 1961; old synonym), Ericameria parryi (Carril et al., 2023; GBIF record 3421481239) Euthamia occidentalis (Laberge, 1961; old synonym), Gutierrezia (Illinois Natural History Survey, 2025; GBIF record 3801941089), Helianthus (Illinois Natural History Survey, 2025; GBIF record 3801337126), Isocoma sp (Laberge, 1961); Lorandersonia linifolia (Carril et al., 2023; GBIF record 3421529199), Senecio sp. (Carril et al., 2023; GBIF record 3421450645), Senecio flaccidus (Carril et al., 2023; GBIF record 3421522175), Senecio lugens (Illinois Natural History Survey, 2026; GBIF record 3801939121), Senecio spartioides (Carril et al., 2023; GBIF record 3421531243), Stephanomeria tenuifolia (Carril et al., 2023; GBIF record 3421496322), Tamarix sp. (Carril et al., 2023; GBIF record 3421445063), Thelypodium integrifolium (Carril et al., 2023; GBIF record 3421445952). These flower records are derived from 482 M. semilupinus records, 116 of which had floral data attached. As sufficient floral data is now present, an analysis of flower preference can be given. Contrary to Laberge (1961), the flower genus with the highest number of M. semilupinus collections and highest number of unique events (collections where latitude, longitude, and date are different) is Ericameria with a total of 53 records and 30 unique events. Another notable genus is Chrysothamnus, seemingly the second most favored genus of flower by M. semilupinus with a total of 25 collections and 19 unique events. These plant genera comprise 67.24% of collections and 74.24% of unique events. It’s somewhat unsurprising that M. semilupinus favors these genera, especially with how similar Ericameria and Chrysothamnus are to one another.
Synonyms
Melissodes menuacha semilupina, Cockerell, 1905; Melissodes chrysothamni, Cockerell, 1905; Melissodes semilupina, Laberge, 1961.
Melissodes stearnsi

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes stearnsi
Melissodes stearnsi is perhaps one of the most strikingly marked and highly distinctive species in Oregon. This is due to the sternal structure of the males, with most sterna being widely hyaline apically, the tergal vestiture of males and females being diffuse and puberulent, and the females having long curled galeal hairs. Neither sex seems to be directly related to, or remotely resemble any other Oregonian, or for that matter genus-wide, Melissodes, although they can bear a resemblance to a few Anthophorula and sometimes at afar, Diadasia. Although largely undersampled, from the known data, it’s interesting to see that M. stearnsi seems to not be an oligolege, data presented in Laberge (1961) show that this species has been abundantly found on flowers from the family Asclepiadaceae, albeit the majority of these were males (see “Flower preference”). Given that no other Melissodes are known to have the diffused puberulent tergal pubescence of M. stearnsi, and that other species of bees are believed that metasomal vestiture may play a part in floral-host camouflage to aid in some sort of evolutionary advantage (Portman et al., 2016), it isn’t outside the realm of possibilities that the setal morphology of M. stearnsi may be linked to an unknown preferred floral host; although, this is just speculation. Three publicly available records of this species have been collected in Oregon (2 mentioned by Laberge, 1961), a similar commonality to that of M. saponellus in this state, placing M. stearnsi the sixth rarest Melissodes in Oregon. Of these records (sourced from GBIF), only one has coordinates attached, that being in Josephine County, Oregon, and therefore a distribution map as well as the state line in the phenology graph will be omitted. This species was historically known to fly from March to October with peak activity occurring in June through August (Laberge, 1961). Newer data seem to agree, however, M. stearnsi has been collected 16 times in May as well (Fig. ??).
Field Markers
♂ A very pale bee with no lightning or darkening on the vertex or dorsal thoracic areas as in
other Melissodes. The mandibular bases and clypeus are cream to pale yellow and the labrum (having
its margin not darkened) is yellow. The eyes are bluish gray to gray, and on rare occasions, greenish
blue and the underside flagellar segments 2-11 are red to yellow (these segments are dark brown on top).
The wing veins are black to brown and the apical areas of terga 1-5 are often near-hyaline to brownish
hyaline. The first flagellar segment’s minimum length is less than one-sixth of the second segment’s
maximum length, and segments 5-9 (sometimes also on 10, never on 11) have shiny, incredibly shallow
and indistinct longitudinal dorsolateral depressions if noticeable at all. The length of the penultimate
segment is more than twice its width and the ultimate segment’s length is less than, or equal to,
two times its width. The length of the ultimate flagellar segment is less than three times its width,
but more than twice its width. The galeae bear many straight hairs that are very short. The fourth
sternum has a wide hyaline flap that is medially emarginate and, in length, equal to the basal opaque
sternal area or longer. The second and third sterna are similar to the fourth, but the wide hyaline
flap is shorter and less produced and the fifth sternum is apically convex. The basal area of the
first tergum bears long pale ochraceous basal hairs that reach the apex of the tergum laterally, but
not medially. The apical area is glabrous. The basal area of the second tergum bears many dense white
hairs. The interband zone of the second tergum has suberect pale hairs mixed with largely spaced more
complex pale hairs (more complex hairs present than that of the female). The apical area of the second
tergum is often apubescent with the diffused pubescence not reaching the apical margin. The third and
fourth terga are similar to that of the second, however, the basal tomentum is often paler brown. The
fifth tergum is similar to that of the second, apart from the apical area, which has diffused puberulent
pubescence reaching the apical margin medially, often unworn. The sixth and seventh terga are mostly
covered in dark ochraceous hairs apart from the lateral tufts, which are white.
♀ Generally a very pale bee, however, if the diffuse tergal pubescence is severely worn, the darker
integument may show through. The eyes are often gray to bluish-gray the wing veins are dark brown.
The clypeus is usually rufescent anteriorly in the medial areas. The underside of F3-F10, and
usually the apical area of F2 are yellow to reddish beneath the apical areas of terga 1-4 are
dark brownish red. The galeae have abundant long hooked hairs above. The head hairs are mostly
pale ochraceous, becoming more of a yellow towards and on the vertex. The thoracic hairs are mostly
laterally ochraceous and dorsally yellow, but the mesoscutum has a bare posteromedial area
(sometimes having a small amount of brown hairs fringing this area) and the scutellum has a few
brown medial hairs. The basal area of the first tergum bears long pale ochraceous basal hairs that
reach the apex of the tergum laterally, but not medially. The apical area is glabrous. The basal
area of the second tergum bears many dense white hairs. The interband zone of the second tergum has
suberect pale hairs mixed with largely spaced more complex pale hairs. The apical area of the
second tergum bears diffused, short, very pale ochraceous to white, appressed hairs that are
somewhat puberulent in vestiture (usually the medial area of the basal portion of this tergum
has these fine hairs worn to expose the integument). The third and fourth terga are similar to
that of the second, however, the basal tomentum is often paler brown, and the interband zones bear
much more puberulent light hairs (these terga often being entirely covered in diffuse pubescence).
The fifth and sixth terga are mostly covered in dark ochraceous hairs apart from the lateral tufts,
which are white.
Similar Species
From what may be apparent above, M. stearnsi does not resemble any other species of Melissodes in Oregon and for this reason, sex-specific comparisons will be omitted. In place of the omitted comparisons, diagnostic characters sufficient for species-level identification for each sex will be listed. However, even though this species is highly distinctive compared to other Melissodes in both sexes mostly by the vestiture (more so females than males), M. stearnsi can resemble a few other bees in different genera (again, more so females than males). This species is readily separated from other genera by the generic Melissodes characteristic of anteriorly narrowed tegulae.
♂ The fourth sternum’s posterior apical area is developed into a thin, broad, colorless hyaline flap that’s the same length of the rest of the sternum medially and is medley weakly emarginate. The second sternum is widely convex and the apical area is hyaline. The third sternum has a hyaline, bilobed, wide flap that is similar to the second sternum, although the flap is shorter. The fifth sternum is the same as the second. The clypeus is usually white to cream-colored and on rare occasions, sometimes pale yellow
♀ The galeae have abundant long hooked hairs above and the second, third, and fourth terga are largely covered in short, pale, diffused, pubescence unless worn. The fifth and sixth terga have long, dark ochraceous to ochraceous medial hairs, and long white lateral hairs.
Fig. ??. A graph representing the phenology of M. (Callimelissodes) stearnsi. The x value is the month, and the y value is the number of documented observations occurring in that month. Data compiled from Data derived from (Ikerd, 2019; Johnson, 2020; Ikerd & Engler, 2023; Bentley & Osborn, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026a; Cheadle Center for Biodiversity and Ecological Restoration, 2026b; Gibbs, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Kelly et al., 2026; Kenneth S. Norris Center for Natural History, 2026; Mertz et al., 2026; Museum of Southwestern Biology, 2026; Orrell & Informatics, 2026a; Orrell & Informatics, 2026b; The International Barcode of Life Consortium, 2026; University of California, Davis, 2026; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC0 1.0, and CC BY-NC 4.0 as applicable (see "Dataset liscences").
Flower preference
Melissodes stearnsi, although sufficient data is lacking, seems as if it may prefer flowers from the family Asteraceae, but not necessarily an oligolectic bee (Laberge, 1961). Although the vast majority of collections outlined by Laberge (1961) are from the plant family Asclepiadaceae (204 out of 319 total), only 8 of these are females. Moreover, the highest number of female floral records for a specific family was Asteraceae, similar to most Melissodes (1961). However, only 71 female M. stearnsi floral records are known (44 on Asteraceae and only 9 on Asclepiadaceae), each of these mostly spread out across many families (10) with no real noticeable preference (Laberge, 1961). More work is needed to definitely determine the flower preference and/or oligolecty towards any one family or genus.
Synonyms
Exomalopsis stearnsi, Cockerell, 1906.
Melissodes subagilis

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes subagilis
Melissodes subagilis, although the name suggests otherwise, is a fairly distinctive Oregonian bee with females bearing somewhat of a resemblance to M. verbesinarum in the coverage and color of the vestiture. However, males aren’t necessary allied with one Oregonian species in particular (range wide, quite similar to M. coreopsis, but this species has no state records) but can resemble a few other Eumelissodes just based on the somewhat largely shared flagellar structure and pale vestiture (see similar species section). Males are distinctive in that they are somewhat small ranging between 9-11 mm in length; the galeae are often dull above due to shagreening, minimally in the apical halves and sometimes more; the apical area of the first tergum doesn’t have hairs obscuring it at lateral areas; the overall vestiture is often somewhat yellow to ochraceous; the second tergum’s distal pale pubescent band is usually narrower than the apical area; the dorsal areas of the scutellum and mesoscutum do not have dark hairs; the punctures on the interband zone of the second tergum are larger and mostly separated by two puncture diameters or less; each puncture is notably wider than the bases of the hairs that arise from them; The mandibular bases and labrum do not have a yellow maculation; the clypeus is entirely light excluding the dark spots denoting the tentorial depressions or the anterior notches and margin; the first flagellar segment’s maximum length is less than, or equal to, one-third of the third segment’s minimum length. Females are distinctive in that the galeae are dull above due to dense reticular shagreening and the hairs on the inner surfaces of the hind basitarsi are often red to yellow, and on rare occasions, dark reddish brown; the mesoscutum usually doesn’t have a dark patch of hairs, but if it does, then it’s smaller than that of the scutellum’s dark patch (rarely a dark patch present in Oregon; Laberge, 1961); the overall pale vestiture is ochraceous to yellow, prominently on the mesoscutum; the interband zone of the second tergum has punctures that are small, irregular, and mostly separated by two puncture diameters, but also bears pale subappressed hairs; the underside of flagellar segments 3-10, and usually the second in the apical one-third are red to yellow; the apical area of the third tergum is obscured due to the distal band which reaches the apex of the tergum, minimally laterally and often more. M. subagilis has a total of 15 state records, placing it as the 13th most common Oregonian Melissodes, most localized in Mid-Eastern Oregon and a few occurring in the South Central region as well (Fig. ??). This species was historically known to be active between the months of June and November, with peak activity occurring in July, August, and September (Laberge, 1961). Newer data present a similar pattern, however, M. subagilis seems to have been collected as early as April and August seems to be the apex of activity (Fig. ??).
Field Markers
♂ Usually a somewhat pale to ochraceous bee, a similar vestiture color range to M. agilis (liklely where the name was derrived).
The eyes are often blue to greenish blue, and sometimes gray to yellowish gray. The clypeus is yellow apart from the darkened
tentorial depressions and the testaceous apical margin and the wing veins are usually dark red to yellow (sometimes, be it
rarely, the wing veins are entirely brown). The underside of the flagella are red to yellow and dark reddish brown to brown
on top (F1 sometimes entirely dark brown) and the terga, which are colorless to faintly yellow hyaline in the apical areas.
The mandibular bases and labrum do not have a yellow maculation. The first flagellar segment’s minimum length is usually
equivalent to one-fifth to one-eighth the second segment’s maximum length, and on rare occasions it can be as short at one-tenth
the maximum length of the second segment. The minimum length of the first flagellar segment (smaller side of F1) is often more
than half the maximum length (longer side of F1). The length of the penultimate flagellar segment is longer than three times its
width. The first flagellar segment’s maximum length is less than, or equal to, one-third of the third segment’s minimum length.
The flagellum reaches the pterostigma when in repose. Makes are often somewhat small ranging between 9-11 mm in length and the
galeae are often dull above due to shagreening, minimally in the apical halves and sometimes more. The apical area of the first
tergum doesn’t have hairs obscuring it at lateral areas. The second tergum’s distal pale pubescent band is usually narrower than
the apical area. The punctures on the interband zone of the second tergum are larger and mostly separated by two puncture diameters
or less with each puncture being notably wider than the bases of the hairs that arise from them. The head hairs are often entirely
ochraceous, but in western individuals, they can be white. The thoracic hairs are mostly pale ochraceous to white laterally,
and ochraceous dorsally, except for western individuals, which have white dorsal hairs, with brown patches never present. The
first tergum bears long ochraceous to white basal hairs, and in apical areas, these hairs become appressed, somewhat simple with
few branches, and exceed the apical margins, but only obscure it in the lateral areas, not obscuring it medially. The distal
pale pubescent band of the second tergum is medially wider than the tergum’s apical area. The apical area of the second tergum
bears comparably simple, long, suberect to subappressed, yellow to white hairs (no brown hairs present). The third, fourth, and
fifth terga are similar to that of the second, except the apical areas posterior to the pubescent bands become shorter with each
subsequent tergum until the band reaches the apex across the apex across the entire tergum on T5. The sixth and seventh terga
are completely covered in white to dark ochraceous hairs.
♀ Definetly one of the paler Melissodes in Oregon, and although its vestiture may range, the most commonly found Oregonian
females bear very pale ochraceous to near white hairs. The eyes are often blue, but sometimes they can be a gray or greenish blue.
The wing veins are usually reddish brown to black, and often dark red and the underside of flagellar segments 3-10, usually the
second segment’s apical one-third as well, are red to yellow (dark brown on top). The apical one-third of the first tergum, which
is usually rufescent. The galeae are dull above due to dense reticular shagreening and the hairs on the mesoscutum usually doesn’t
have a dark patch of hairs, but if it does, then it’s smaller than that of the scutellum’s dark patch. However, a dark dorsal thoracic
patch is quite rare for individuals in Oregon as outlined in Laberge (1961). The interband zone of the second tergum has punctures
that are small, irregular, and mostly separated by two puncture diameters, but also bears pale subappressed hairs. The underside of
flagellar segments 3-10, and usually the second in the apical one-third are red to yellow. The head hairs are mostly white to pale
ochraceous, apart from a few brown hairs on the vertex, but never abundant. The thoracic hairs are dorsally ochraceous and laterally
white with the following additions: the mesoscutum has no brown hairs posteromedially (or less than the scutellum) in northern and
eastern individuals, and a large dark patch often less than twice the size (rarely more) of the scutellar dark patch in southern and
western individuals; the scutellum is often entirely covered in brown hairs, and peripherally fringed with pale ochraceous hairs.
The hairs on the anterior portion of the mesoscutum are subappressed to appressed, somewhat decumbent. The first tergum’s basal area
bears pale ochraceous hairs, but in the apical areas, the tergum is glabrous. The basal pale pubescent band of the second tergum is
white. The interband zone of the second tergum bears short, completely to primarily ochraceous, suberect to subappressed hairs. The
distal pale pubescent band of the second tergum is white to pale ochraceous, often uninterrupted medially, medially wider than that
of the apical area, and the apical hairs of the band are of similar length, or longer than, the hairs of the basal pubescent band,
and simple. The third tergum is similar to that of the second, except the distal pale pubescent band reaches the apical margin minimally
laterally and often more, and the basal tomentum is brown. The fourth tergum’s distal pale pubescent band is pale ochraceous to white,
complete, and reaches the apex across the entire tergum. The fifth tergum bears long wide lateral tufts of pale hairs and the sixth
tergum is medially orange-ish brown to brown with similar lateral tufts of pale hairs. The hairs on the inner hind basitarsal surfaces
are red to yellow in individuals from the east, and dark brownish red to red in individuals from the west, but intergrades may occur
(in general, these hairs are mostly reddish-orange to yellow).
Similar Species
Although the males are fairly distinct in Oregon, the lack of mandibular and labral maculations as well as the fact that the flagellum reaches the pterostigma when in repose are characteristics similar to the M. grindeliae male as well as most of the males in the druriellus group. Females are slightly more distinct than the males due to the tergal vestiture covering most of terga 2-4 with many bands reaching the apical margin across the entire tergum. A characteristic shared by M. verbesinarum (and, to an extent M. stearnsi, but can be separated from this species as outlined in the Melissodes stearnsi treatment).
