Melissodes communis

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes communis
Melissodes communis, the common long-horned bee, is likely one of the most common and wide-spread species of Melissodes (similar in range and commonality to that of M. agilis), stretching across the majority of the United States and Mexico (Fig. 23). Like all Melissodes, male M. communis have long antennae, and the females have short antennae in comparison (see "Genus" page for more information). This polytypic species resides in the subgenus Melissodes Latreille, containing two distinct subspecies, M. c. communis and M. c. alopex. Both sexes, and subspecies, resemble a few other Melissodes in their subgenus; specifically M. comptoides in eastern portions of its range and gilensis in the western areas. Females are highly variable (as can be seen in Laberge’s 1956 key), ranging widely, and most notably, in thoracic and head vestiture (among other characters). Males of this species are distinctive in the distal pale band of the second tergum being complete (only medially interrupted in specimens that are worn, but band can be identified via the presence of puncture from which the hairs originally arose); the first flagellar segment’s minimum length often being equal to, or slightly longer than, one-sixth of the second segment’s maximum length (not always); the apical areas of the second and third terga being either impunctate or only bearing minute punctures that are less than twice the width of the hairs that arise from them; the distal pale band of the fourth tergum being distinctly narrower than four times the medial portion of the apical area; the distal pale band of the second tergum often being as narrow as, or narrower than, half the apical area’s width, although if wider (approximately of similar size to the apical area), then the apical portions of the terga are hyaline; the band of the fifth tergum often being pale and medially interrupted or absent, and the scutellum and mesoscutum often bearing many dark hairs (Laberge, 1956). Females are much more variable than their male counterparts with some individuals having entirely dark thoracic hairs, some bearing these dark hairs on the ventral mesepisternal surfaces (often on the lower-lateral surfaces as well), and some having these hairs entirely pale apart from the large dark mesoscutal patch and the dark scutellar patch (Laberge, 1956). Due to the wide variation females present, a more in-depth description will be given below that lists distinctive traits (see “Description and Identification” for more information on both sexes). Similarly to many other species of the subgenus Melissodes, M. communis is polylectic with no significant known floral preference (Laberge, 1956; Wright, 2018). It is, however, interesting that from the datasets contributing the known floral data of M. communis (24), the general species-level collections seem to occur most predominantly on Asteraceae while subspecific data shows neither subspecies has an abundance of collections from this floral family (a discrepancy between total specific identifications and total subspecific identifications; see “Flower Records” below for more information). Given the presence of two subspecies, two subspecific treatments will be given below following the general specific description.
Description and Identification
Based on Laberge's (1956) description, Melissodes communis are setacouse bees that greatly range in size but tend to be somewhat larger in general. Females range from 12 to 16 millimeters in length and 4.0 to 6 millimeters in width (width measured at the widest portion of the metasoma). Males are a bit smaller, being about 10 to 15 millimeters in length and 3.5 to 5.5 millimeters in width (width measured at the widest portion of the metasoma). The female's first flagellar segment is on average 2.01 times the size of the second flagellar segment (standard deviation 0.019). The males are the opposite where the second flagellar segment is on average 4.80 times the size of the first flagellar segment (standard deviation 0.115). Female wing length is 4.57 millimeters on average (standard deviation 0.384 millimeters), and male wing length is 4.18 millimeters on average (standard deviation 0.371 millimeters). Females have an average of 16.97 hamuli (standard deviation 0.285), while males have an average of 15.05 (standard deviation 0.328).