♂ Males are probably most simalar to those of M. grindeliae in Oregon on account of both species having entirely black labrums and mandibular bases. However, M. subagilis greatly differs from M. grindeliae, and for that matter most of the druriellus group in that the first flagellar segment’s maximum length is less than, or equal to, one-third of the third segment’s minimum length (unlike group 2 of M. grindeliae), the tergal rims are hyaline (unlike M. druriellus) and the length of the penultimate flagellar segment is longer than three times its width (unlike M. pallidisignatus, group one of M. grindeliae, and group one of M. druriellus).
♀ M. subagilis females are undoubtably most superficially similar to those of M. verbesinarum, but can somewhat resemble M. steransi and M. utahensis, though to a much less degree. This species can be separated from M. verbesinarum in that the galeae are dull above due to dense reticular shagreening instead of the shiny unshagreened galeae (apart from the tips) of M. verbesinarum. However, M. subagilis and M. utahensis both have densely shagreened, matte and dull galeae, therefore, other characters are needed to separate the two. M. utahensis differs from that of M. subagilis in that the apical areas of the second and third terga are mostly glabrous and the bands are noticeably separated from the apical margin, the terga have fewer and overall less dense punctures, and the mesoscutum is also overall less crowded with punctures.
Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) subagilis in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Johnson, 2020; Illinois Natural History Survey, 2026). Data liscenced under CC BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Eumelissodes) subagilis in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Ikerd, 2019; Johnson, 2020; Illinois Natural History Survey, 2026) and range-wide data compiled from (Eardley & Ranwashe, 2017; Ikerd, 2019; Johnson, 2020; Carril et al., 2023; Ikerd & Engler, 2023; South Dakota State University, Severin-McDaniel Insect Research Collection, 2023; Texas A&M University Insect Collection, 2023; North Carolina State University Insect Collection, 2024; Droege & Maffei, 2025; European Bioinformatics Institute (EMBL-EBI), GBIF Helpdesk, 2025; Scott, 2025; Sánchez & Comisión, 2025; Zhuang, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Brigham Young University, Arthropod Collection, 2026; Clemson University Arthropod Collection, 2026; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Frost Entomological Museum, 2026; Gibbs, 2026; Grinter et al., 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Orrell & Informatics, 2026a; San Diego Natural History Museum, 2026; The International Barcode of Life Consortium, 2026; University of Arizona Insect Collection, 2026a; University of Arizona Insect Collection, 2026b; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
M. subagilis is an oligolege of the plant family Asteracea and seems to have a preference specifically for the genus Grindelia (Laberge, 1961). Given that fewer publicly available digitized specimen records with floral data are currently present than those in Laberge’s (1961) paper, floral analyses will be derived from his work. Of the 345 M. subagilis collected with floral data attached, 212 were found atop Grindelia, 131 of which were females and 81 of which were males. Two percentages will be given below, one representing the total specimens collected, and one representing females collected as this sex is what determines oligolecty and preference. This renders Grindeliae to be favored 61.44% of the time in totality, and 64.85% of the time by females.
Synonyms
Melissodes agilis var. subagilis, Cockerell, 1905; Melissodes pecosella, Cockerell, 1905; Tetraloniella excurrens melanaspis, Cockerell, 1925; Xenoglossodes albertensis, Cockerell, 1937.
Melissodes tepidus

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes tepidus
Melissodes tepidus is one of two currently known Oregonian Melissodes (M. tepidus and M. communis) that reside in the subgenus Melissodes. Due to the paucity of species in this subgenus that also redside in Oregon, M. tepidus is quite distinctive based on subgeneric characteristics alone. On top of this, M. communis and M. tepidus are one somewhat of the opposite ends of morphology within their subgenus, specifically in regards to their vestiture, making identification even simpler. Males are distinctive in that the labrum is entirely pale with no darkened margin; the second tergum’s wide distal pale band is equal to, or almost equals, the medial portion of the apical area; the fourth tergum’s wide pale band is more than, or equal to, fourth times the medial portion of the apical area. Females are distinctive in that the metanotum is as long as, or longer than, the propodeum’s dorsal surface medially; the first tergum is usually only narrowly hyaline marginally; the second tergum’s distal pale band is often never medially interrupted and the anterior margin of this band is straight while the posterior margin is evenly curved; the second and third terga have very wide pubescent bands that cover a large area of the tergum, prominently wide on the second tergum; the apical areas of the second and third terga are distinctly medially punctate, with punctures that are notably wider than the hairs that arise from them posterior to the bands. However, although these apply to all M. tepidus, two of the three subspecies are present in Oregon, tepidus and timberlakei, each with slight variation from one another in both morphologies and range. The latter of the two tends to be darker in both sexes than the former and also occupies a much larger region of the state. The former has only been found in the south western corner of Oregon, and given that the other subspecies (M. t. timberlakei) is also present in this area, intergrades between the two are known to occur (Laberge, 1961). The total combination of the two subspecies’ ranges combined shows that M. tepidus seems to occur across most of inland Oregon, and although none have currently been found across the coastal area of this state (Fig. ??), more sampling may find a presence in these regions. This, however, is just speculatory. M. tepidus has been historically known to fly between the months of April and October with peak activity occurring during June and July in the timberlakei subspecies and May to August with peak activity occurring during July in the tepidus subspecies (Laberge, 1956a), although both likely have similar phenologies and the larger timeframe will be used. Newer data seem to reflect this pattern (Fig. ??).
Field Markers
♂ Males are fairly pale oracheous to ochraceous-yellow, but most notably, similar to the other Oregonian male in the subgenus
Melissodes (M. communis), the labrum is entirely cream to whtie with no dark margin. The eyes are bluish green to a light pale
blue and the wing veins are black to dark brownish red, often darkening in the apical and anterior areas. The clypeus is cream
to pale yellow and the mandibles bear a large basal yellow macula that is larger than the concave triangular basal area, often
covering the entire mandibular base. The underside of flagellar segments 2-11 are red to yellow (these segments black to brownish
red on top) and the first flagellar segment is dark brownish red and usually bearing a ventral pale red macula. The sterna and
lateral tergal surfaces are often rufescent and the apical areas of terga two, three, four, and five are usually somewhat
translucent (often not transparent, usually having some sort of coloration), but in older worn individuals, these translucent
areas may become opaque or piceous. The first flagellar segment’s minimum length ranges from being faintly less than one-sixth
of the second flagellar segment’s maximum length, to faintly more than one-third (most commonly measuring one-fifth; see
subspecies descriptions). The maxillary palpal segments decrease in length from basal to apical in a ratio of about 3.0:3.0:2.5:1.0,
with the length of the ultimate segment sometimes being greater. Given the presence of two subspecies, a base-line male setal
description will be given, along with subspecific descriptions that are meant to be applied to, and override portions of, this
generic description (including the foregoing structural description).
The head hairs are mostly white apart from the vertex, which is usually near-white to white with no dark hairs on the vertex.
The thoracic hairs are near-white to white except as follows: the scutellar hairs are mostly near-white to white with a fairly
small dark scutellar patch medially. The first tergum’s basal two-thirds (or less) bear long near-white to white hairs that reach
the apical margin laterally, and the apical area of this tergum is more or less bare, but a few dark brown, appressed, simple hairs
are present at extreme lateral areas. The basal pubescent band of the second tergum connects to the distal band at extreme lateral
areas and minimally partially consists of basal plumose hairs and narrowly apical spatulate hairs. The interband zone of the second
tergum consists of appressed to suberect, erect black to dark brown hairs and the distal pale ochraceous band is wide (much wider
than that of M. communis). The apical area of the second tergum consists of appressed to suberect, erect black to dark brown hairs
(similar to the interband zone). The third tergum’s distal near-white to white band often reaches the basal area of the tergum somewhat
obscuring the interband zone. The apical area of the third tergum is similar to that of the second tergum. The fourth tergum is similar
to that of the second tergum, but the pubescent band is very wide (often covers most of the tergum) and sometimes medially interrupted
by a small rectangular or triangular notch of suberect dark brown hairs. The sixth and seventh terga are both shrouded in appressed,
black to dark brown hairs, and the sixth tergum (sometimes the seventh as well) usually has some long pale hairs laterally.
M. tepidus ssp. tepidus
The first flagellar segment’s minimum length usually ranges between being one-fourth to one-fifth of the second segment’s maximum length, but sometimes, on rare occasions, it can measure to be either one-third or one-sixth of the second segment’s maximum length. The head hairs are entirely pale ochraceous to white with no dark present on the vertex. The thoracic hairs are also almost entirely pale ochraceous to white, usually with no to very few dark hairs present on the mesoscutum (if present, only on the posteromedial area), but the scutellum has a small dark patch of hairs peripherally lined with pale hairs. The light hairs on all of the terga tend to take on a pale ochraceous to white color and the interband zone of the second tergum is narrow and bears yellow to white (at most brown in the medial one-third of the tergum), erect to suberect hairs. The hairs in the apical area of the third tergum are suberect and minimally pale on the lateral one-fourth of the tergum (if this is the case, then brown suberect hairs are present in the medial one-half or less), but usually there are pale hairs across the entire apical area. The fourth tergum is similar to that of the third. The fifth tergum often has no apicomedial dark hairs, or if present, than a minute amount. The sixth and seventh terga are mostly covered in yellowish or near-yellow hairs, but medially, a few brown hairs are often present (only on rare occasions are the terga shrouded entirely in yellow). The sternal hairs range from yellow to white, but on the ultimate and penultimate sterna, these often become brownish yellow. The leg hairs are mostly white, differing at the basitibial plates, which are usually brown; the inner tarsal surfaces, which are orange to yellow; and the hind tibiae, which are also orange to yellow.
M. tepidus ssp. timberlakei
The first flagellar segment’s minimum length often ranges between one-fourth to one-fifth of the second segment’s maximum length, rarely ever measuring one-sixth but sometimes as long as one-third as well. The head hairs are almost entirely ochraceous to faintly ferruginous (sometimes pale ochraceous to white, most often in older individuals or specimens) often bearing a few brown hairs on the vertex. The thoracic hairs are also almost entirely ochraceous to faintly ferruginous (sometimes pale ochraceous to white, most often in older individuals or specimens) except as follows: usually a few brown hairs are present on the mesoscutum posteromedial mesoscutal area creating a dark setal patch of similar size, or larger than, the scutellar dark patch; the scutellum which has a small dark patch of hairs, peripherally lined with pale hairs. The light hairs on all of the terga tend to take on a pale ochraceous to white color and the interband zone of the second tergum is narrow and bears yellow to white (at most brown in the medial one-third of the tergum), erect to suberect hairs. The hairs in the apical area of the third tergum are suberect and at most, pale on the lateral one-fourth of the tergum with the rest of the apical area covered in dark brown suberect hairs (very rarely are dark hairs absent). The fourth tergum is similar to that of the third. The fifth tergum often has a few apicomedial dark hairs. The sixth and seventh terga are mostly covered in yellowish or near-yellow hairs, but medially, brown hairs are present. The sternal hairs range from yellow to white, but on the ultimate and penultimate sterna, these often become brownish yellow. The leg hairs are mostly white, differing at the basitibial plates, which are usually brown; the inner tarsal surfaces, which are orange to yellow; and the hind tibiae, which are also orange to yellow.
♀ Often a very pale bee with distinctively wide tergal bands that shroud most of the surface on terga 2-4 (in this manner,
faintly reminiscent of M. subagilis and M. verbesinarum females). The eyes are bluish green to a light pale blue and the wing
veins are black to dark brownish red, often darkening in the apical and anterior areas. The underside of flagellar segments 3-10
are red to yellow and the sterna and lateral tergal surfaces are rufescent. The apical margin of the first tergum is very narrowly
hyaline and the metanotum is as long as, or longer than, the propodeum’s dorsal surface medially. The second and third terga have
very wide pubescent bands that diffuse over most of the tergum (much wider than those of M. communis). The apical areas of the
second and third terga are distinctly medially punctate, with punctures posteriorly to the bands that are notably wider than the
hairs that arise from them. In a similar fashion to that of the male, a base-line setal description will be given below, to which
subspecific descriptions can be applied, and in areas, override. Unlike the males however, these subspecific characteristics rely
solely on setal coloration with no structural variations (at least none of significance) among subspecies.
The head hairs are mostly white apart from the vertex, which is usually pale ochraceous, but often having a small amount to
several dark brown to black hairs. The thoracic hairs are white to pale ochraceous laterally and dorsally white to ochraceous
(sometimes faintly ferruginous) except as follows: the mesoscutum has a large posteromedial dark patch (somewhat square-shaped)
that often reaches the anterior tegular margins, sometimes extending beyond but usually falling short; the scutellar hairs are
mostly dark brown, but peripherally, this dark brown patch is margined with pale ochraceous hairs. The first tergum’s basal two-thirds
(or less) bear long pale ochraceous hairs that reach the apical margin laterally, and the apical area of this tergum is more or less
bare, but a few dark brown, appressed, simple hairs are present at extreme lateral areas. The basal pubescent band of the second
tergum connects to the distal band at extreme lateral areas and minimally partially consists of basal plumose hairs and narrowly
apical spatulate hairs. The interband zone of the second tergum consists of appressed to suberect, erect black to dark brown hairs
and the distal pale ochraceous band is wide (much wider than that of M. communis). The apical area of the second tergum consists of
appressed to suberect, erect black to dark brown hairs (similar to the interband zone). The third tergum’s distal pale band often
reaches the basal area of the tergum somewhat obscuring the interband zone. The apical area of the third tergum is similar to that
of the second tergum. The fourth tergum is similar to that of the second tergum, but the pubescent band is very wide (often covers
most of the tergum), positioned apically, and sometimes medially interrupted by a small rectangular or triangular notch of suberect
dark brown hairs. The fifth and the sixth terga are both shrouded in appressed, black to dark brown hairs, and the fifth tergum
(sometimes the sixth as well) usually has some long pale hairs laterally.
M. tepidus ssp. tepidus
The head hairs are mostly pale ochraceous to white, apart from the vertex, which sometimes has a minute amount of long dark hairs (approximately 20), although the head hairs are often entirely light. On the head surfaces where hairs are present between the compound eye apices and the lateral ocelli, these hairs are often white. The thoracic hairs are mostly pale ochraceous to white as well (sometimes, be it rarely, rufescent anterior to the dark mesoscutal patch), except as follows: the mesoscutum, which has a dark posteromedial patch of hairs that sometimes reaches, but often just falls short of extending to the anterior regular margins; the mesoscutal hairs anterior to the dark patch, which are often pale but have dark hairs from the dark patch mixed, creating somewhat of an indistinct transition; and the scutellar hairs, which are mostly dark, but peripherally fringed with pale hairs. The distal pale band of the second tergum is usually two times the length of the medial portion of the apical area and sometimes, be it rarely, narrower than one and one-half of the medial portion of the apical area. The interband zone of the second tergum bears suberect yellow to white hairs (at most, becoming brown in the medial one-third) and is narrower than the width of the distal pale band. The apical area of the third tergum is often narrower than one-third of the distal pale band medially, and bears suberect, short, often white hairs that are minimally present one either side of the lateral one-fourths of the first tergum, but usually occur across the entire tergum. The areas near the apex of the fourth tergum often lack dark brown hairs medially. The sternal hairs are often brownish yellow, becoming white laterally and fringed with white apically on all but the ultimate sterna, which bears entirely brownish yellow hairs. The leg hairs are mostly pale ochraceous to white, differing at the apical areas of the outer surfaces of the middle tibiae, which are brown; the outer surfaces of the fore basitarsi, which are brown; the dititarsi, which are rufescent to ochraceous; the basitibial plates, which are brown; and the inner surfaces of the hind basitarsi, which are red to yellow.
M. tepidus ssp. timberlakei
The head hairs are mostly pale ochracous, apart from the vertex, which is covered in ochraceous hairs that may become white in worn and older individuals (mostly in specimens) and a few to many long dark brown hairs (minute amounts may be present, but these hairs are never absent). The areas of the vertex between the apicices of the compound eyes and lateral ocelli are often covered in short brown hairs that tend to reach down into the facial area medially. The thoracic hairs are pale ochraceous to white (often becoming paler in the lower lateral areas) except as follows: the mesoscutum, which has a large posteromedial dark patch of hairs that becomes wider anteriorly and often reaches, or surpasses, the anterior regular margins; the pale hairs on the mesoscutum, which are usually pale ochraceous to white, but can sometimes be rufescent; and the scutellum, which is mostly covered in dark brown hairs and posteriorly margined by pale hairs. The interband zone of the second tergum often bears dark brown suberect hairs in the medial one-third of the tergum (sometimes, although rarely, less), these dark hairs becoming absent laterally (dark hairs are almost always present). The medial portion of the distal pale band of the second tergum is often narrower than two times the width of the apical area due to wear, but often, in width, approximately one and one-half times the width of the apical area (sometimes narrower). The medial portion of the distal pale band of the third tergum is often slightly less than, or equal to, three times the width of the apical area. The apical area of the third tergum often has large black to dark brown suberect area of hairs medially, these hairs becoming absent in extreme lateral areas, with the lateral one-fourth of the tergum only rarely bearing pale hairs. The four tergum often minimally has a noticeable, small amount of dark brown hairs apicomedially. The sternal hairs are often omstly dark brown, becoming white to pale ochraceous laterally and apically fringing the terga, apart from the ultimate sterna, which bears completely dark brown hairs. The leg hairs are mostly pale ochraceous to white, differing at the apical areas of the outer surfaces of the middle tibiae, which are brown (a faintly larger brown patch than M. t. tepidus); the outer surfaces of the fore basitarsi, which are brown (also a faintly larger brown patch than M. t. tepidus); the dititarsi, which are rufescent to ochraceous; the basitibial plates, which are brown; the hairs on the fore tarsi, which are dark brown (darker than those of M. t. tepidus); and the inner surfaces of the hind basitarsi, which are red to yellow.
Subspecies and variation
As apparent from the subspecific descriptions of both sexes, M. tepidus is fairly variable in multiple characteristics. For this reason, this subsection of M. tepidus’ treatment will be dedicated to identification of the subspecies and variation tables. The lighter of the two Oregonian subspecies, M. t. tepidus, currently occupies only a small portion of Oregon, present in the eastern one-fourth of the state. However, the darker subspecies, M. t. timberlakei, seems to occur across the majority of Oregon, with the only areas in which no collections have taken place and its range likely doesn’t overlap being the north western corner.
♂ The males of M. t. tepidus can be separated from those of M. t. timberlakei by the three following
traits, although a combination of two are sufficient for identification: the suberect hairs on the apical area of the fourth
tergum are minimally pale in the lateral one-fourth of the tergum, and often pale across the entire apical area; the mesoscutal
hairs lack a distinctive dark patch; and the interband zone hairs of all the terga are at most darkened in the medial one-third
of the tergum, but often entirely pale. For comparison, male of M. timberlakei have the apical hairs of the fourth tergum pale
at most in the lateral one-fourth of the tergum, but often the entire apical area is covered in dark hairs; usually a few brown
hairs are present on the mesoscutum posteromedial mesoscutal area creating a dark setal patch of similar size, or larger than,
the scutellar dark patch; the scutellum which has a small dark patch of hairs, peripherally lined with pale hairs; and the
interband zone hairs are also mostly pale, but sometimes, they become brown in the medial one-third (similar to M. tepidus,
though given that this subspecies is darker, brown hairs are likely more common).
♀ Females of M. t. tepidus can be separated from those of M. t. timberlakei by the lack of dark medial hairs on the fourth tergum.
Similar to the male however, there are three more characters that can separate M. t. tepidus from M. t. timberlakei, two of which
are sufficient for identification: the vertex ranges from either being entirely covered in pale hairs to having approximately 20
dark hairs (the short hairs are mostly to completely pale); the suberect, short hairs on the apical area of the third tergum are
minimally pale on both sides of the tergum’s lateral one-fourth to all these hairs on the interband zone being pale; and the suberect
hairs on the interband zone of the second tergum are at most darkened in the tergum’s medial one-third and often entirely pale. For
comparison, M. t. timberlakei females have dark medial hairs on the apical area of the fourth tergum; the vertex is usually covered
in ochraceous hairs (darker than the surrounding pale hairs) and a few to many long dark brown hairs (minute amounts may be present,
but these hairs are never absent); the apical area of the third tergum often bears a large dark patch of black to dark brown hairs
medially that become absent in extreme lateral areas, but the lateral one-fourth of the tergum only rarely bears pale hairs; and the
interband zone of the second tergum often bears dark brown suberect hairs in the medial one-third of the tergum (rarely less) that
start to become absent laterally, but are almost always present medially.
Due to the ranges of these two subspecies overlapping, the Eastern areas of Oregon are home to many intergrades that likely differ from the subspecific descriptions and the base-line description. Therefore, a singular description (or 2 for each Oregonian subspecies for that matter) cannot sufficiently capture all variation. To address this, Laberge (1956a) created a list of 7 female characteristics and 5 male characteristics that consistently differ and delimit subspecies (subspecific descriptions are partially based on these as well) with 2-4 variations per character. A similar structure derived from Laberge’s (1956a) findings will be presented here in the form of a table along with the character values in regards to their melanism, including characteristics that aren’t directly implicative of melanistic value (i.e. the ratio of the male’s first flagellar segment to their second). Characters as such are given their melanistic value based on which subspecies they are most commonly observed in and that specific subspecies melanistic value. The characters in the tables below, similar to the subspecific descriptions, are meant to be applied to, and override portions of, the base-line M. tepidus description given above. Many characters have binary choices and others have multiple. In such scenarios (e.g. the first character of the female), they will be set into the respective characters' extremes (dark or light). Characters that were originally given 4 variations in that of Laberge (1956a) have been reduced to three for simplicity. Note that each value per character (light, intermediate, or dark) are independent from one another (although, many are correlated), therefore a female M. tepidus may have no dark hairs on the vertex (palest choice for the second female character) and also have dark hairs on the inner surfaces of the hind basitarsi (darkest choice for the seventh female character). Although variation between all three subspecies is unnecessary given only two exist in Oregon, this table will encompass all possible variants, even intergrades between subspecies that are currently unknown in this region.
Table 5. 5 characters pertaining to the vestiture and antennal structure of males of M. tepidus each with three different melanistic variations. This table accounts for all possible morphs of the males of M. tepidus when the characters are either added to, or replace, portions of the base-line pale description.
| Character | Light | Intermediate | Dark |
|---|---|---|---|
| The first flagellar segment’s minimum length is less than one-sixth of the second segment’s maximum length. | The first flagellar segment’s minimum length is one-fifth to one-sixth of the second segment’s maximum length. | The first flagellar segment’s minimum length is one-fourth or more of the second segment’s maximum length. | |
| The mesoscutal dark patch | The entire mesoscutum is covered in pale hairs, dark patch absent. . | The patch is smaller in size than the area of the scutellum. | The patch is of similar size to, or greater size than the scutellum. |
| The interband zone hairs of the second tergum | The hairs are entirely pale, brown hairs absent. | The hairs are at most brown in the medial one-third, but often less (brown hairs still present). | The hairs are minimally brown in more than the medial one-third to entirely brown. |
| The hairs on the apical area of the third tergum | The hairs are entirely pale, brown hairs absent. | The hairs are at most brown in the medial one-half, but often less (brown hairs still present). | Minimally brown in the medial one-half and often all hairs are brown. |
| The apical area of the fourth tergum | The apical hairs are pale (no dark hairs present). | Dark hairs are present minimally in the medial areas, sometimes more. |
Table 6. 7 characters pertaining to the vestiture of the females of M. tepidus each with three different melanistic variations. This table accounts for all possible morphs of the females of M. tepidus when the characters are either added to, or replace, portions of the base-line pale description.
| Character | Light | Intermediate | Dark |
|---|---|---|---|
| The mesoscutal patch of dark hairs | The dark mesoscutal patch of hairs fall short of reaching the anterior tegular margins. | The dark mesoscutal patch of hairs extends to, or surpasses the anterior tegular margins. | |
| The vertex hairs | The vertex hairs are entirely pale. . | A small amount of dark hairs are present on the vertex. | Many dark hairs are present on the vertex. |
| The interband zone hairs of the second tergum | The hairs on the second tergum’s interband zone are entirely pale. | The hairs in the lateral one-third, or slightly less, of the second tergum’s interband zone are pale. | The majority of, to all of the second tergum’s interband zone hairs are black to dark brown. |
| The hairs on the apical area of the third tergum | The majority of, to all of the hairs are pale. | The hairs range from being dark in the medial one-third to medial one fourth (pale hairs laterally). | The hairs are minimally dark in more than the medial one-third to entirely dark brown across the entire tergum. |
| The distal band of the second tergum | The distal pale bane is medially equal to, or wider than, one and one-half times the medial width of the apical area, but narrower than two times the apical areas width. | The distal pale band is medially narrower than one and one-half times the medial width of the apical area. | |
| The distal pale band of the fourth tergum | The apicomedial brown patch is absent. | A patch of brown apicomedial hairs is present. | |
| The hairs on the inner surfaces of the hind basitarsi | The hairs range from red to reddish orange to yellow, but not highly contrasting the the rest of the scopae. | The hairs are brownish red. | The hairs are dark brown to black |
Similar Species
In comparison to all other Melissodes in Oregon, M. tepidus (both subspecies as well) are quite distinctive. With this species falling into the subgenus Melissodes, the only resemblance it truly bears is to M. communis in both sexes, although, the vestiture of females in particular can sometimes resemble M. subagilis, M. verbesinarum, and to an extent, M. stearnsi (although, mostly the former two). Male’s on the other hand have no real resemblance to one specific species, although they are most closely related to M. communis.
♂ Males of M. tepidus and M. communis both share a pale cream to white colored labrum with no darkening marginally, but this is where most of the similarities stop. M. tepidus has a very wide band across the second tergum that’s equal to, or more than, three-fourths of the medial portion of the apical area. For contrast, the second tergum’s distal pale band of M. communis is usually narrow and equal toor just slightly more than, one-third of the width of the medial portion of the apical area. Also, the apical areas of the second and third (often most noticeably so on the second) terga of M. tepidus are distinctly punctate with punctures that are approximately three times the the width of the hairs arising from them, whereas in M. communis, the apical areas are mostly impunctate, or if a minute amount present, then the punctures are fairly indistinct and less than two times the width of the hairs arising from them. Another characteristic to separate these species is the distal pale band on the fourth tergum, that being in M. tepidus, this band is medially equal to, or more than, four times the width of the apical area, whereas in M. communis, this band is noticeably narrower than fourth times the width of the apical area medially. Given the fact that M. tepidus has a pale cream to white labrum with no margin and can readily be separated from M. communis, males of this species are fairly distinctive in Oregon.
♀ Females of M. tepidus are similar to M. communis in the subgeneric female characteristic of the metanotum being as long as, or longer than, the propodeum’s dorsal surface medially and the presence of only a narrow hyaline margin on the first tergum. However, the two distinctly differ in the tergal vestiture. The second and third terga of M. tepidus have very wide pubescent bands that cover most of the tergum (the second tergum’s band is noticeably wide). Whereas in M. communis, the second and third terga have narrower white bands, the second tergum’s band resembling more of a thin line than a wide band. Also The apical areas of the second and third terga of M. tepidus are distinctly medially punctate, with punctures that are notably wider than the hairs that arise from them, posteriorly of the bands. However, in M. communis the apical areas of the second and third terga are not distinctly medially punctate posteriorly of the bands, or if there are punctures, then they are no wider than the base of the hairs that arise from them. With the presence of these wide bands, M. tepidus can faintly resemble M. subagilis, M. verbesinarum, and to an extent, M. stearnsi. M. tepidus can be separated from all foregoing species in the subgeneric Melissodes ss. characteristic of the metanotum being as long as, or longer than, the propodeum’s dorsal surface medially, and can specifically separated from the former two in the fact that the distal pale band on the third tergum doesn’t reach the apical margin across most of, or all of, the tergum.
Fig. ??. Map showing an estimation for the known distribution of M. (Melissodes) tepidus in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026). Data liscenced under CC BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Melissodes) tepidus< in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (Ikerd, 2019; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026) and range-wide data compiled from (Ikerd, 2019; Johnson, 2020; Droege & Maffei, 2025; Scott, 2025; Sánchez & Comisión, 2025; Bentley & Osborn, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026a; Florida State Collection of Arthropods, 2026; Gibbs, 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Johnson, 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Orrell & Informatics, 2026a; San Diego Natural History Museum, 2026; The International Barcode of Life Consortium, 2026; University of Arizona Insect Collection, 2026a; University of California, Davis, 2026; University of Minnesota Insect Collection, 2026). Data licensed under CC BY 4.0, CC0 1.0, and CC BY-NC 4.0 as applicable (see "Dataset Licenses").
Flower preference
Similar to most other Melissodes in the subgenus Melissodes ss., M. tepidus seems to be polylectic, having been found atop 19 families with Leguminosae having the highest toll (Laberge, 1956a). However, as mentioned in his paper, the Leguminosae toll seems to be fairly unrepresentive of a true floral preference as these records are mostly the result of intensive sampleings of bees in alfalfa fields (a plant in Leguminosae) for studies of alfalfa pollinators (Laberge, 1956a). A non-biased sampling effort will have to occur to gain the fullest understanding of the floral preference of M. tepidus.
Synonyms
Melissodes tepida, Cresson, 1878.
Melissodes tristis

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes tristis
Melissodes tristis, the dark faced long-horn bee, is likely one of the easiest identifiable and most recognisable Oregonian Melissodes, especially in the male sex. The vernacular name derived from the male’s diagnostic character of an entirely black clypeus, and although a few other Melissodes males have this pattern, no others are known to occur, or likely occur, in Oregon. Both sexes of these species are very bright white bees with little to no dark tergal hairs (apart from the distal most two terga), and females can especially look very light due to the broadly hyaline margin of the first tergum creating somewhat of a whitish color. Males are distinctive in that the length of the penultimate flagellar segment is noticeably greater than three times its width and when in repose, the flagella reach, or almost reach, the apical area of the marginal cell; the first flagellar segment’s maximum length is less than, or equal to, one-third of the third segment’s minimum length; the clypeus is completely black and usually has a shiny boss medially; the vestiture of the body is usually mostly white; the flagellae bear no depressions, and the underside of flagellar segments 2-11 are red with the last one or two flagellar segments being darker than the rest. Females are distinctive in that the apical area of the first tergum is widely colorless hyaline (approximately the apical one-eighth) and the hairs on the inner surfaces of the hind basitarsi are black to dark brown; the overall vestiture is white to ochraceous and the clypeus has a median shiny boss. Given how distinctive both sexes of this species are, very few characteristics can be used to determine them. Although previously believed to be an oligolect of Asteraceae, as in most other Melissodes (Laberge, 1961), it seems as though M. tristis may in fact be highly polylectic (Wright, 2018; see flower preference). Given its singular Oregonian occurrence in Warm Springs (Ikerd, 2019; likely a valid datapoint due to its highly diagnostic nature), a state-wide distribution map and state phenology line will be omitted, but a distribution wide phenology graph will be presented below (Fig. ??). Historically, M. tristis was known to be active between the months of March and November with peak activity occurring in early July (Laberge, 1961). Newer data seem to support a similar pattern, but peak activity seems to occur throughout June as well (Fig. ??).
Field Markers
♂ Males are very pale, most of their vestiture consisting of near-white to white setae. The eyes are grayish
blue to greenish and the wing veins are brownish red to dark red. The underside of flagellar segments 2-11 are
yellow to yellowish red (these segments black on top) and the first flagellar segment is usually entirely brownish
red. However, the underside of the distal most one to two flagellar segments are often darker than the preceding
ones. None of the flagellar segments have dorsolateral, longitudinal, shiny depressions. The apical areas of the
terga are often colorless (sometimes, although rarely, yellowed) hyaline, transitioning to a thin margin of
rufescence anteriorly before becoming opaque on the rest of the terga. The clypeus, labrum, and mandibles are
entirely dark (highly distinctive). The clypeus is uniquely shaped from that of other Melissodes, it’s rather
flat, short, somewhat wide (although noticeably narrower than that of the female), distinctly converging near
the mandibles, and the oculoclypeal distance falls between one-half to three-fourths of the first flagellar
segment’s smallest diameter, though usually more than the former and less than the latter. The clypeal surface
has a somewhat distinct (less prominent than that of the female) shiny apical boss positioned medially. The
first flagellar segment’s minimum length is often minimally two-thirds of the second segment’s maximum length,
although the first segment often measures about one-tenth of the maximum length of the second segment. Also,
the first flagellar segment’s maximum length is less than, or equal to, one-third of the third segment’s minimum
length. The length of the penultimate flagellar segment is noticeably greater than three times its width. When
in repose, the flagella reach, or almost reach, the apical area of the marginal cell. The head hairs are mostly
white, apart from the vertex of the head, which bears very pale ochraceous hairs (no brown hairs present). The
thoracic hairs are also white, apart from the dorsum, which bears very pale ochraceous hairs (no brown hairs
present). All terga are mostly shrouded in white hairs with no brown present, apart from the basal regions of
terga three, four, five, and six, which only sometimes bear a few brown hairs. The basal area of the first tergum
bears long white hairs that become subappressed near the apical area of the basal portion, these hairs often
extending to, or surpassing the, apex of the terga. The apical area of the second tergum bears distinct, white,
appressed, apical plumes of pubescence that are present on no more than the lateral one-fourth of each side.
The interband zone of the second tergum bears suberect, long, wire-like hairs. The distal white pubescent band
of the second tergum consists of multiple layers, creating somewhat of a “thick” appearance, and medially measures
to a similar length, or longer than, the medial portion of the apical area. The apical area of the second tergum
is either glabrous, or bears simple, white, subappressed hairs. The third and fourth terga are similar to that of
the second.
♀ Females are one of the palest Oregonian Melissodes, bearing almost entirely white tergal hairs on T2-T4;
their paleness accentuated by the apical area of the first tergum. The eyes are grayish green to bluish; and
the wing veins are brownish red to black. The underside of flagellar segments 3-10 and usually the apical
areas of the second are red and the apical one-eighth of the first tergum is colorless hyaline, transitioning
to a faintly rufescent hyaline basally before becoming opaque. The apical areas of terga two, three, and four
are faintly rufescent. The clypeus is uniquely shaped from that of other Melissodes, being rather flat, short,
broad, and bearing a distinct medial boss. The head hairs are mostly white, apart from the vertex, which often
has a few sparse brown hairs. The thoracic hairs are laterally white, and mostly pale ochraceous dorsally except
as follows: the mesoscutum has a medium-sized to small posteromedial brown patch that is of similar size, to
slightly larger than, the scutellar dark patch; and the scutellum is mostly shrouded in brown hairs medially.