Female
Females can be separated from other M. (Melissodes) in three groups: 1) the head hairs and thoracic hairs are almost entirely black, apart from a few pale hairs between the mesoscutum and scutellum, and on the fossae of the antennae; 2) the thoracic hairs are largely pale with a dark dorsal thoracic patch and dark brown hairs ventrally on the mesepisterna; 3) the thoracic hairs are mostly pale apart from the dorsal thoracic patch (mesepisternal hairs all pale). The following lists of the foregoing M. communis female groups are characters that define this species. The first group is distinctive in that the distal pale band of the second tergum is narrow, complete, and white; the band on the fourth tergum is mostly white, often with a medial interruption of dark hairs, and reaches the apex across the entire tergum; the distal band of the third tergum is white to pale ochraceous (not dark); the scopal hairs are pale ochraceous to yellow with the inner surfaces of the hind basitarsi bearing dark red to orange hairs; and the apical areas of the second and third terga often bearing more than, or equal to, six semi-indistinct, irregular rows of simple, short dark brown hairs. The second and third groups are distinctive the following characters until listed otherwise: the hairs on the pronotal lobes are entirely pale; the majority of the scopal hairs are white to yellow but the inner surfaces of the hind basitarsi are bright red; the apical areas of the second and third terga are often entirely impunctate or if punctate, then only bearing punctures that are less than twice the width of the hairs that arise from them; the mesoscutum is shiny and unshagreened, or if so, then only delicately, and bears large, round, deep punctures that are separated by one puncture diameter or less in the lateral and anterior areas; the distal pale band of the second tergum, when unworn, is complete; the basal band of the second tergum is composed of typical plumose hairs, lacking spatulate hairs apically. The second group is distinctive in that the mesepisterna have dark ventral hairs; the distal pale band of the second tergum is distinctly much narrower than the interband zone throughout the majority of the tergum; the clypeus noticeably protrudes from the face in profile view, its medial length from this angle measuring faintly longer than its width; the distal white band of the second tergum often arches evenly across the entire tergum and is usually less than, or equal to one-third of the apical area’s width; the interband zone of the second tergum is either medially impunctate, or if punctate, then bearing shallow minute indistinct punctures that are often obscured due to dense shagreening. The third group is distinctive in that the mesepisterna bear no dark hairs (including the ventral areas); the pronotal lobes and anterior tegular hairs bear no dark hairs; the pale hairs on the mesoscutum and scutellum are white to ochraceous, sometimes will pale rufescence or ferruginous colors anteriorly, or if bearing orange hairs (see the Mexican M. c. communis variation records below), then the distal pale band of the second tergum is narrower than the interband zone across the entire tergum; the mesoscutum bears a large patch of dark hairs posteromedially; the apical areas of the second and third terga are impunctate and dulled due to fine, be it distinctly dense, shagreening; the interband zone of the second tergum bears small distinct punctures on either side of the lateral one-fourths of the tergum; and the apical margin of the first tergum is translucent to hyaline yellowish in more than, or equal to, the apical one-sixth.
According to Laberge (1956), the base-line description of female M. communis is as follows. The integument is black, differing at the basitarsi, which are reddish brown; the distitarsi, which are often rufescent; the underside flagellar segments 3-10, which are reddish-brown to red; the apical area of the first tergum, which can be narrowly hyaline (though, this area too can be black or only extremely narrowly margined with hyaline colorations); terga 2-4’s apical areas, which are usually reddish brown; and the eyes, which are often green-yellow, and sometimes yellow-brown to gray. The clypeus is moderately shiny to shiny due to either no, or delicate, shagreening and has mostly deep round punctures that are separated by equal to, or less than, one puncture width. The clypeus also usually has its medial carina positioned in the apical half shiny and often protrudes from the face, in profile view, its medial length measuring faintly longer than its width. The supraclypeal area is somewhat shiny, but often dulled due to delicate shagreening and has punctures that are large, round, and minimally present laterally, and usually more. The flattened lateral areas of the vertex that reach relatively posterior from, and positioned medially to, the apical portions of the compound eyes are shiny, either not, or very delicately shagreened, and have noticeable round punctures that are mostly separated by one-half to one puncture diameter. In facial view, the eyes faintly converge near the mandibles and are just about one-third as wide as long. The four maxillary palpal segments are in a ratio of approximately 6:8:5:1, with the second segment the longest, and the fourth segment varying in size (sometimes twice its length). The galeae are shiny with no shagreening, apart from the tips, and have widely spaced punctures with hairs dorsally arising from them. The mesoscutum is shiny with no, or delicate, shagreening (Fig. 1). The lateral, anterior, and posterior sloped areas of the mesoscutum have large, round, deep, crowded punctures, but in the flattened posteromedial area, the punctures become larger and mostly separated by more than one puncture diameter (Fig. 1). The scutellum is shiny and bears punctures of similar size to that of the surrounding mesoscutal area, but these punctures become smaller peripherally and near the midline. The lateral mesapisternal surfaces are shiny, usually delicately shagreened, and have punctures that are of similar size to those on the anterior areas of the mesoscutum, and tend to be separated by less than one puncture diameter. The metanotum is usually shiny and not shagreened on the medial dorsal surface, but becomes dulled due to delicate shagreening on the remaining surfaces, and bears punctures that are small and crowded. In length, the metanotum is medially as long, or longer than, the dorsal propodeal surface. The basal propodeal face is sometimes shiny, often basally reticulopunctate, but the punctures become noticeably separated in the apical areas. The sloping propodeal and lateral surfaces are often dulled due to dense coarse shagreening and have large crowded punctures, apart from the upper impunctate area of the sloping surface.
The first tergum dulled due to dense minute shagreening and the basal one-half to three-fifths bear small shallow punctures that are mostly separated by one-half to two puncture diameters, minimally medially and sometimes more. The apical area of the first tergum is impunctate. The second tergum is densely finely shagreened, has very tiny round punctures positioned beneath the basal band of pubescence, and has small round interband punctures that are mostly separated medially by more than one puncture diameter, becoming somewhat larger and denser laterally (these punctures are sometimes indistinct and often obscured due to dense shagreening). The apical area of the second tergum is either impunctate, or has very tiny apical punctures that are no more than two times the width of the hairs that arise from them (Fig. 2). The third tergum’s apical area is similar to that of the second tergum, apart from the surface, which is instead densely finely shagreened (Fig. 2). The third and fourth terga’s small rounded punctures usually merge into one another laterally in the basal areas, and are medially separated by less than one puncture diameter.
The base-line female setal description is as follows. The head hairs are mostly white to ochraceous apart from the abundant dark brown vertex hairs (these can at times be very sparse or absent entirely). However, on rare occasions, these hairs are almost entirely dark brown with a few pale hairs around the fossae of the antennae. The thoracic hairs are mostly pale colored (usually ochraceous), however, these hairs are often pale rufescent in western individuals and whitish in eastern individuals. The mesoscutum has a large posteromedial patch of dark hairs (these can at times be very sparse or absent entirely, similar to those of the vertex). The scutellar hairs are mostly dark with ochraceous to white hairs on the periphery. However, on rare occasions, these hairs are also almost entirely dark brown with a narrow row of pale hairs fringing the anterior scutellar margin and the posterior mesoscutal margin. The first tergum’s basal two-thirds (or less) bear long pale hairs that reach the apical margin laterally, and the apical area of this tergum is more or less bare, but a few dark brown, appressed, simple hairs are present at extreme lateral areas. The basal pubescent band of the second tergum connects to the distal band at extreme lateral areas and minimally partially consists of basal plumose hairs and narrowly apical spatulate hairs. The interband zone of the second tergum consists of appressed ot suberect, erect black to dark brown hairs. The distal pale band is distinctly thin, approximately more than, or equal to, one-third of the apical area medially. The apical area of the second tergum consists of appressed to suberect, erect black to dark brown hairs (similar to the interband zone). The third tergum’s distal pale band is positioned medially and somewhat denser apically and more diffuse basally. The basal area of the third tergum has many erect to suberect, black to dark brown hairs and the apical area is similar to that of the second tergum. The fourth tergum is similar to that of the second tergum, but the pubescent band is wide, positioned apically, and usually medially interrupted by a small rectangular or triangular notch of suberect dark brown hairs (Fig. 3). The fifth and the sixth terga are both shrouded in appressed, black to dark brown hairs, and the fifth tergum (sometimes the sixth as well) usually has some long pale hairs laterally. The scopae are ochraceous to white, and the hairs on the inner surfaces of the hind tibiae and hind basitarsi are yellow to bright rufescent (Fig. 4).