The basal hairs of the first tergum are entirely white. The basal band of the second tergum is white and broadly
connects to the distal pale band laterally. The interband zone of the second tergum bears sparse, suberect,
short, brown hairs. The distal pale band of the second tergum is separated from the apical margin, often composed
of multiple layers of hairs, creating a “thick” appearance and rarely, if ever, medially notched. The apical
area of the second tergum ranges from bearing sparse, brown, short, simple, subappressed, hairs to being glabrous.
The third tergum is similar to that of the second, differing at the basal tomentum, which is brown; and the apical
area, which often bears brown hairs, and when present, of higher abundance. The distal pale band of the fourth
tergum is complete and reaches the apical margin across the entire tergum. The fifth and sixth terga are entirely
covered in dark brown hairs with no pale lateral tufts present. The sternal hairs are entirely dark brown, staying
this color both apically and laterally. The leg hairs are mostly white, but become distinctly dark reddish brown
on the inner surfaces of the hind basitarsi.
Similar Species
Due to the dark clypeus of the males and the broad hyaline apical margin of the females, this species doesn’t specifically resemble any other species in particular. The only faint resemblance females bear are to those of M. saponellus and M. vernalis given the shared hyaline apical margins of the first tergum. However, M. saponellus and M. vernalis also have wide hyaline apical margins on terga two, three, and four as well, whereas M. tristis has these areas opaque to faintly rufescent (noticeably not hyaline). As such, further comparisons seem unnecessary.
Fig. ??. A graph representing the phenology of M. (Eumelissodes) tristis. The x value is the month, and the y value is the number of documented observations occurring in that month. Data compiled from (Eardley & Ranwashe, 2017; Ikerd, 2019; Johnson, 2020; Carril et al., 2023; Texas A&M University Insect Collection, 2023; Droege & Maffei, 2025; European Bioinformatics Institute (EMBL-EBI), GBIF Helpdesk, 2025; Luis & Comisión, 2025; Navarro & Comisión, 2025; Quiroz & Comisión, 2025; Romero & Comisión, 2025; Scott, 2025; Sánchez & Comisión, 2025; Vergara & Comisión, 2025; Zhuang, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Brigham Young University, Arthropod Collection, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026a; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Fägerström, 2026; Gibbs, 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Natural Science Research Laboratory, Museum of Texas Tech University (TTU), 2026; New Mexico State Collection of Arthropods, 2026; Northern Arizona University, 2026a; Northern Arizona University, 2026b; Oram, 2026; Orrell & Informatics, 2026a; San Diego Natural History Museum, 2026; The International Barcode of Life Consortium, 2026; University of Arizona Insect Collection, 2026a; University of Arizona Insect Collection, 2026b; University of Minnesota Insect Collection, 2026; Wild, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset liscences").
Flower preference
M. tristis, unlike many other Eumelissodes, seems to be highly polylectic rather than an Asteracea oligolege (Laberge, 1961; Wright, 2018). This species has been collected atop 13 different families and 23 differing genera, similar patterns to the dietary preferences of those in the subgenus Melissodes ss. (Wright, 2018). Although many of these collections are from newer publications (i.e. Wright, 2018), from the data available at the time, Laberge (1961) reasoned that M. tristis likely has 3 generations each year in Texas, New Mexico, and Arizona; likely having more southward and less northward. This was based on phenological activity peaks, in which M. tristis was recorded to have high peaks during April and the first half of May, then in mid July (highest abundance), and finally in the first half of September (Laberge, 1961). However, newer data instead seem to show a similar phenological activity pattern to that of most other Melissodes species, with the peak activity occurring in June and July and without noticeable early or later peaks suggestive of multiple generations per year. Furthermore, M. agilis has been documented to likely only have one generation per year (Parker et al., 1981), and although separate species will have different reproductive ecologies, it seems unlikely that M. tristis would differ at this substantial amount.
Synonyms
Melissodes pallidicinta, Cockerell, 1896; Melissodes tristis var. malvina, Cockerell 1902; Melissodes semitristis, Cockerell, 1902; Melissodes pallidicinta var. erythrina, Cockerell, 1925.
Melissodes utahensis

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes utahensis
Melissodes utahensis seems to be most similar to M. subagilis, but males are very similar to those of M. lutulentus and M. vernalis as well. Although not the most distinctive species in Oregon, the following male characteristics separate a group of four Melissodes from the rest, including M. utahensis: The first flagellar segment’s maximum length is less than, or equal to, one-third of the third segment’s minimum length; the clypeus is entirely light excluding the dark spots denoting the tentorial depressions or the anterior notches and margin; the mandibular bases do not have a yellow maculation and the labrum does or does not have a mediobasal pale macula (rarely do both the labrum and mandibular bases have pale macula, but if so, then the macula on the labrum is less than one-third of the area of the labrum and/or the macula on the bases of the mandibles are tiny, but rarely are there small mandibular macula present and no labral macula); the wing veins are dark rather than pale reddish brown. Only four males in Oregon share the combination of these characteristics, M. utahensis, M. vernalis, M. lutulenus, and somtimes M. subagilis. Although M. bicoloratus males share many of the same characteristics, their wing veins are distinctly pale reddish-yellow. Females are distinctive in that the apical area of the third tergum is often obscured by the distal pale band, which minimally reaches the apex laterally (if not reaching medially, then often having an apubescent triangular notch); the underside of flagellar segments 3-10, and usually the second in the apical one-third, are red to yellow (dark brown on top); the pale hairs are white; the interband zone of the second tergum has white, appressed to subappressed pubescence with no darker spine-like hairs and bears punctures; the galeae are dull above due to shagreening, minimally in the apical one-third to one-half and often more; the hairs on the mesoscutum are usually darkened posteromedially. In general, this species is very light, both sexes often having no dark thoracic hairs (dorsum of thorax just slightly darkening from white in the lateral areas to pale ochraceous). This species has been documented in Oregon three times according to The International Barcode of Life Consortium (2026), these specimens determined via their DNA barcodes. It’s unknown whether the collected specimens’ barcodes were identified as M. utahensis by an algorithm (i.e. BOLD or BLAST) in tandem with morphological traits, or if they were determined purely by algorithmic matches. However, when omitting these datapoints, M. utahensis’ range still has a high likelihood of overlapping Oregon, being found in Washington (McElrath, 2023), Nevada (Ikerd, 2019), Idaho (Ikerd, 2019), and northern California (Bentley & Osborn). Given that three occurrences, each from largely different portions of Oregon, have been recorded and the known range of this species overlaps most of Oregon, M. utahensis will herein be included an Oregonian resident (Fig. ??; see note in “Species treatments” for examples of the contrary). Historically, this species has been recorded to fly from July to September, although a month of peak activity had yet to be discerned (Laberge, 1961). Newer data suggest a similar pattern, but M. utahensis has been recorded in October, and peak activity seems to occur in August (Fig. ??).
Field Markers
♂ Males are very pale with little to no darkening on the vertex and bears finer punctures than that of M.
lutulentus as described below. The eyes are often blue to greenish blue, but sometimes gray to yellowish
gray and the clypeus is yellow except for the darkened tentorial depressions and the testaceous apical margin.
The labrum sometimes bears a small cream mediobasal macula, but most notably so in Arizonan individuals (unknown
in Oregon) and the mandibles lack basal macula. The wing veins are dark brown to brownish red and the underside
of the flagellum is red to yellow (F1 often red underneath as well or entirely brown). The terga are colorless
to faintly yellow hyaline in the apical areas. The galeae are dull above due to dense reticular shagreening and
the pygidial plate is wide, with its width about two-thirds of its length. The punctures on the interband zone
of the second tergum are extremely tiny and mostly separated by more than, or equal to, three puncture diameters;
rarely any wider than the bases of the hairs that arise from them. The interband zone punctures of the third and
fourth terga are also very tiny to small, indistinct, shallow, and mostly separated by two puncture diameters
(usually separated by less on the fourth tergum). The first flagellar segment’s maximum length is less than, or
equal to, one-third of the third segment’s minimum length, and the first flagellar segment’s minimum length is
usually equivalent to one-fifth to one-eighth the second segment’s maximum length (on rare occasions, it can be
as short at one-tenth the maximum length of the second segment). The minimum length of the first flagellar segment
(smaller side of F1) is often more than half the maximum length (longer side of F1). The length of the penultimate
flagellar segment is longer than three times its width. The flagellum reaches the pterostigma when in repose. The
head hairs are entirely pale ochraceous to white, often somewhat paler ochraceous on the vertex, but in general,
all head hairs are pale. The thoracic hairs are white in the lateral areas, becoming paler ochraceous on the dorsum
except as follows: the scutellum usually bears many brown medial hairs (most distinctly so in Arizonan individuals,
but unknown in Oregonian specimens); and the mesoscutum, which ranges from being entirely pale ochraceous, to having
a patch of brown posteromedial hairs. The first tergum bears ochraceous hairs that, in the apical areas, become
appressed and sometimes nearly obscure the apical margin. The basal band of the second tergum is white. The interband
zone of the second tergum usually bears erect to suberect, ochraceous hairs, but in Arizonan individuals, these
hairs may be dark brown (unknown in Oregon). The distal pale band of the second tergum is approximately medially
as wide, or wider than, the apubescent apical area and pale ochraceous to white. The apical area of the second
tergum ranges from being entirely glabrous, to bearing suberect, long, usually pale hairs, but in Arizonan and
Nebraskan individuals, these may be brown (unknown in Oregon). Terga three, four, and five are similar to that
of the second, but the interband zones often bear brown hairs, the apical areas are absent to narrow, and the
basal areas tend to bear brown hairs. However, this isn’t always the case. The allotype male lacks all these
additive features, except on the fifth tergum. The sixth and seventh terga are shrouded in ochraceous hairs.
♀ Females are some of the palest Oregonian Melissodes with their vestiture being overall white, often having
little to no darkening on the dorsal thoracic areas and vertex, as well as many hairs present on these hairs,
surrounding the dark integument as described below. The eyes are grayish green to gray and the wing veins are
usually reddish brown to black, but often dark red. The underside of flagellar segments 3-10, and usually the
second in the apical one-third, are red to yellow (dark brown on top) and the apical one-third of the first
tergum is usually rufescent. The galeae are dull above due to shagreening, minimally in the apical one-third
to one-half and often more and the hairs on the mesoscutum are sometimes dark posteromedially, although many
individuals bear all light hairs. The interband zone of the second tergum has white, appressed to subappressed
pubescence with no darker spine-like hairs. The interband zone of the second tergum bears distinct, irregular,
small punctures. The head hairs are entirely pale ochraceous to white with no darkening on the vertex. The
thoracic hairs are mostly white in the lateral areas, becoming pale ochraceous on the dorsum except as follows:
the scutellum, which has a patch of brown medial hairs; and the mesoscutum, which has a brown posteromedial patch
of hairs, at most of similar size to the dark scutellar patch, but usually less. The first tergum’s basal area
bears pale ochraceous hairs, but in the apical areas, the tergum is glabrous. The basal pale pubescent band of
the second tergum is white. The interband zone of the second tergum bears short, pale ochraceous to white, suberect
to subappressed hairs. The distal pale pubescent band of the second tergum is near-white to white, often
uninterrupted medially, usually medially wider than that of the apical glabrous area (or of similar size),
often reaches the apex of the tergum in extreme lateral areas, and the apical hairs of the band are simple and
of similar length, or longer than, the hairs of the basal pubescent band. The third tergum is similar to that
of the second, except the distal pale pubescent band reaches the apical margin minimally in the lateral
one-thirds and often more (glabrous apical area narrower than that of the second tergum), the hairs on the
interband zone are dark brown, and the basal tomentum is dark brown. The fourth tergum is similar to that of
the third, except the distal pale pubescent band reaches the apex across the entire tergum. The fifth and sixth
terga are mostly shrouded with dark brown hairs, but laterally, pale tufts are present. The leg hairs are mostly
pale ochraceous to white, but the hairs on the inner surfaces of the hind basitarsi are yellow to red.
Similar Species
M. utahensis can resemble a few other Eumelissodes in both sexes, but likely more so in the males than females. Males are most similar to M. lutulenus, M. vernalis, and somtimes M. subagilis as outlined above, but likely more so the former than the rest. Range-wide, females can somtimes bear a striking resemblance to M. brevipyga, especially so in specimens that lack dark dorsal thoracic hairs. However, M. brevipyga has not been documented in, nor does it likely occur in Oregon. Therefore, Oregonian females of M. utahensis seem to instead be most similar to M. verbesinarum and M. subagilis due to the shared characteristics of near-white vestiture (more so with M. verbesinarum than M. subagilis), and the large distal band of the third tergum that minimally reaches the apex laterally.
♂ Males can be separated froEumelissodestepm those of M. lutulentus and M. subagilis in that The punctures on the interband zone of the second tergum are extremely tiny and mostly separated by more than, or equal to, three puncture diameters; rarely any wider than the bases of the hairs that arise from them. Also, the interband zone punctures of the third and fourth terga are very tiny to small, indistinct, and shallow and mostly separated by two puncture diameters (usually separated by less on the fourth tergum). Whereas in M. lutulentus and M. subagilis, the punctures on the interband zone of the second tergum are larger and mostly separated by two puncture diameters or less; each puncture is notably wider than the bases of the hairs that arise from them. And the interband zones of the third and fourth terga have large, deep, round punctures that are mostly separated by one puncture diameter. However, the tergal sculpturing as described above is shared by both M. utahensis and M. vernalis. As such, these two species are separated by labral markings, galeal sculpture, and pygidial plate structure. The labrum of M. utahensis often lacks a medial pale macula, but if bearing one, then the macula is less than one-third of the labral area. In M. vernalis, the labrum always bears a pale macula that is more than, or equal to, one-half of the labral area. The galeae of M. utahensis are dull above due to dense reticular shagreening and the pygidial plate is wide, with its width about two-thirds of its length. In M. vernalis, the galeae are shiny and unshagreened above apart from the tips and the pygidial plate is often narrow, with its width often less than two-thirds of its length (usually less than one-half).
♀ Females can be separated from those of M. subagilis by the overall pale vestiture, which is white rather than ochraceous to yellow. However, females bear a more direct resemblance to M. verbesinarum given the dulled galeae (although, dulled to a higher degree in M. utahensis) and overall white vestiture. M. utahensis can be separated from those of M. verbesinarum by distal pale band of the second tergum, which, in M. utahensis, is distinctly separated from the apical margin across most of, or all of the tergum medially so by approximately the same length of the distal pale band. In M. verbesinarum, this band usually reaches the apex across the tergum, or is minimally smaller than the medial portion of the distal band.
Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) utahensis in Oregon. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (The International Barcode of Life Consortium, 2026). Data liscenced under CC BY 4.0 (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Eumelissodes) utahensis< in Oregon and outside of Oregon (blue represents documentations in Oregon and red represents documentations outside of Oregon). The x value is the month, and the y value is the number of documented observations occurring in that month. Oregon data compiled from (The International Barcode of Life Consortium, 2026) and range-wide data compiled from (Ikerd, 2019; Johnson, 2020; Carril et al., 2023; Texas A&M University Insect Collection, 2023; Scott, 2025; Bentley & Osborn, 2026; Brigham Young University, Arthropod Collection, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Oram, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
At the time of his publication, Laberge (1961) only had a total of 36 M. utahensis specimens, 25 female and 11 male, which seems to be the full breadth of what was available during that period. Because of this, Laberge (1961) opted to omit a bionomics section, detailing floral records and preference, due to the paucity of his data (although, specimen labels with floral accounts were included). However, since 1961, 412 new publicly available records have been documented, 93 of which have floral data attached. From these data, a list of floral records as well as an approximation of M. utahensis’ floral preference will be detailed below. All flower records included in this list are from reports in the literature or datasets. Each flower has a parenthesized reference listed after it, corresponding to the literary work or dataset in which it was recorded. Currently, M. utahensis has been found atop Chrysothamnus sp. (Laberge, 1961), Chrysothamnus depressus (Carril et al., 2023; GBIF record 3421447849), Chrysothamnus viscidiflorus (Carril et al., 2023; GBIF record 3421490128), Cleomaceae serrulata (Carril et al., 2023; old synonym; GBIF record 3421519359), Cleome lutea (Carril et al., 2023; GBIF record 3421498116), Cryptantha sp. (Carril et al., 2023; GBIF record 3421497216), Ericameria nauseosa (Laberge, 1961: old synonym), Erigeron utahensis (Carril et al., 2023; GBIF record 3421447740), Eriogonum ovalifolium (Carril et al., 2023; GBIF record 3421474538), Euthamia occidentalis (MT James Entomological Collection, Washington State University, 2026; GBIF record 5141245836), Grindelia squarrosa (Scott, 2025; GBIF record 5283835842), Gutierrezia californica (Laberge, 1961), Gutierrezia sarothrae (Carril et al., 2023; GBIF record 3421481405), Haplopappus croceus (Illinois Natural History Survey, 2026; GBIF record 3801337921), Helianthus petiolaris (Carril et al., 2023; GBIF record 3421481386), Heterotheca villosa (Bentley & Osborn, 2026; GBIF record 1913918156), Lepidium montanum (Carril et al., 2023; GBIF record 3421491447), Lorandersonia linifolia (Carril et al., 2023; old synonym; GBIF record 3421445869), Machaeranthera sp. (Illinois Natural History Survey, 2026; GBIF record 3801339911), Melilotus officinalis (Carril et al., 2023; GBIF record 3421479989), Petradoria pumila (Carril et al., 2023; GBIF record 3421446849), Senecio flaccidus (Carril et al., 2023; old synonym; GBIF record 3421472520), Senecio spartioides (Carril et al., 2023; GBIF record 3421470134), Solidago sp. (Bentley & Osborn, 2026; GBIF record 658128379), Sphaeralcea grossulariifolia (Carril et al., 2023; GBIF record 3421465083), Stephanomeria exigua (Carril et al., 2023; GBIF record 3421480406), Stephanomeria tenuifolia (Carril et al., 2023; GBIF record 3421496315), Tamarix sp. (Carril et al., 2023; GBIF record 3421497255). Although the current data is fairly sparse, it seems as though M. utahensis has been collected atop eight plant families in the following order of abundance, Asteraceae, Boraginaceae, Cleomaceae, Polygonaceae, Brassicaceae, Fabaceae, Malvaceae, and Tamaricaceae. Of these families, Asteraceae, Boraginaceae, and Cleomaceae are the only ones from which M. utahensis has been collected more than once, and Asteraceae is the only one from which M. utahensis has been collected more than twice. Also, from all but Asteraceae, M. utahensis was only collected upon one taxon from each family. This is suggestive of Asteraceae oligolecty, with this species being collected from 19 different Asteraceae taxa, however, a non-biased sampling study will have to occur to truly understand the dietary preferences and/or restrictions of M. utahensis.
Synonyms
None.
Melissodes verbesinarum

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes verbesinarum
Melissodes verbesinarum is an interesting species in that rather than the females having two separate forms (as in M. bimatris) males seem to form two distinct groups. This species, seemingly in both sexes, is extremely pale, females having most (if not all) of their terga covered in fairly appressed pubescent bands that reach the apex, and males often bearing distinctly large basal mandibular macula. Although originally described as an Asteraceae oligolege (Laberge, 1961), this species has been collected from a total of 9 families (see “flower preference” below). Given the three distinct groups of males, distinctive features will be outlined for each separate group. The first group of males are distinctive in that the labrum is entirely light cream-colored to white with no darkening apically (much as in M. communis and M. tepidus), and the mandibular bases are almost entirely covered in pale macula that span the distance of the basal width. The second group of males are distinctive in that the labrum is often somewhat darkened apically; the mandibles bear yellow basal macula, be it smaller than those of group one; the penultimate flagellar segment is longer than three times its width and the flagella reach the pterostigma when in repose; the apical areas of the terga are opaque; the first flagellar segment’s maximum length is less than, or equal to, one-third of the third segment’s minimum length; the clypeus is entirely light excluding the dark spots denoting the tentorial depressions or the anterior notches and margin; the galeal are often shiny with no noticeable shagreening and the dorsal thoracic hairs are white to cinereous; and the first flagellar segment’s maximum length is much less than one-third of the third segment’s maximum length, and the minimum length of the first flagellar segment is barely, if at all, longer than the pedicel’s length on the same side. Something of note, although the first group of males is distinctive in a combination of only two characteristics, males from the two groups still share many traits and the lack of descriptive factors does not represent a lack of traits. Females vary much less than their male counterparts, only having a single group, and are distinctive in that the distal band of the third tergum usually reaches the apex across the entire tergum (if apical area present, then much narrower than the second tergum’s distal band medially); the underside of flagellar segments 3-10 are rufescent; the interband zone of the second tergum has distinct punctures that are mostly separated by faintly more than, or equal to, one puncture diameter and bears white, appressed to subappressed pubescence with no darker spine-like hairs; the distal pale band on the second tergum often reaches the apex across the entire tergum (if apical area present, then narrower than one-half of the medial width of the distal pale band); and the galea are shiny with no shagreening except near the tips. This species has yet to be documented in Oregon, but given its presence in northern California (Ikerd, 2019), Idaho (MT James Entomological Collection, Washington State University, 2026), and Washington (Laberge, 1961; not plotted below due to lack of coordinates), this species has a high likelihood of occurring in this region and is herein included in this guide (Fig. ??). Historically, this species has been known to be active through the months of April to November with peak activity occurring in June, July, and August (Laberge, 1961; based on 284 specimen records). Newer data show a similar pattern, but two distinct peaks in phenology seem to be present, one in May and the other around August and September (Fig. ??; based on 437 records). However, this may be due to sampling bias and until dedicated sampling efforts occur, the full bredth of this species’ phenology is unknown.
Field Markers
Although males fall into two groups (apparent in the key above), both groups of males are very similar in
vestiture and structure, only differing in a few key areas. As such, two male descriptions (as for females
in M. bimatris or for both sexes in M. ablusus) seem redundant. Instead, one description will be given that
highlights and addresses variable characteristics.
♂ Similar to most males, M. verbesinarum is very pale, but does have a medially interrupted apical band,
similar to those of M. bimatris or M. semilupinus, but not distinctly obscuring the medial portion. The eyes
are bluish gray to gray and the clypeus is yellow except for the darkened tentorial depressions and the
testaceous apical margin. The labrum is either entirely near-white to white, is near-white to white but has
its apical margin testaceous, or is black with a large mediobasal pale macula. The mandibles either bear
comparatively "normal-sized" macula for a male Melissodes, or bear large yellow macula basally that often
encompass the entire basal area (no distinct yellow triangular area, whole base yellow). The wing veins are
red to yellow and the flagella are reddish yellow on the underside, brownish red dorsally, and the first
segment is wholly brown to red. The terga are colorless to faintly yellow hyaline in the apical areas. The
clypeus is flat without a large bulge or concavity. The oculoclypeal distance is less than or equal to half
of the first flagellar segment’s minimum width. The first flagellar segment’s minimum length is usually
equivalent to one-ninth the second segment’s maximum length, and on rare occasions it can be as long as
one-eighth the maximum length of the second segment. The minimum length of the first flagellar segment
(smaller side of F1) is often more than half the maximum length (longer side of F1). Also, the first
flagellar segment’s maximum length is much less than one-third of the third segment’s maximum length,
and the minimum length of the first flagellar segment is barely, if at all, longer than the pedicel’s
length on the same side. The length of the penultimate flagellar segment is longer than three times its
width. The flagellum reaches the pterostigma when in repose. The galeae are shiny above with no shagreening,
except for the tips. The head hairs are entirely white. The thoracic hairs are mostly white, but become faintly
cinereous on the dorsal thoracic areas. The first tergum bears long ochraceous to white basal hairs, and in
apical areas, these hairs become appressed, somewhat simple with few branches, wire-like, and exceed the apical
margins, but only obscure it in the lateral areas, not entirely obscuring it medially. The distal pale pubescent
band of the second tergum is always medially wider than, or of similar width to, the tergum’s apical area. The
apical area of the second tergum bears comparably simple, long, suberect to subappressed, yellow to white hairs
(no brown hairs present). The third, fourth, and fifth terga are similar to that of the second, except the apical
areas posterior to the pubescent bands become shorter with each subsequent tergum until the band reaches the
apex across the apex across the entire tergum on T5. The sixth and seventh terga are completely covered in
white to dark ochraceous hairs.
♀ Females are strikingly and beautifully marked, with the second, third, and fourth terga often being entirely
shrouded in pale ochraceous to white hairs (as described below), in this regard, similar to those of M. subagilis.
The eyes are bluish gray to gray and the wing veins are usually brown to reddish brown. The underside of flagellar
segments 3-10, which are rufescent and the underside of the second flagellar segment is often entirely black,
but if rufesenet, then only so in the apical most areas. The apical one-third of the first tergum, which is
usually rufescent. The galeae are mostly shiny and unshagreened apart from the tips and the interband zone of
the second tergum is distinctly punctate with punctures that are mostly separated by faintly more than, or equal
to, one puncture diameter. Key diagnostic characteristics of M. verbesinarum females, although many exist within
the structure and sculpture, are mostly derived from their vestiture. As such, a full vestiture description will
be given below due to its importance in identification. The head hairs are mostly to entirely white, but if not
entirely so, then a few brown hairs are present on the vertex. The thoracic hairs are white in the lateral areas,
becoming pale ochraceous dorsally except as follows: the scutellum, which is mostly shrouded in brown hairs,
peripherally margined with white; and the mesoscutum, which bears a small dark posteromedial patch that’s of
similar size or smaller (very rarely larger) than the dark patch of scutellar hairs, but on occasion, all mesoscutal
hairs are pale ochraceous. The basal band of the second tergum bears long white hairs. The interband zone of the
second tergum bears subappressed, short, white (medially often mixed with short brown hairs) hairs. The distal pale
band of the second tergum is white, complete, wider than the interband zone’s width, and reaches the apex of the
tergum, minimally so in the lateral one-thirds of either side of the tergum, and often medially as well. However,
if the distal pale band is distinctly separated from the apical area, then by no more than half of the width of
the distal pale band’s width. The third and fourth terga are similar to that of the second, but the distal pale
bands are rarely largely separated from the apical margin of the tergum and the basal tomentum is brown. If the
third tergum is separated from the apex of the tergum, then by much less than the width of the second tergum’s
band. The fifth and sixth terga are shrouded in dark brown hairs, becoming white tufts laterally. The sternal hairs
are mostly brownish red to gold-colored, becoming white in the lateral areas. However, on the second to last sterna,
the hairs become white apically. The leg hairs are mostly white, differing at the inner surfaces of the hind
basitarsi, which range from brownish red, to red, to yellow; the basitibial plates, which are brown; the scopal
hairs, which are white; the fore tarsi, which are pale brown; and the apical portions of the outer surfaces of
the middle tibiae, which are brownish orange.
Similar Species
Given how variable males are, M. verbesinarum will have two comparisons, each corresponding to the group’s traits listed above. Females seem to mostly resemble those of M. subagilis, but to a degree M. stearnsi and M. utahensis as well. The first group of males are counterintuitively superficially most similar to those of the subgenus Melissodes ss. due to the non-darkened labral margin. However, the second group of males are fairly similar to M. menuachus.
♂ (1) This group of M. verbesinarum males are fairly similar to M. communis and M. tepidus in that the labrum is entirely white with no darkened apical margin. This is interesting given that a classical subgeneric characteristic of the subgenus Melissodes s.s. is the coloration of the labrum as described above. M. verbesinarum can be separated from those of M. communis by the hairs on the distal most two terga being dark ochraceous to white rather than black, and from those of M. tepidus by the distinctly transparent to faintly yellow translucent hyaline tergal rimes rather than the darkened translucent to testaceous rims of M. tepidus. Along with this, the first group of M. verbesinarum can be separated from both these species by the large mandibular macula that encompasses the majority of the basal mandibular width.
♂ (2) This group of M. verbensinarum males are fairly similar to the first group in that the basal mandibular macula are abnormally large and often span the width of the basal mandibular area, but the labrum is instead black with a large mediobasal pale macula. Although the large yellowed base of the mandible is highly diagnostic of M. verbesinarum, this group of males can superficially M. menuachus. The two species can be separated by the first flagellar segment’s length. That being, in M. verbesinarum, the maximum length of this segment is much less than one-third of the third segment’s maximum length, and the minimum length of the first flagellar segment is barely, if at all, longer than the pedicel’s length on the same side. However, in M. menuachus, the first flagellar segment’s maximum length is equal to, or almost equal to, one-third of the third segment’s maximum length, and the minimum length of the first flagellar segment is notably longer than the pedicel’s length on the same side.
♀ Females can be separated from those of M. utahensis by the galeal sculpture. That being, in M. verbesinarum, the galeae are shiny with no shagreening apart from the tips, whereas in M. utahensis, the galeae are distinctly shagreened, minimally so in the apical one-half and often more. M. verbesinarum can also be separated from M. utahensis by the distal pale band of the second tergum, which often reaches the apex across the entire tergum, but if not, then the apical area is narrower than one-half of the distal pale band’s width. In M. utahensis, the distal pale band of the second tergum usually reaches the apex laterally, but the apical area is medially distinct and of similar size, or slightly narrower than, the width of the distal pale band medially. M. stearnsi, a Callimelissodes, although not immensely similar to M. verbesinarum is listed in this comparison due to the diffuse white tergal pubescence which often reaches the apical margin of all but the first tergum. However, as can be determined from the different subgenera (Eumelissodes vs. Callimelissodes), the two species greatly differ in many traits. The greatest distinction between the two, surprisingly, is the tergal vestiture. In M. verbesinarum, the terga bear long plumous hairs and form distinct wide bands across each terga. However, in M. stearnsi, the terga are covered in diffuse short pubescence with no distinct bands being formed. Also, the two largely differ in that M. stearnsi has noticeably long hooked (almost curled) galeal hairs, whereas M. verbesinarum lacks these hairs. Lastly, the most prominent resemblance females of this species bear to any other Oregonian Melissodes is to M. subagilis. While these two species are incredibly similar in the tergal vestiture (both having distinctly wide tergal bands that often reach the apex across multiple terga), M. subagilis has very dull galeae due to dense shagreening (similar in sculpture to M. agilis), whereas M. verbesinarum has shiny and fairly unshagreened galeae as described above.
Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) verbesinarum. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Johnson, 2020; Carril et al., 2023; Texas A&M University Insect Collection, 2023; Droege & Maffei, 2025; Scott, 2025; Sánchez & Comisión, 2025; Bentley & Osborn, 2026; Gibbs, 2026; Grinter et al., 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Northern Arizona University, 2026a; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026; University of Arizona Insect Collection, 2026b). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Eumelissodes) verbensinarum. The x value is the month, and the y value is the number of documented observations occurring in that month. Data compiled from (Ikerd, 2019; Johnson, 2020; Carril et al., 2023; Texas A&M University Insect Collection, 2023; Droege & Maffei, 2025; Scott, 2025; Sánchez & Comisión, 2025; Bentley & Osborn, 2026; Gibbs, 2026; Grinter et al., 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Northern Arizona University, 2026a; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026; University of Arizona Insect Collection, 2026b). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0 as applicable (see "Dataset Licenses").
Flower preference
M. verbesinarum has been categorized as an Asteraceae oligolege (Laberge, 1961), and although the current data are few, newer records seem to suggest a similar scenario. Given that only 87 floral records are publicly available, and Laberge (1961) examined 175 M. verbesinarum with floral records attached, the percentages below will be derived from his paper. Although he didn’t fully list all floral records, Laberge (1961) did state that of the 175 specimens collected with floral data attached, 159 were collected atop Asteraceae. This renders Asteraceae to be favored by M. verbesinarum 90.85% of the time.
Synonyms
Melissodes pecosella verbesinarum, Cockerell, 1905.
Melissodes vernalis

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes vernalis
Although generally males are the most distinctive and easily identifiable sex in Melissodes, M. vernalis is the opposite. Females, just like the females of M. saponellus, are highly distinctive in that the tergal rims of terga 2-4 (and portions of 1 as well) are largely hyaline, creating somewhat of a male-like integument. This characteristic is very rare within Melissodes, with only three species range-wide sharing it (M. vernalis, M. saponellus, and M. velutinus), two of which occur in Oregon. Although speculatory, this is somewhat suggestive of an ancient similar ancestor between the two hyaline-edged Oregonian females given the characteristic’s rareness. Interestingly, M. vernalis (apart from integumental color) seems to be more closely related to M. limbus, which is in-turn somewhat related to M. subagilis, than it is to M. saponellus or M. velutinus, the two latter seemingly forming a clade (although, not in terms of physiology). It is, however, undeniable that these three species are extremely closely related, but an in-depth phylogenetic analysis with an inclusion of these species may reveal new insights. Males are distinctive in that the labrum has a pale macula that is more than, or equal to, one-half of the labral area; the galeae are shiny and unshagreened above apart from the tips; the pygidial plate is often narrow with its width often less than two-thirds of its length (usually less than one-half); the punctures on the interband zone of the second tergum are extremely tiny and mostly separated by more than, or equal to, three puncture diameters (rarely any wider than the bases of the hairs that arise from them); the interband zone punctures of the third and fourth terga are also very tiny to small, indistinct, and shallow and mostly separated by two puncture diameters (usually separated by less on the fourth tergum); the mandibular bases are entirely black; the clypeus is entirely light excluding the dark spots denoting the tentorial depressions or the anterior notches and margin; the first flagellar segment’s maximum length is less than, or equal to, one-third of the third segment’s minimum length; the apical areas of the terga are hyaline; The length of the penultimate flagellar segment is longer than three times its width and the flagellum reaches the pterostigma when in repose. Females are distinctive in the presence of hyaline tergal rims on terga 2-4; the mesoscutum, which has a large brown patch of hairs; the hairs on the apical areas of the second and third terga, which are dark brown; and the hairs across the fifth and sixth terga, which are usually dark brown as well. Although this species has yet to be found in Oregon, occurrence reports in Nevada (Bentley & Osborn), Washington (MT James Entomological Collection, Washington State University, 2026), California (Johnson, 2020), and Wyoming (Droege & Maffei, 2025), along with Laberge’s (1961) original distribution map (Fig. 35 in Laberg, 1961) are suggestive of an Oregonian presence and is therefore included in this field guide (Fig. ??). M. vernalis has been historically believed to be active throughout the months of March to October with peak activity occurring in April, May, and June (Laberge, 1961). Although the current data is sparse (41 records), a similar pattern seems to appear, but M. vernalis may have even been collected as early as February (Fig. ??).