Fig. 1. A labeled diagram of the mesoscutal microsculpturing and punctation pattern of a typical female M. communis Photo credit: Christopher Wilson (All Rights Reserved).

Fig. 2. A labeled diagram of the microsculpturing and punctation pattern of the apical areas of the second and third terga of a typical female M. communis. Note that the punctures are increadibly small on both terga. Photo credits: Christopher Wilson (All Rights Reserved).
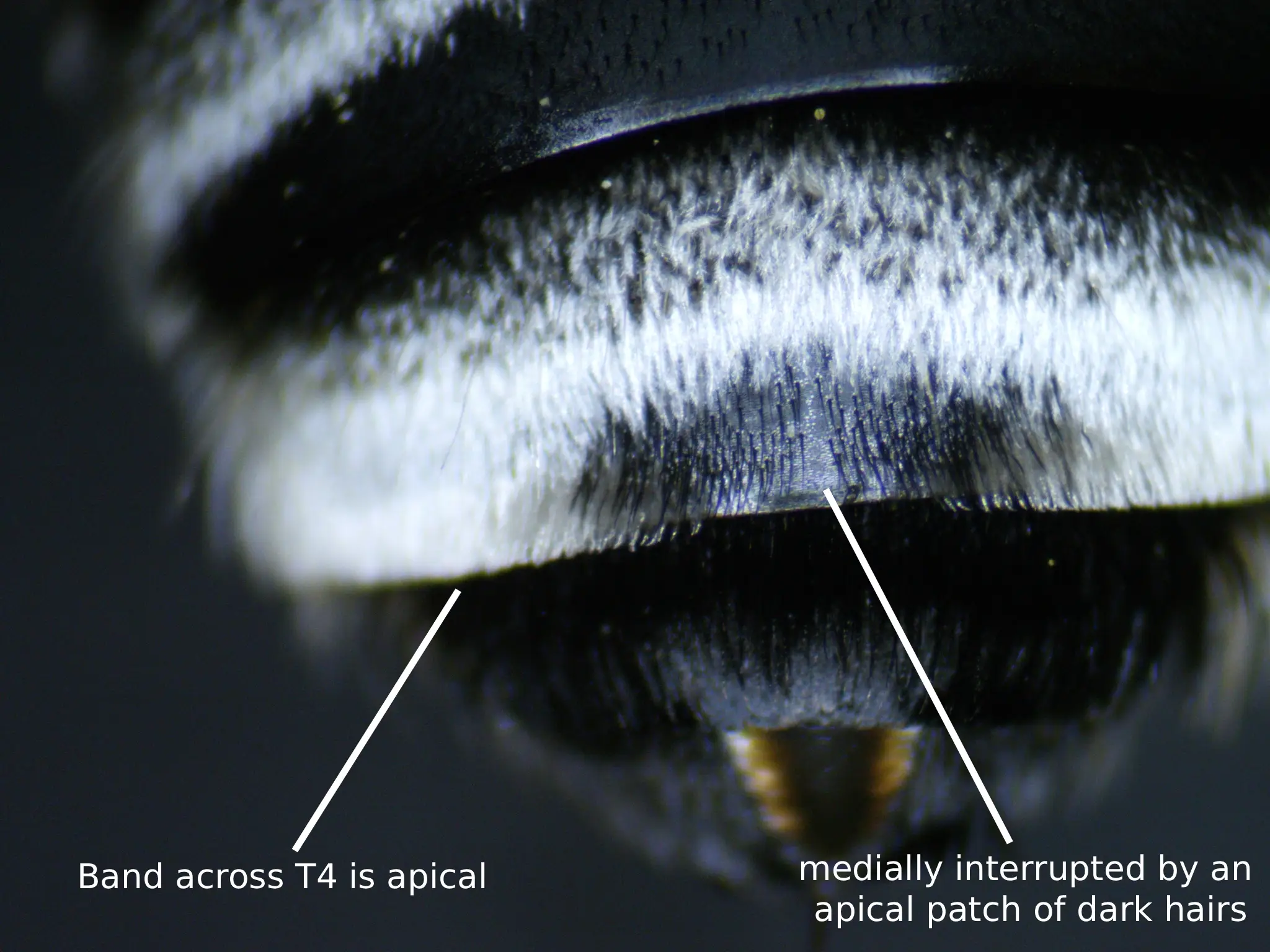
Fig. 3. A labeled diagram of the band across the fourth tergum illustrating the apical nature as well as the dark medial patch of hairs. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 4. A labeled diagram illustrating the light scopal hairs and yellow to reddish hairs on the innner surfaces of the hind basitarsi. Photo credits: Christopher Wilson (All Rights Reserved).