Field Markers
♂ Males are fairly pale with entirely light head hairs, but the dorsal thoracic area does bear a dark patch
as described below. The eyes are often blue to greenish blue, and sometimes gray to yellowish gray. The
clypeus is yellow apart from the darkened tentorial depressions and the testaceous apical margin. The labrum
bears a large mediobasal cream to white macula that is minimally more than one-third of the labrum’s area,
and usually more than one-half and the wing veins are black to dark reddish brown. The mandibular bases are
entirely dark with no yellow macula. The underside of the flagella are red to yellow (F1 sometimes entirely
dark brown) and the terga are colorless to faintly yellow hyaline in the apical areas. The first flagellar
segment’s minimum length is usually equivalent to one-sixth to one-eighth the second segment’s maximum length.
The minimum length of the first flagellar segment (smaller side of F1) is often more than half the maximum
length (longer side of F1). The first flagellar segment’s maximum length is less than, or equal to, one-third
of the third segment’s minimum length. The length of the penultimate flagellar segment is longer than three
times its width. The flagellum reaches the pterostigma when in repose. The length of the pygidial plate is
often twice the medial width (long and narrow) and has indistinct indentations apicolaterally. The galeae are
shiny and unshagreened above apart from the tips and the punctures on the interband zone of the second tergum
are extremely tiny and mostly separated by more than, or equal to, three puncture diameters; rarely any wider
than the bases of the hairs that arise from them. The interband zone punctures of the third and fourth terga
are also very tiny to small, indistinct, and shallow and mostly separated by two puncture diameters (usually
separated by less on the fourth tergum). The thoracic hairs are mostly white, differing at the scutellum,
which bears many brown hairs posteromedially; and the mesoscutum, which bears a large brown patch that’s
usually of similar size to, or faintly larger than, the scutellar dark patch. The first tergum bears long
white basal hairs, and in apical areas, these hairs become appressed, somewhat simple with few branches,
and exceed the apical margins, but only obscure it in the lateral areas, not obscuring it medially. The
distal white pubescent band of the second tergum is medially a similar width to, or faintly narrower than,
the tergum’s medial portion of the apical area. The apical area of the second tergum bears comparably simple,
long, suberect to subappressed, yellow to white hairs (often white, no brown hairs present). The third,
fourth, and fifth terga are similar to that of the second, except the apical areas posterior to the pubescent
bands become shorter with each subsequent tergum until the band reaches the apex across the apex across the
entire tergum on T5. The sixth and seventh terga are completely covered in white to dark ochraceous hairs
(often white).
♀ Females, although not distinctly light or dark in vestiture, can look extremely pale (almost to the same
level as M. vernalis and M. subagilis) due to the hyaline tergal rims appearing light-colored. The eyes are
vibrant green to gray and the wing veins are black to dark brown. The underside of flagellar segments 3-10,
and usually the second in the apical one-third are red to yellow (dark brown on top) and the apical area of
the first tergum is widely colorless to reddish hyaline. The apical areas of the second, third, and fourth
terga are widely colorless hyaline to somewhat smokey translucent (much as in most male Melissodes). Although
the characteristic of hyaline tergal rims on a female Melissodes is distinctive enough to separate this
species from all but one in Oregon (M. saponellus), the role that vestiture plays in female identification
is fairly important, and therefore, a setal description will be given below. The head hairs are mostly white,
differing at the vertex, which bears long brown hairs. The thoracic hairs are laterally white, becoming white
to pale ochraceous on the dorsum except as follows: the scutellum, which bears abundant brown hairs; and the
mesoscutum, which bears a large brown patch of hairs that ranges in size from being similar in area to that of
the scutellar dark patch, to twice the area of the scutellar dark patch. The basal area of the first tergum
bears long white hairs, but apically becomes glabrous apart from the rounded lateral projections, which usually
bear simple, brown, minute, closely spaced, appressed hairs. The basal band of the second tergum is white. The
interband zone of the second tergum bears suberect to appressed, short, dark brown hairs, and in width, is
approximately equal to the distal pale band medially. The distal pale band of the second tergum is white, and
the apical area bears many dark brown suberect hairs. The third tergum is similar to that of the second, but
the apical area is narrower to entirely absent and the basal tomentum is brown. The fourth tergum is similar
to that of the third, but the distal pale band reaches the apex across the entire tergum. The fifth and sixth
terga are mostly shrouded in pale brown hairs that are basally darkened and become white laterally forming
distinct pale tufts. The leg hairs are mostly white, but the inner surfaces of the basitarsi (including the
hind basitarsi) are red to yellow.
Similar Species
From what can be discerned above, females of M. vernalis only resemble one other species, that is M. saponellus, and are incredibly distinct from all other Melissodes. Males on the other hand aren’t as easily identifiable, often resembling a few other Eumelissodes, most notably M. utahensis due to similar interband zone punctuation, and M. subagilis due to similar structure, integumental coloration, and somewhat similar vestiture.
♂ Males of M. vernalis can be separated from those of M. utahensis by the labrum, which has a pale macula that is more than, or equal to, one-half of the labral area; the galeae, which are shiny and unshagreened above apart from the tips; and the pygidial plate, which is often narrow, with its width often less than two-thirds of its length (usually less than one-half). For contrast, the labrum of M. utahensis often doesn't have a medial pale macula, or if it does, then the macula is less than one-third of the labral area; the galeae are dull above due to dense reticular shagreening; and the pygidial plate is wide, with its width about two-thirds of its length. Males can also be separated from M. subagilis by the punctures of the interband zone of the second tergum being extremely tiny and mostly separated by more than, or equal to, three puncture diameters (rarely any wider than the bases of the hairs that arise from them); and the punctures of the interband zone of the third and fourth terga, which are also very tiny to small, indistinct, shallow, and mostly separated by two puncture diameters (usually separated by less on the fourth tergum). For contrast, the punctures on the interband zone of the second tergum of M. subagilis are fairly large and mostly separated by two puncture diameters or less (each puncture is notably wider than the bases of the hairs that arise from them); and the interband zones of the third and fourth terga have large, deep, round punctures that are mostly separated by one puncture diameter.
♀ Females, as state above, only resemble M. saponellus in Oregon and are surprisingly simple to separate. M. vernalis has a large dark mesoscutal patch of hairs, the hairs on the apical areas of the second and third terga are dark brown, and the hairs across the fifth and sixth terga are usually dark brown as well. In M. saponellus, the mesoscutal hairs are entirely to mostly pale (only a minute amount brown if present at all), the apical hairs on the second and third terga are white, and the hairs across the fifth and sixth terga are ochraceous to white.
Fig. ??. Map showing an estimation for the known distribution of M. (Eumelissodes) vernalis. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data compiled from (Ikerd, 2019; Johnson, 2020; Droege & Maffei, 2025; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026). Data licensed under CC BY 4.0, CC0 1.0, and CC BY-NC 4.0 as applicable (see "Dataset Licenses").
Fig. ??. A graph representing the phenology of M. (Eumelissodes) vernalis. The x value is the month, and the y value is the number of documented observations occurring in that month. Data compiled from (Ikerd, 2019; Johnson, 2020; Droege & Maffei, 2025; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Orrell & Informatics, 2026a; The International Barcode of Life Consortium, 2026). Data licensed under CC BY 4.0, CC0 1.0, and CC BY-NC 4.0 as applicable (see "Dataset Licenses").
Flower preference
M. vernalis seems to be an oligolege of Asteraceae (as are most Melissodes). This species was historically known to be an Asteraceae oligolege, but recent studies allude to the fact that M. vernalis may in fact be a generalist with a strong Asteraceae preference (Wright, 2018). A deeper species specific analysis and non-bias sampling efforts will have to take place for the full breadth of M. vernalis’ diet to be understood.
Synonyms
None.
References
Best, L. R., Marshall, C. J., Feuerborn, C., Kincaid, S., Melathopoulos, A., & Robinson, S.V.J. (2021b). Oregon Bee Atlas: native bee findings from 2018. Catalog: Oregon State Arthropod Collection, 5(1). https://doi.org/10.5399/osu/cat_osac.5.1.4647.
Best, L. R., Engler, J., Feuerborn, C., Larson, J., Marshall, C. J., Kincaid, S., Melathopoulos, A., & Robinson, S. V. J. (2022b). Oregon Bee Atlas: wild bee findings from 2019. Catalog: Oregon State Arthropod Collection, 6(1). https://doi.org/10.5399/osu/cat_osac.6.1.4906
LaBerge, W.E. (1961) ‘A revision of the bees of the genus melissodes in north and Central America. part III (hymenoptera, Apidae)’, The University of Kansas science bulletin, 42(5), pp. 283–663. doi:10.5962/bhl.part.9821.
LaBerge, W.E. (1956a) ‘A revision of the bees of the genus melissodes in north and Central America. part I. (Hymenoptera, Apidae)’, The University of Kansas science bulletin, 37(18), pp. 911–1194. doi:10.5962/bhl.part.24549.
LaBerge, W.E. (1956b) ‘A revision of the bees of the genus melissodes in north and Central America. part II (hymenoptera, Apidae)’, The University of Kansas science bulletin, 38(8), pp. 533–578. doi:10.5962/p.376392.
Wilson, J.S. et al. (2025) ‘A checklist of the Bees of Utah’, Diversity, 17(3), p. 212. doi:10.3390/d17030212.
LaBerge, Wallace E. (1963), "New Species and Records of Little-known Species of Melissodes from North America (Hymenoptera:Anthophoridae)". Bulletin of the University of Nebraska State Museum. 9. http://digitalcommons.unl.edu/museumbulletin/9
North American Native Bee Collaborative. (2020). Bees of Ohio: A Field Guide. Ohio, USA.
LaBerge, Wallace E. (1957), "The Genera Bees of the Tribe Eucerini in North and Central America (Hymenoptera, Apoidea)". Kr. Paper 4. https://digitalcommons.usu.edu/bee_lab_kr/4
Michener, C.D. (2007) “The bees of the world” Vol 2. Baltimore: Johns Hopkins University Press.
accessed via GBIF.org on 2025-12-06.
Gloria DeGrandi-Hoffman, Mona Chambers, Judith E Hooper, Stanley S Schneider, (2004), "Description of an Intermorph Between a Worker and Queen in African Honey Bees Apis mellifera scutellata (Hymenoptera: Apidae)", Annals of the Entomological Society of America, 97(6), p. 1299–1305, https://doi.org/10.1603/0013-8746(2004)097[1299:DOAIBA]2.0.CO;2
Washington State Department of Agriculture. (2025). Washington Bee Atlas [Online citizen-science project]. https://agr.wa.gov/departments/insects-pests-and-weeds/insects/apiary-pollinators/pollinator-health/bee-atlas
Wright, K.W., Miller, K.B. and Song, H. (2020) ‘A molecular phylogeny of the long-horned bees in the genus Melissodes latreille (hymenoptera: Apidae: Eucerinae)’, Insect Systematics & Evolution, 52(4), pp. 428–443. doi:10.1163/1876312x-bja10015.
Wright, Karen W. "THE EVOLUTION OF DIET BREADTH IN MELISSODES BEES (APIDAE: EUCERINI)." (2018). https://digitalrepository.unm.edu/biol_etds/301
PORTMAN, Z.M., NEFF, J.L. and GRISWOLD, T. (2016) ‘Taxonomic revision of Perdita subgenus Heteroperdita Timberlake (hymenoptera: Andrenidae), with descriptions of two ant-like males’, Zootaxa, 4214(1). doi:10.11646/zootaxa.4214.1.1.
Cruden, R. W., & Jensen, K. G.. (1979) Viscin Threads, Pollination Efficiency and Low Pollen-Ovule Ratios. *American Journal of Botany*, *66*(8), 875. https://doi.org/10.2307/2442226
Halbritter H. 2016. Clarkia unguiculata. In: PalDat - A palynological database. https://www.paldat.org/pub/Clarkia_unguiculata/301507; accessed 2026-03-19
Graham M, Best L, Moldenke A (2021). Digital record of specimens, including voucher material, from the study of a pollinator habitat restoration site under a commercial solar array in Jackson County, Oregon, 2019.. Oregon State University. Occurrence dataset https://doi.org/10.5399/osu/cat_osac.5.2.4855 accessed via GBIF.org on 2026-03-10.
Best L (2026). Oregon Bee Atlas. Oregon State University. Occurrence dataset https://doi.org/10.15468/fqxchw accessed via GBIF.org on 2026-03-10.
Frost Entomological Museum (2026). Frost Entomological Museum. Occurrence dataset https://doi.org/10.15468/epw1ws accessed via GBIF.org on 2026-03-10.
MT James Entomological Collection, Washington State University (2026). Washington State University Collection. Occurrence dataset https://doi.org/10.15468/c8yk6t accessed via GBIF.org on 2026-03-10.
Best L, Engler J, Feuerborn C, Larsen J, Lindh B, Kincaid S, Melathopoulos A, Marshall C J, Marshall C (2022a). Oregon Bee Atlas Survey Data: 2019. Version 1.3. Oregon State University. Occurrence dataset https://doi.org/10.15468/kuwm6h accessed via GBIF.org on 2026-03-10.
Gibbs J (2025). J. B. Wallis / R. E. Roughley Museum of Entomology. Version 1.2. University of Manitoba. Occurrence dataset https://doi.org/10.5886/hgagiy accessed via GBIF.org on 2026-03-10.
Droege S, Maffei C (2025). Insect Species Occurrence Data from Multiple Projects Worldwide with Focus on Bees and Wasps in North America. Version 1.27. United States Geological Survey. Sampling event dataset https://doi.org/10.15468/6autvb accessed via GBIF.org on 2026-03-10.
Best L, Feuerborn C, Holt J, Kincaid S, Marshall C, Melathopoulos A (2021a). Oregon Bee Atlas Survey Data: 2018. Version 1.5. Oregon State University. Occurrence dataset https://doi.org/10.15468/dmyc73 accessed via GBIF.org on 2026-03-10.
Ikerd H, Engler J (2023). Bee Fauna of National Wildlife Refuges in the Pacific Northwest, 2010-2016. USDA-ARS Pollinating Insect-Biology, Management, Systematics Research. Occurrence dataset https://doi.org/10.15468/ppjnys accessed via GBIF.org on 2026-03-10.
The International Barcode of Life Consortium (2026). International Barcode of Life project (iBOL). Occurrence dataset https://doi.org/10.15468/inygc6 accessed via GBIF.org on 2026-03-10.
Colorado State University, C.P. Gillette Museum of Arthropod Diversity (2026). C.P. Gillette Museum of Arthropod Diversity. Occurrence dataset https://doi.org/10.15468/k0aett accessed via GBIF.org on 2026-03-10.
Motz G (2026). Entomology Division, Yale Peabody Museum. Yale University Peabody Museum. Occurrence dataset https://doi.org/10.15468/95waq3 accessed via GBIF.org on 2026-03-10.
Bentley A, Osborn R (2026). Snow Entomological Museum Collection. University of Kansas Biodiversity Institute. Occurrence dataset https://doi.org/10.15468/fhntpy accessed via GBIF.org on 2026-03-10.
Johnson N (2026). Triplehorn Insect Collection, The Ohio State University. Museum of Biological Diversity, The Ohio State University. Occurrence dataset https://doi.org/10.15468/efb17f accessed via GBIF.org on 2026-03-10.
Illinois Natural History Survey (2026). Illinois Natural History Survey Insect Collection. Occurrence dataset https://doi.org/10.15468/eol0pe accessed via GBIF.org on 2026-03-10.
Gross J, Oboyski P (2026). Essig Museum of Entomology. Version 121.432. Berkeley Natural History Museums. Occurrence dataset https://doi.org/10.15468/0saucj accessed via GBIF.org on 2026-03-10.
Ikerd H (2019). Bee Biology and Systematics Laboratory. USDA-ARS Pollinating Insect-Biology, Management, Systematics Research. Occurrence dataset https://doi.org/10.15468/anyror accessed via GBIF.org on 2026-03-10.
Johnson C (2020). hymenoptera. Version 1.3. American Museum of Natural History. Occurrence dataset https://doi.org/10.15468/mvtuf5 accessed via GBIF.org on 2026-03-10.
Grinter C, Diaz-Bastin R, Fong J (2026). CAS Entomology Type (TYPE). Version 1.332. California Academy of Sciences. Occurrence dataset https://doi.org/10.15468/gak5hc accessed via GBIF.org on 2026-03-10.
Grant S, Webbink K, Turcatel M, Shuman R (2025). Field Museum of Natural History (Zoology) Insect, Arachnid and Myriapod Collection. Version 12.64. Field Museum. Occurrence dataset https://doi.org/10.15468/0ywfpc accessed via GBIF.org on 2026-03-10.
Orrell T, Informatics and Data Science Center - Digital Stewardship (2026a). NMNH Extant Specimen Records (USNM, US). Version 1.106. National Museum of Natural History, Smithsonian Institution. Occurrence dataset https://doi.org/10.15468/hnhrg3 accessed via GBIF.org on 2026-03-10.