Male
According to Laberge (1956), the base-line description of male M. communis is as follows. The integument is black, differing at the clypeus and labrum, which are bright yellow to pale; the mandibular bases, which have yellow maculations that are minimally wider than the triangular basal depressions and often completely cover the basal half (Fig. 5); the underside of flagellar segments 2-11, which are yellow to red; the eyes, which are greenish-yellow to brownish-gray; the legs which are minimally red on the distitarsi, but usually entirely red; the wing membranes, which are mostly hyaline to very faintly infumate; the wing veins, which are reddish brown; the tegulae, which can be rufescent; and the terga’s apical areas, which range from being translucent, to infumate, to entierly reddish brown or black opaque. The first flagellar segment’s minimum length is about one-sixth of the second segment's maximum length, sometimes less but usually more, and always longer than the length of the pedicel on the same side (Fig. 6). The four maxillary palpal segments are in a ratio of approximately 6:8:5:1, with the second segment the longest and sometimes, be it rarely, having a tiny fifth segment. The remainder of the sculptural characteristics are similar to that of the females except as follows: the clypeal punctures are less coarse; the mesoscutal posteromedial area occasionally has its punctures more crowded; the first tergum’s basal three-fifths or more bear small shallow punctures and the surface is distinctly densely shagreened.
The length of the lateral plate of the seventh sternum is less than twice its width and often less than one-half of the tergum’s length measured from the apex of the apodeme to the plate’s tip. The medial carinae on the lateral plates of the seventh sternum converge, creating a broad V-shaped projection that does not reach the apical area of the sternum medially. The medial plate of the seventh sternum is large (in comparison to those of other species), produced into a semi-rounded hollow one-half to three-fourths cylindrical structure (slightly flattened), and measures, in length, approximately twice its width. The apodeme of the medial plate of the seventh sternum is narrow, rounded to pointed apically, and long. The eighth sternum is apically emarginate, bears several to many long simple hairs apicolaterally (distinctly so on the large rounded apical projections), has a ventral, distinct, longitudinal, medial carina that medially surpasses the apical margin of the sternum, and bears short lateral apodems that narrow to form a small neck-like constriction as well as a small knob-shaped projection that reaches basally. The gonostylus is short in length, bears tufts of long, simple hairs on the outer areas basally that lean distally. The inner surface of the gonocoxite positioned below the gonostylus bears many simple short, some of which are blunt and the others normally pointed (both types intermixed), and the ventral areas below the gonostylus bear many extremely short, simple, normally pointed hairs. The spatha has a small notch across its distal margin in the lateral areas that fits the penis valve’s dorsal lamella. The lateral protrusions of the spatha become somewhat rectangular and shape and reach laterally as well as anteriorly towards the gonobase.
The male base-line setal description is as follows. The vestiture is similar to that of the female, differing at the head, which very rarely has any dark hairs; the mesoscutum, which lacks dark hairs; the scutellum, which sometimes doesn’t have dark hairs; the ventral, anterior, and lateral surfaces of the mesepisterna, which lack dark hairs; the first tergum, which isn’t usually apically bare and often has some appressed dark brown, usually yellow colored medially, hairs across the apical area; the distal pale band of the second tergum is wider than that of the females, often more than, or equal to, one-third of the medial portion of the apical area; the hairs present on the second tergum’s interband zone and apical areas are usually pale, but can be black to dark brown; the fourth tergum’s pubescent band is complete and separated from the apical margin of the tergum similar to the separation from that of the second and third tergum; the fifth tergum has an uninterrupted pale band that reaches the apex across the anterior tergum; the sixth and seventh terga are completely covered in dark hairs apart from the long lateral pale tufts.

Fig. 5. A labeled diagram showing the large basal mandibular macula of a male M. communis. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 6. A labeled diagram illustrating the ratio of the first flagellar segment's minimum length to the second flagellar segment's maximum length, as well as the small side of F1 to the length of the pedicle. Photo credits: Christopher Wilson (All Rights Reserved).
Subspecies
Melissodes communis is comprised of two separate subspecies, a western one (M. c. alopex) located in a range including the following states and provinces: extremely southern British Columbia, Washington, Oregon, California, Arizona, Utah, likely Nevada, likely western portions of Montana, Idaho, and extremely southern Alberta; and an eastern one (M. c. communis), occupying the majority of the eastern and east-central United States (Laberge, 1956). However, following Laberge’s (1956) publication, three M. c. communis identifications (one identified by Dr. K. Wright, one by Dr. W. Laberge, and the other whose identifier wasn’t recorded) much farther westward than its original proposed range have been documented. These records include states such as California (Florida State Collection of Arthropods, 2026; GBIF record 5220126120) and Nevada (Bentley & Osborn, 2026; GBIF record 657848537), suggesting that the hypothesized zone of intergradation from that of Laberge (1956) may be more expansive than originally assumed. The former of the two subspecies seems to be paler as well as less variable in vestiture and integumental characteristics than the latter; the latter containing the only currently published female description of significant melanistic value (Laberge, 1956; see M. c. communis description for more information). Females of these subspecies seem to only differ in vestiture coloration, while males, in addition to vestiture differ in the integumental color of the apical tergal. As a species, M. communis varies largely in size (which can be seen from the measurements above) with the largest documented individuals occurring in Utah, an interesting observation made by Laberge (1956) given that the intergradation zone overlaps this state. While males and females of the current M. communis subspecies are documented and described below, it should be noted that, in general, males are more variable than females in the sense that they follow less of a distinct pattern; hence, subspecific delamination is predominantly placed on female traits with males in general often being less determinative of subspecific presence (Laberge, 1956). The descriptions below are meant to be added to, and override portions of, the baseline M. communis description of both sexes given above. All subspecies descriptions below are derived from Laberge (1956) unless cited otherwise.
M. c. communis
This subspecies of M. communis seems to have the largest range, its current known distribution overlapping the majority of the Eastern United States, dipping down into Mexico, and a small portion of the Southwestern United States as well. As stated above, M. c. communis has the highest variability between the two subspecies, and while not exceedingly difficult to identify from its corresponding taxa (M. c. alopex), the ranges of size and vestiture can lead to some confusion during identification. Due to this, a full additive description will be presented below (see “Female description”) which can be used in tandem with the female table of variable characteristics. Females of M. c. communis can be identified from those of M. c. alopex by the distal pale band of the second tergum being narrower; the pale tergal pubescence of the bands often being white; and the dark hairs on the ventral mesepisternal surfaces. Males differ from those of M. c. alopex primarily in the distal pale band of the first tergum being entirely absent, or at least medially interrupted, and the apical tergal rims being dark-colored opaque to dark smokey translucent.
Female
According to Laberge (1956), the additive description of female M. c. communis is as follows. The first tergum’s apical margin is often entirely opaque black with no hyaline edge, or if a hyaline margin is present, then it’s extremely and distinctly narrow (only sometimes is the apical hyaline margin wider than this; Fig. 7). The wing membranes are a translucent infumate dark brown to yellow color (Fig. 8). In the darkest individuals that are located in the southeastern portions of the United States, the wing veins often range from black to dark red rather than the dark brownish red of M. c. alopex. The supraclypeal areas are usually dulled due to fine shagreening, but sometimes, this area may instead be shiny and unshagreened (although fairly rarely). The upper impunctate triangular area of the propodeal slope is often large, but sometimes this area is smaller and ovular. The small basal punctures of the first tergum are mostly separated by more than, or equal to, one puncture diameter in the medial areas (Fig. 9).