San Diego Natural History Museum (2026). San Diego Natural History Museum Entomology Department. Occurrence dataset https://doi.org/10.15468/dpyqml accessed via GBIF.org on 2026-03-10.
Texas A&M University Insect Collection (2023). Texas A&M University Insect Collection. Occurrence dataset https://doi.org/10.15468/caprqh accessed via GBIF.org on 2026-03-10.
University of Minnesota Insect Collection (2026). University of Minnesota Insect Collection. Occurrence dataset https://doi.org/10.15468/ahwyqb accessed via GBIF.org on 2026-03-10.
Oram R (2026). Royal Saskatchewan Museum - Entomology. Version 2.2. Royal Saskatchewan Museum. Occurrence dataset https://doi.org/10.5886/osnmhn accessed via GBIF.org on 2026-03-10.
Scott V (2025). UCM Entomology Collection. Version 6.6. University of Colorado Museum of Natural History. Occurrence dataset https://doi.org/10.15468/jsgtns accessed via GBIF.org on 2026-03-10.
Museum of Southwestern Biology (2026). Museum of Southwestern Biology Division of Arthropods. Occurrence dataset https://doi.org/10.15468/jtovgy accessed via GBIF.org on 2026-03-10.
Kenneth S. Norris Center for Natural History (2026). Kenneth S. Norris Center for Natural History, University of California Santa Cruz, Insect Collection. Occurrence dataset https://doi.org/10.15468/hzkqkp accessed via GBIF.org on 2026-03-10.
Carril O, Wilson J, Griswold T, Ikerd H I (2023). Wild bees of Grand Staircase-Escalante National Monument. USDA-ARS Pollinating Insect-Biology, Management, Systematics Research. Occurrence dataset https://doi.org/10.7717/peerj.5867 accessed via GBIF.org on 2026-03-10.
University of Arizona Insect Collection (2026a). University of Arizona Insect Collection. Occurrence dataset https://doi.org/10.15468/hzkbpg accessed via GBIF.org on 2026-03-10.
Brigham Young University, Arthropod Collection (2026). Brigham Young University Arthropod Museum. Occurrence dataset https://doi.org/10.15468/gqf6no accessed via GBIF.org on 2026-03-10.
Mertz W, Kung G, Xie W (2026). LACM Entomology Collection. Version 5.33. Natural History Museum of Los Angeles County. Occurrence dataset https://doi.org/10.15468/kc9hyp accessed via GBIF.org on 2026-03-10.
Mississippi Entomological Museum (2026). Mississippi Entomological Museum. Occurrence dataset https://doi.org/10.15468/iuhxpw accessed via GBIF.org on 2026-03-10.
Cheadle Center for Biodiversity and Ecological Restoration (2026a). UCSD Holway Lab - Longterm Habitat Fragmentation Study. Occurrence dataset https://doi.org/10.15468/74ktmt accessed via GBIF.org on 2026-03-10.
Luther College (2026). Luther Entomological Research Collection. Occurrence dataset https://doi.org/10.15468/vy5e54 accessed via GBIF.org on 2026-03-10.
UMMZ Insects Division Data Group, LSA IT A (2026). University of Michigan Museum of Zoology, Division of Insects. Version 1.107. University of Michigan Museum of Zoology. Occurrence dataset https://doi.org/10.15468/tmxd7n accessed via GBIF.org on 2026-03-10.
Wild A (2026). University of Texas, Biodiversity Center, Entomology Collection (UTIC). Version 1.304. University of Texas at Austin, Biodiversity Collections. Occurrence dataset https://doi.org/10.15468/sanyq7 accessed via GBIF.org on 2026-03-10.
Natural Science Research Laboratory, Museum of Texas Tech University (TTU) (2026). Museum at Texas Tech University Invertebrate Zoology Collection. Occurrence dataset https://doi.org/10.15468/ga4bmd accessed via GBIF.org on 2026-03-10.
Cheadle Center for Biodiversity and Ecological Restoration (2026b). University of California Santa Barbara Invertebrate Zoology Collection. Occurrence dataset https://doi.org/10.15468/w6hvhv accessed via GBIF.org on 2026-03-10.
University of Kentucky (2026). University of Kentucky Insect Collection. Occurrence dataset https://doi.org/10.15468/fi4vfu accessed via GBIF.org on 2026-03-10.
Veit M, McFarland K (2024). Bee Specimens from Michael Veit Collection. Version 1.3. Vermont Center for Ecostudies. Occurrence dataset https://doi.org/10.15468/b8sg4a accessed via GBIF.org on 2026-03-10.
Holm M, Houle M, Salem M (2025). U.S. Fish and Wildlife Service Great Lakes Native Bee Survey. Version 1.14. United States Fish and Wildlife Service. Sampling event dataset https://doi.org/10.15468/jr59af accessed via GBIF.org on 2026-03-10.
Florida State Collection of Arthropods (2026). Florida State Collection of Arthropods. Occurrence dataset https://doi.org/10.15468/25xdz4 accessed via GBIF.org on 2026-03-10.
Virginia Tech Insect Collection (2026). Virginia Polytechnic Institute and State University Insect Collection. Occurrence dataset https://doi.org/10.15468/m2htqa accessed via GBIF.org on 2026-03-10.
Johnson N (2019). Cleveland Museum of Natural History. Museum of Biological Diversity, The Ohio State University. Occurrence dataset https://doi.org/10.15468/bmfgag accessed via GBIF.org on 2026-03-10.
Quiroz Martínez H, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Abejas silvestres (Hymenoptera: Apoidea) de la región Praderas de Tokio-El Potosí - Sierra Mazmorras - Cañón de Iturbide, Coahuila y Nuevo León, México. Version 1.17. Comisión nacional para el conocimiento y uso de la biodiversidad. Occurrence dataset https://doi.org/10.15468/xf37pd accessed via GBIF.org on 2026-03-10.
Zhuang V (2025). UTEP Insects (Arctos). Version 1.99. University of Texas at El Paso Biodiversity Collections. Occurrence dataset https://doi.org/10.15468/cfddho accessed via GBIF.org on 2026-03-10.
Northern Arizona University (2026a). Northern Arizona University - Arthropod Collection. Occurrence dataset https://doi.org/10.15468/du1hci accessed via GBIF.org on 2026-03-10.
University of Arizona Insect Collection (2026b). University of Arizona Insect Collection RL Minckley Insect Collection. Occurrence dataset https://doi.org/10.15468/sj3uvh accessed via GBIF.org on 2026-03-10.
Archbold Biological Station (2023). Archbold Biological Station Arthropod Collection. Occurrence dataset https://doi.org/10.15468/mwyr3t accessed via GBIF.org on 2026-03-10.
Soon V. University of Tartu Natural History Museum and Botanical Garden Zooloogical Collections (n.d.). University of Tartu, Natural History Museum and Botanical Garden. Occurrence dataset https://doi.org/10.15468/6hfnux accessed via GBIF.org on 2026-03-10.
New Mexico State Collection of Arthropods (2026). New Mexico State Collection of Arthropods. Occurrence dataset https://doi.org/10.15468/nn3cel accessed via GBIF.org on 2026-03-10.
Pozo de la Tijera M D C, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Fortalecimiento de las colecciones de ECOSUR. Primera fase (Abejas). Version 1.10. Comisión nacional para el conocimiento y uso de la biodiversidad. Occurrence dataset https://doi.org/10.15468/ydeiae accessed via GBIF.org on 2026-03-10.
Kelly S, Dal Pos D, Sharanowski B (2026). University of Central Florida Collection of Arthropods (UCFC). University of Central Florida Collection of Arthropods (UCFC). Occurrence dataset https://doi.org/10.15468/kyulwg accessed via GBIF.org on 2026-03-10.
Clemson University Arthropod Collection (2026). Clemson University Arthropod Collection. Occurrence dataset https://doi.org/10.15468/fvigdc accessed via GBIF.org on 2026-03-10.
Purdue Entomological Research Collection (2023). The Purdue Entomological Research Collection. Occurrence dataset https://doi.org/10.15468/hyexfq accessed via GBIF.org on 2026-03-10.
North Carolina State University Insect Collection (2024). North Carolina State University Insect Collection. Occurrence dataset https://doi.org/10.15468/3ck2nw accessed via GBIF.org on 2026-03-10.
Eardley C, Ranwashe F (2017). ARC-PPRI: Collection of Southern African bees (1800-2007). Version 1.1. South African National Biodiversity Institute. Occurrence dataset https://doi.org/10.15468/itdvb2 accessed via GBIF.org on 2026-03-10.
Sánchez Cordero Dávila V, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Digitalización y Sistematización de las Colecciones Biológicas Nacionales del Instituto de Biología, UNAM (CNIN). Version 1.7. Comisión nacional para el conocimiento y uso de la biodiversidad. Occurrence dataset https://doi.org/10.15468/gy3jna accessed via GBIF.org on 2026-03-10.
European Bioinformatics Institute (EMBL-EBI), GBIF Helpdesk (2025). INSDC Sequences. Version 1.168. European Nucleotide Archive (EMBL-EBI). Occurrence dataset https://doi.org/10.15468/sbmztx accessed via GBIF.org on 2026-03-10.
A.J. Cook Arthropod Research Collection (2026). The Albert J. Cook Arthropod Research Collection. Occurrence dataset https://doi.org/10.15468/mtagq6 accessed via GBIF.org on 2026-03-10.
Hughes H, Parys K A, Huntzinger K T, Lankford C, Fortuin C, Davis J B (2025). Bees (Hymenoptera: Anthophila) of seasonal herbaceous wetlands in Mississippi. Version 2.1. Specimen Micropublications. Occurrence dataset https://doi.org/10.5886/wv0tub accessed via GBIF.org on 2026-03-10.
Hardy S (2026). Vermont Wild Bee Survey: Miscellaneous Records. Version 1.2. Vermont Center for Ecostudies. Occurrence dataset https://doi.org/10.15468/9ek5m8 accessed via GBIF.org on 2026-03-10.
López-Uribe Lab (2024). López-Uribe Lab Collection. Occurrence dataset https://doi.org/10.15468/fyyzub accessed via GBIF.org on 2026-03-10.
Hardy S, Sharp N, Sefchick J, Veit M (2023). Native Bee Inventory of Missisquoi National Wildlife Refuge. Version 1.9. Vermont Center for Ecostudies. Sampling event dataset https://doi.org/10.15468/cv5uwp accessed via GBIF.org on 2026-03-10.
Hardy S, McFarland K, Sharp N, Milam J, Veit M, Richardson L, Droege S (2025). Vermont Wild Bee Survey (2019-2021). Version 1.19. Vermont Center for Ecostudies. Sampling event dataset https://doi.org/10.15468/yjw5fk accessed via GBIF.org on 2026-03-10.
Ricketts T, Mazurowski J, Hardy S, Nicholson C, Zawawi A (2024). Long-term Bee Monitoring in Intervale Area in Burlington, Vermont, USA. Version 1.2. Vermont Center for Ecostudies. Occurrence dataset https://doi.org/10.15468/fvvhru accessed via GBIF.org on 2026-03-10.
Mazurowski J, Hardy S (2024). Surveys of the bee (Hymenoptera: Apiformes) community in and around solar installations. Version 1.10. Vermont Center for Ecostudies. Sampling event dataset https://doi.org/10.15468/y3y1nb accessed via GBIF.org on 2026-03-10.
Jacobson M, Schummer M, Fierke M, Leopold D (2024). Pollinating insects of restored, managed freshwater marshes in central New York, USA.. Version 1.6. Vermont Center for Ecostudies. Sampling event dataset https://doi.org/10.15468/wy9hgh accessed via GBIF.org on 2026-03-10.
Connecticut Agricultural Experiment Station (2026). Connecticut Agricultural Experiment Station Arthropod Collection. Occurrence dataset https://doi.org/10.15468/nvkb6d accessed via GBIF.org on 2026-03-10.
South Dakota State University, Severin-McDaniel Insect Research Collection (2023). South Dakota State University, The Severin-McDaniel Insect Research Collection. Occurrence dataset https://doi.org/10.15468/hl5tkx accessed via GBIF.org on 2026-03-10.
István M (2026). University of New Hampshire Collection of Insects and Arthropods (UNHC-UNHC). University of New Hamsphire Collection of Insects and other Arthropods. Occurrence dataset https://doi.org/10.15468/n36jmy accessed via GBIF.org on 2026-03-10.
Telfer A (2019). Centre for Biodiversity Genomics - Canadian Specimens. Version 1.5. University of Guelph. Occurrence dataset https://doi.org/10.15468/mbwnw9 accessed via GBIF.org on 2026-03-10.
New Brunswick Museum (2025). New Brunswick Museum - Insects. Occurrence dataset https://doi.org/10.15468/d8dv3a accessed via GBIF.org on 2026-03-10.
Paiero S, Paiero S (2017). University of Guelph Insect Collection (DEBU). Version 2.2. University of Guelph. Occurrence dataset https://doi.org/10.5886/4pp2vt5a accessed via GBIF.org on 2026-03-10.
Vermont Agricultural and Environmental Laboratory (2026). State of Vermont Forest Biology Lab. Occurrence dataset https://doi.org/10.15468/s3gj9m accessed via GBIF.org on 2026-03-10.
Richardson L, Hardy S (2025). Leif Richardson Vermont Bee Specimens. Version 1.12. Vermont Center for Ecostudies. Occurrence dataset https://doi.org/10.15468/nddf7z accessed via GBIF.org on 2026-03-10.
Harvard University M, Morris P J (2026). Museum of Comparative Zoology, Harvard University. Version 162.502. Museum of Comparative Zoology, Harvard University. Occurrence dataset https://doi.org/10.15468/p5rupv accessed via GBIF.org on 2026-03-10.
McKenna M, Zitani N (2025). Zoological Collections, Bees of North America. Version 2.4. University of Western Ontario. Occurrence dataset https://doi.org/10.5886/kofv27 accessed via GBIF.org on 2026-03-10.
Hardy S (2025). Middlebury College Insect Collection: Bees (Anthophila). Version 1.7. Vermont Center for Ecostudies. Occurrence dataset https://doi.org/10.15468/t8dlqy accessed via GBIF.org on 2026-03-10.
Roberts D (2025). CHAS Entomology Collection (Arctos). Version 13.102. Chicago Academy of Sciences. Occurrence dataset https://doi.org/10.15468/i5oupp accessed via GBIF.org on 2026-03-10.
Natural History Museum (2026). Natural History Museum (London) Collection Specimens. Occurrence dataset https://doi.org/10.5519/qd.4fmbrm00 accessed via GBIF.org on 2026-03-10.
Hardy S, McFarland K (2023). Miscellaneous Historical Bee Records from Vermont, USA. Vermont Center for Ecostudies. Occurrence dataset https://doi.org/10.15468/z43gqg accessed via GBIF.org on 2026-03-10.
York University (n.d.a). Bees of Canada / Gschwendtner property collection. Version 1.0. Occurrence dataset https://doi.org/10.15468/i6absz accessed via GBIF.org on 2026-03-10.
York University (n.d.b). Morandin PhD Thesis / La Crete, Alberta. Version 1.0. Occurrence dataset https://doi.org/10.15468/0bse1m accessed via GBIF.org on 2026-03-10.
York University (n.d.c). Bees of Canada / Joker's Hill collection. Version 1.0. Occurrence dataset https://doi.org/10.15468/0zftjm accessed via GBIF.org on 2026-03-10.
Orrell T, Informatics and Data Science Center, Digital Stewardship Team (2026b). NMNH Material Samples (USNM). Version 1.87. National Museum of Natural History, Smithsonian Institution. Occurrence dataset https://doi.org/10.15468/jb9tdf accessed via GBIF.org on 2026-03-11.
Santa Barbara Museum of Natural History (2026). Santa Barbara Museum of Natural History Entomology Collection. Occurrence dataset https://doi.org/10.15468/74wfv6 accessed via GBIF.org on 2026-03-11.
University of California, Davis (2026). R. M. Bohart Museum of Entomology. Occurrence dataset https://doi.org/10.15468/bvzxln accessed via GBIF.org on 2026-03-11.
Fägerström C (2026). Lund University Biological Museum - Insect collections Inventory. Version 367.883. Lund University Biological Museum. Occurrence dataset https://doi.org/10.15468/dahk2a accessed via GBIF.org on 2026-03-11.
Native Bee Society of BC (2026). Native Bee Society of BC Bee Atlas Collection. Occurrence dataset https://doi.org/10.15468/aqkjev accessed via GBIF.org on 2026-03-11.
Prescott D, Wells M, Best L (2023). Survey of wild bees in central Alberta – 2018. Alberta Environment and Parks - Species at Risk program. Occurrence dataset https://doi.org/10.5886/gjdrpe accessed via GBIF.org on 2026-03-11.
Sheffiled C (2019). The bees of British Columbia (Hymenoptera: Apoidea, Apiformes) - Occurrences Dataset. Royal Saskatchewan Museum. Occurrence dataset https://doi.org/10.5886/inga8z accessed via GBIF.org on 2026-03-11.
Northern Arizona University (2026b). Northern Arizona University - Walnut Canyon National Monument Collection. Occurrence dataset https://doi.org/10.15468/hfnekq accessed via GBIF.org on 2026-03-11.
Luis Martínez M A, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Computarización de la colección de abejas (Hymenoptera: Apoidea) del Museo de Zoología Alfonso L. Herrera, de la Facultad de Ciencias de la UNAM. Comisión nacional para el conocimiento y uso de la biodiversidad. Occurrence dataset https://doi.org/10.15468/wzfnf2 accessed via GBIF.org on 2026-03-11.