The additive female setal description is as follows. The mandibular and labral hairs are often brown (Fig. 10), as are the hairs on the apical one-half of the clypeus. The dark patch of hairs on the scutellum is sometimes shrunken, composed of only several hairs rather than the entier medial area (Fig. 11), or at times entirely absent (although, both are uncommon, often entire). The thoracic pale hairs are usually ochraceous to pale rufescent, and sometimes white (Fig. 12). Minimally, the ventral mesepisternal hairs are black to dark brown and often the lower areas of the lateral surfaces and anterior faces bear dark hairs as well. The pale hairs of the pubescent bands of the terga are often entirely white, and the medial portion of the distal pale band of the second tergum often measures less than, or equal to, one third of the medial portion of the apical area (Fig. 13). This band, at times, may be faintly wider, but never measures to be one-half of the apical area’s width medially. The distal pale band of the fourth tergum minimally bears a small medial darkened patch of hairs in the apical areas, these hairs often becoming dark brown to black (see Fig. 3). The fifth and sixth terga are often entirely shrouded in dark hair, bearing no pale lateral tufts. The sternal hairs are mostly black to dark brown, often becoming pale laterally to form lateral tufts (Fig. 14). The leg hairs are highly variable, but in general, these hairs are often white to pale ochraceous, differing at the inner surfaces of the hind tibiae and hind basitarsi, which are yellow to red; the fore tarsi, which are dark brownish red; the femora, which are usually dark brown, noticeably and distinctly so on the underside and posterior areas; the inner surfaces of the fore and middle tibiae, which are usually brownish, minimally so apically and sometimes more; and the scopae, which are often ochraceous, but range between ochraceous to pale ochraceous (see Fig. 4).

Fig. 7. A labeled diagram showing the thin hyaline margin on the first tergum of a female M. c. communis. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 8. A labeled diagram showing the color of the wing membranes of a female M. c. communis. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 9. A labeled diagram showing the punctation pattern of the small basal punctures on the first tergum of a female M. c. communis. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 10. A labeled diagram showing the brown labral and mandibular hairs of a female M. c. communis. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 11. A labeled diagram showing the dark scutellar patch of hairs of a female M. c. communis and how somtimes, this patch doesn't engulf the entiere dorsal surface of the scutellum. Note that this character varies as described above in the text. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 12. A labeled diagram showing the color of the pale dorsal thoracic hairs of a female M. c. communis and how these hairs are rather ochraceous to pale rufescent rather than pale ochraceous to white. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 13. A labeled diagram showing the white colorations of the metasomal hair bands of a female M. c. communis and the ratio of the width of the second tergum's distal pale band to the width of the apical area. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 14. A labeled diagram showing the colorations of the sternal hairs of a female M. c. communis. Photo credits: Christopher Wilson (All Rights Reserved).
Male
According to Laberge (1956), the additive description of Male M. c. communis is as follows. The apical rims of the terga are often black to reddish brown opaque and only sometimes hyaline (Fig. 15), but very rarely are these rims colorless hyaline. The supraclypeal area is often dulled due to fine shagreening. The first flagellar segment’s minimum length is often approximately one-sixth of the second segment’s maximum length, but may be faintly shorter (very rarely of greater length; see Fig. 6).