Vergara Briceño C H, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Actualización de la base de datos del proyecto H278 Apoidea (Hymenoptera) del Valle de Zapotitlán de las Salinas, Puebla. Comisión nacional para el conocimiento y uso de la biodiversidad. Occurrence dataset https://doi.org/10.15468/wv3xpz accessed via GBIF.org on 2026-03-11.
Navarro Sigüenza A G, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Apoyo a las colecciones biológicas de la Facultad de Ciencias de la UNAM: Fase 1 (Abejas). Version 1.10. Comisión nacional para el conocimiento y uso de la biodiversidad. Occurrence dataset https://doi.org/10.15468/gucqr3 accessed via GBIF.org on 2026-03-11.
Romero Nápoles J, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Catálogo de insectos de la colección del Centro de Entomología. Comisión nacional para el conocimiento y uso de la biodiversidad. Occurrence dataset https://doi.org/10.15468/ksnkfh accessed via GBIF.org on 2026-03-11.
Dataset Licenses
Each dataset liscence is structured as follows: (1) in-text citation, (2) dataset title, (3) short-hand liscence.
Graham M, Best L, Moldenke A (2021). Digital record of specimens, including voucher material, from the study of a pollinator habitat restoration site under a commercial solar array in Jackson County, Oregon, 2019. (CC BY-NC 4.0)
Best L (2026). Oregon Bee Atlas. Oregon State University. (CC BY-NC 4.0)
Frost Entomological Museum (2026). Frost Entomological Museum. (CC0 1.0)
MT James Entomological Collection, Washington State University (2026). Washington State University Collection. (CC BY-NC 4.0)
Best L, Engler J, Feuerborn C, Larsen J, Lindh B, Kincaid S, Melathopoulos A, Marshall C J, Marshall C (2022a). Oregon Bee Atlas Survey Data: 2019. Version 1.3. (CC BY-NC 4.0)
Best L, Feuerborn C, Holt J, Kincaid S, Marshall C, Melathopoulos A (2021a). Oregon Bee Atlas Survey Data: 2018. Version 1.5. (CC BY 4.0)
Gibbs J (2025). J. B. Wallis / R. E. Roughley Museum of Entomology. Version 1.2. (CC BY 4.0)
Droege S, Maffei C (2025). Insect Species Occurrence Data from Multiple Projects Worldwide with Focus on Bees and Wasps in North America. Version 1.27. (CC0 1.0)
Ikerd H, Engler J (2023). Bee Fauna of National Wildlife Refuges in the Pacific Northwest, 2010-2016. (CC BY 4.0)
The International Barcode of Life Consortium (2026). International Barcode of Life project (iBOL). (CC BY 4.0)
Colorado State University, C.P. Gillette Museum of Arthropod Diversity (2026). C.P. Gillette Museum of Arthropod Diversity. (CC0 1.0)
Motz G (2026). Entomology Division, Yale Peabody Museum. Yale University Peabody Museum. (CC0 1.0)
Bentley A, Osborn R (2026). Snow Entomological Museum Collection. (CC BY 4.0)
Johnson N (2026). Triplehorn Insect Collection, The Ohio State University. (CC BY 4.0)
Illinois Natural History Survey (2026). Illinois Natural History Survey Insect Collection. (CC BY 4.0)
Gross J, Oboyski P (2026). Essig Museum of Entomology. Version 121.432. (CC BY 4.0)
Ikerd H (2019). Bee Biology and Systematics Laboratory. (CC BY 4.0)
Johnson C (2020). hymenoptera. Version 1.3. (CC BY 4.0)
Grant S, Webbink K, Turcatel M, Shuman R (2025). Field Museum of Natural History (Zoology) Insect, Arachnid and Myriapod Collection. (CC0 1.0)
Orrell T, Informatics and Data Science Center - Digital Stewardship (2026a). NMNH Extant Specimen Records (USNM, US). Version 1.106. (CC0 1.0)
San Diego Natural History Museum (2026). San Diego Natural History Museum Entomology Department. (CC0 1.0)
Texas A&M University Insect Collection (2023). Texas A&M University Insect Collection. (CC BY-NC 4.0)
University of Minnesota Insect Collection (2026). University of Minnesota Insect Collection. (CC0 1.0)
Grinter C, Diaz-Bastin R, Fong J (2026). CAS Entomology Type (TYPE). Version 1.332. (CC0 1.0)
Oram R (2026). Royal Saskatchewan Museum - Entomology. Version 2.2. (CC BY 4.0)
Scott V (2025). UCM Entomology Collection. Version 6.6. (CC BY-NC 4.0)
Museum of Southwestern Biology (2026). Museum of Southwestern Biology Division of Arthropods. (CC BY-NC 4.0)
Kenneth S. Norris Center for Natural History (2026). Kenneth S. Norris Center for Natural History, University of California Santa Cruz, Insect Collection. (CC BY-NC 4.0)
Carril O, Wilson J, Griswold T, Ikerd H I (2023). Wild bees of Grand Staircase-Escalante National Monument. (CC BY-NC 4.0)
University of Arizona Insect Collection (2026). University of Arizona Insect Collection. (CC0 1.0)
Brigham Young University, Arthropod Collection (2026). Brigham Young University Arthropod Museum. (CC BY-NC 4.0)
Mertz W, Kung G, Xie W (2026). LACM Entomology Collection. Version 5.33. (CC0 1.0)
Mississippi Entomological Museum (2026). Mississippi Entomological Museum. (CC0 1.0)
Cheadle Center for Biodiversity and Ecological Restoration (2026a). UCSD Holway Lab - Longterm Habitat Fragmentation Study. (CC BY-NC 4.0)
Cheadle Center for Biodiversity and Ecological Restoration (2026b). University of California Santa Barbara Invertebrate Zoology Collection. (CC0 1.0)
Luther College (2026). Luther Entomological Research Collection. (CC BY-NC 4.0)
UMMZ Insects Division Data Group, LSA IT A (2026). University of Michigan Museum of Zoology, Division of Insects. Version 1.107. (CC BY-NC 4.0)
Wild A (2026). University of Texas, Biodiversity Center, Entomology Collection (UTIC). Version 1.304. (CC0 1.0)
Natural Science Research Laboratory, Museum of Texas Tech University (TTU) (2026). Museum at Texas Tech University Invertebrate Zoology Collection. (CC BY-NC 4.0)
University of Kentucky (2026). University of Kentucky Insect Collection. (CC BY-NC 4.0)
Veit M, McFarland K (2024). Bee Specimens from Michael Veit Collection. Version 1.3. (CC BY-NC 4.0)
Holm M, Houle M, Salem M (2025). U.S. Fish and Wildlife Service Great Lakes Native Bee Survey. Version 1.14. (CC0 1.0)
Florida State Collection of Arthropods (2026). Florida State Collection of Arthropods. (CC0 1.0)
Virginia Tech Insect Collection (2026). Virginia Polytechnic Institute and State University Insect Collection. (CC0 1.0)
Johnson N (2019). Cleveland Museum of Natural History. (CC BY 4.0)
Quiroz Martínez H, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Abejas silvestres (Hymenoptera: Apoidea) de la región Praderas de Tokio-El Potosí - Sierra Mazmorras - Cañón de Iturbide, Coahuila y Nuevo León, México. Version 1.17. (CC BY 4.0)
Zhuang V (2025). UTEP Insects (Arctos). Version 1.99. (CC0 1.0)
Northern Arizona University (2026a). Northern Arizona University - Arthropod Collection. (CC0 1.0)
University of Arizona Insect Collection (2026b). University of Arizona Insect Collection RL Minckley Insect Collection. (CC0 1.0)
Archbold Biological Station (2023). Archbold Biological Station Arthropod Collection. (CC BY-NC 4.0)
Soon V. University of Tartu Natural History Museum and Botanical Garden Zooloogical Collections (n.d.). University of Tartu, Natural History Museum and Botanical Garden. (CC BY-NC 4.0)
New Mexico State Collection of Arthropods (2026). New Mexico State Collection of Arthropods. (CC0 1.0)
Pozo de la Tijera M D C, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Fortalecimiento de las colecciones de ECOSUR. (CC BY 4.0)
Kelly S, Dal Pos D, Sharanowski B (2026). University of Central Florida Collection of Arthropods (UCFC). (CC BY 4.0)
Clemson University Arthropod Collection (2026). Clemson University Arthropod Collection. (CC0 1.0)
Purdue Entomological Research Collection (2023). The Purdue Entomological Research Collection. (CC0 1.0)
North Carolina State University Insect Collection (2024). North Carolina State University Insect Collection. (CC BY-NC 4.0)
Eardley C, Ranwashe F (2017). ARC-PPRI: Collection of Southern African bees (1800-2007). Version 1.1. (CC BY 4.0)
Sánchez Cordero Dávila V, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Digitalización y Sistematización de las Colecciones Biológicas Nacionales del Instituto de Biología, UNAM (CNIN). Version 1.7. (CC BY 4.0)
European Bioinformatics Institute (EMBL-EBI), GBIF Helpdesk (2025). INSDC Sequences. Version 1.168. (CC BY 4.0)
A.J. Cook Arthropod Research Collection (2026). The Albert J. Cook Arthropod Research Collection. (CC0 1.0)
Hughes H, Parys K A, Huntzinger K T, Lankford C, Fortuin C, Davis J B (2025). Bees (Hymenoptera: Anthophila) of seasonal herbaceous wetlands in Mississippi. Version 2.1. (CC0 1.0)
Hardy S (2026). Vermont Wild Bee Survey: Miscellaneous Records. Version 1.2. (CC BY 4.0)
Hardy S, McFarland K, Sharp N, Milam J, Veit M, Richardson L, Droege S (2025). Vermont Wild Bee Survey (2019-2021). Version 1.19. (CC BY 4.0)
Ricketts T, Mazurowski J, Hardy S, Nicholson C, Zawawi A (2024). Long-term Bee Monitoring in Intervale Area in Burlington, Vermont, USA. Version 1.2. (CC BY 4.0)
Mazurowski J, Hardy S (2024). Surveys of the bee (Hymenoptera: Apiformes) community in and around solar installations. Version 1.10. (CC BY-NC 4.0)
Jacobson M, Schummer M, Fierke M, Leopold D (2024). Pollinating insects of restored, managed freshwater marshes in central New York, USA.. Version 1.6. (CC BY 4.0)
Connecticut Agricultural Experiment Station (2026). Connecticut Agricultural Experiment Station Arthropod Collection. (CC BY-NC 4.0)
South Dakota State University, Severin-McDaniel Insect Research Collection (2023). South Dakota State University, The Severin-McDaniel Insect Research Collection. (CC0 1.0)
István M (2026). University of New Hampshire Collection of Insects and Arthropods (UNHC-UNHC). (CC BY 4.0)
Telfer A (2019). Centre for Biodiversity Genomics - Canadian Specimens. Version 1.5. (CC0 1.0)
New Brunswick Museum (2025). New Brunswick Museum - Insects. (CC BY-NC 4.0)
Paiero S, Paiero S (2017). University of Guelph Insect Collection (DEBU). Version 2.2. (CC0 1.0)
Vermont Agricultural and Environmental Laboratory (2026). State of Vermont Forest Biology Lab. (CC BY-NC 4.0)
Richardson L, Hardy S (2025). Leif Richardson Vermont Bee Specimens. Version 1.12. (CC BY-NC 4.0)
Harvard University M, Morris P J (2026). Museum of Comparative Zoology, Harvard University. Version 162.502. (CC BY-NC 4.0)
McKenna M, Zitani N (2025). Zoological Collections, Bees of North America. Version 2.4. (CC BY 4.0)
Hardy S (2025). Middlebury College Insect Collection: Bees (Anthophila). Version 1.7. (CC BY-NC 4.0)
Roberts D (2025). CHAS Entomology Collection (Arctos). Version 13.102. (CC0 1.0)
Natural History Museum (2026). Natural History Museum (London) Collection Specimens. (CC0 1.0)
Hardy S, McFarland K (2023). Miscellaneous Historical Bee Records from Vermont, USA. Vermont Center for Ecostudies. (CC0 1.0)
York University (n.d.a). Bees of Canada / Gschwendtner property collection. Version 1.0. (CC BY 4.0)
York University (n.d.b). Morandin PhD Thesis / La Crete, Alberta. Version 1.0. (CC BY 4.0)
York University (n.d.c). Bees of Canada / Joker's Hill collection. Version 1.0. (CC BY 4.0)
Orrell T, Informatics and Data Science Center, Digital Stewardship Team (2026b). NMNH Material Samples (USNM). Version 1.87. (CC0 1.0)
Santa Barbara Museum of Natural History (2026). Santa Barbara Museum of Natural History Entomology Collection. (CC0 1.0)
University of California, Davis (2026). R. M. Bohart Museum of Entomology. (CC BY-NC 4.0)
Fägerström C (2026). Lund University Biological Museum - Insect collections Inventory. Version 367.883. (CC BY-NC 4.0)
Native Bee Society of BC (2026). Native Bee Society of BC Bee Atlas Collection. (CC BY-NC 4.0)
Prescott D, Wells M, Best L (2023). Survey of wild bees in central Alberta – 2018. (CC BY 4.0)
Sheffiled C (2019). The bees of British Columbia (Hymenoptera: Apoidea, Apiformes) - Occurrences Dataset. (CC BY 4.0)
Northern Arizona University (2026b). Northern Arizona University - Walnut Canyon National Monument Collection. (CC0 1.0)
Luis Martínez M A, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Computarización de la colección de abejas (Hymenoptera: Apoidea) del Museo de Zoología Alfonso L. Herrera, de la Facultad de Ciencias de la UNAM. (CC BY 4.0)
Vergara Briceño C H, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Actualización de la base de datos del proyecto H278 Apoidea (Hymenoptera) del Valle de Zapotitlán de las Salinas, Puebla. (CC BY 4.0)
Navarro Sigüenza A G, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Apoyo a las colecciones biológicas de la Facultad de Ciencias de la UNAM: Fase 1 (Abejas). Version 1.10. (CC BY 4.0)
Romero Nápoles J, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Catálogo de insectos de la colección del Centro de Entomología. (CC BY 4.0)
Glossary
Pedicel: the second segment of an antennae, in Melissodes, these are often very short.
Flagellum/flagellomeres: all antennal segments except for the scape and pedicel.
Flagellar segment: one of the segments of the flagellum.
Sclerite (something having the property of a sclerite, sclerotized): a hardened pale of an insect, referring to a portion of the exoskeleton.
Clypeus (plural, clypei; in reference to, clypeal): a large facial sclerite that is somewhat equivalent to the beginning of the nose to the beginning of the lip area on humans.
Labrum (in reference to, labral): a medium sized facial sclerite that is smaller than the clypeus and often tucked under the clypeus unless the galeae are extended.
Galea (plural, galeae; in reference to, galeal): the outer covering of the tongue.
Oculoclypeal distance: the distance between the lateral area of the clypeus and the inner margin of the eye just above the mandibles.
Supraclypeal area: the facial sclerite located right above the clypeus.
Vertex: the top portion of the head between the apices of the compound eyes.
Postocular area: the exoskeletal area behind and lateral to the compound eyes (genal area).
Mesoscutum (in reference to, mesoscutal): the large dorsal thoracic plate; when looking at the dorsum of the thorax, this is the anterior plate.
Scutellum (in reference to, scutellar): the medium to smaller sized dorsal thoracic plate; when looking at the dorsum of the thorax, this is the posterior plate.
Metanotum (in reference to, metanotal): the small dorsal thoracic plate located just posteriorly to the scutellum, but anteriorly to the dorsum of the propodeum.
Tegula (plural, tegulae; in reference to, tegular): a small sclerite covering the base of the forewing.
Pronotum (in reference to, pronotal): the anterior most thoracic segment.
Mesepisternum (plural, mesepisterna; in reference to, mesepisternal): the lateral portions of the thorax not including the lateral areas of the propodeum or pronotum.
Mesopleura (in reference to, mesopleural): the lateral area of the thorax including the lateral areas of the propodeum and pronotum.
Mesonotum (in reference to, mesonotal): entirety of the dorsum of the thorax.
Metasoma (in reference to, metasomal): the posterior most body segment (abdomen).
Tergum (plural, terga; in reference to, tergal): the dorsal surface of the metasoma, however, used as meaning each segment or tergite and reference to the dorsum of the metasoma will be stated clearly.
Sternum (plural, sterna; in reference to, sternal): a ventral metasomal segment (i.e. the “other side” of a tergum).
Pygidial plate: a small sclerotized plate on the posterior portion of the metasoma usually forming a sort of pointed process.
Tibia (plural, tibiae; in reference to, tibial): the second major segment of the legs.
Basitibial plate: a small sclerotized plate located at the base of a tibia somewhat resembling a kneecap.
Tarsus (plural, tarsi; in reference to, tarsal): the third major segment of the legs composed of the bastitarsus, mediotarsus and distitarsi. However, the bastitarsus is referred to separately from the rest of the tarsi in this guide.
Bastitarsus (plural, bastitarsi; in reference to, bastitarsal): the largest and basal most tarsal segment.
Scopa (plural, scopae; in reference to, scopal): large pollen collecting hairs. On female Melissodes, these are on the hind tibiae and bastitarsi.