The additive male setal description is as follows. The paler hairs of the head and thorax tend to be ochraceous (Fig. 16), although on the anterior mesoscutal half, these hairs sometimes become a paler rufescent color. The pale tergal hairs are often white (Fig. 17), but can range between white to pale ochraceous (the former more common, but the latter not distinctly rare). The second tergum’s distal pale pubescent band is often narrower than, or of equal width to, half of the medial portion of the apical area; rarely is this band noticeably wider than half the apical area’s width (Fig. 18). The pale pubescent band of the fifth tergum, when present, is apically positioned and medially interrupted; although, it's not uncommon for this band to be absent. The fifth and sixth terga sometimes bear a small amount of long pale lateral hairs, although this is more common on the former than the latter. The sternal hairs can be either ochraceous to red laterally and medially on the basal sterna and minimally brown medially with ochraceous to red hairs laterally on the distal sterna (although, all sterna may bear the distal sternal coloration), or these hairs can be mostly black to dark brown. The leg hairs are ochraceous to white, differing at the inner surfaces of all basitarsi and the hind tibiae, which are rufescent; and the distitarsi, which are also rufescent.

Fig. 15. A labeled diagram showing the integumental colorations of the apical tergal margins of a typical M. c. communis male. Note that males of this subspecies can range as described in the text above. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 16. A labeled diagram showing the ochraceous colorations of the pale thoracic hairs on a typical M. c. communis male. Note that males of this subspecies can range in this characteristic as described above. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 17. A labeled diagram showing the white colorations of the pale tergal hairs on a typical M. c. communis male. Note that males of this subspecies can range in this characteristic as described above. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 18. A labeled diagram showing the ratio of the width of the second tergum's distal pale band to the width of the apical area on a typical M. c. communis male. Photo credits: Christopher Wilson (All Rights Reserved).
Variation records
According to Laberge (1956), throughout M. c. communis’ range, intrasubspecific variability is quite common, especially in populations from different areas of their distribution. Along the Atlantic Coastal states and southwestward to Louisiana along the Gulf states, M. c. communis are often faintly darker and noticeably smaller than populations of this subspecies further inland that inhabit the plains states. Individuals from this Atlantic/Gulf area often have the pale hairs across their entire body very pale ochraceous to white, as opposed to the ochraceous (or pale rufescent on the anterior mesoscutal areas) head and thoracic hairs of individuals in the western areas of its range. In females from this area, the dark dorsal thoracic patch is always present and usually larger than those of westward populations, and while males are similar in this regard, the dark thoracic patch isn’t a guarantee. It is of note that, similar to most variable Melissodes species, females often express these foregoing vestiture characters to a higher magnitude than males. However, males still exhibit distinct setal and integumental coloration differences across their range. Males from the eastern areas commonly have opaque, dark brownish red to black tergal rims and almost always bear a large brownish red dorsal thoracic patch, while males from the western areas of this subspecies’ range often lack a brownish red patch and have translucent tergal rims. While the locational nature of this variation seems to be indicative of two subspecies hidden within M. c. communis (one in the central United states with males having translucent tergal rims with no brownish red thoracic patch and females having their pale hairs ochracous on the head and thorax with pale rufescent on the anterior mesoscutal areas; and an eastern subspecies in which males have opaque brownish red to black tergal rims and usually bear a large brownish red dorsal thoracic patch with females having their pale head and thoracic hairs very pale ochraceous to white), Laberge (1956) noted that characters in which would separate these hypothetical subspecies were not useful for a significant number of specimens, but rather a smooth gradient in color likely occurs across the landscape.
Four female anomalies examined by Laberge (1956), from which he determined to be M. communis based on species-specific characters (described above), exhibited vestiture color ranges outside of the normal M. c. communis range. One of these females bears almost entirely dark brown thoracic and head hairs (Kansas), the other three having their thoracic vestiture much more rufescent than typical of M. c. communis (Mexico). The former anomaly, as explained by Laberge (1956), has its tergal banding similar to that of a normal M. c. communis and is believed to be a specimen of deep melanism (similar to melanistic variants of M. comptoides of Southeastern United States, and M. rufodentatus of the West Indies). The three rufescent Mexican females, while this vestiture pattern isn’t incredibly unique to M. communis in general, distinctly resemble those of M. colliciatus; Laberge (1956) ensuring correct identification of these females via the complete bands of the second tergum and scarce punctures of the posteromedial mesoscutal area. Two males were also collected with the Mexican females and share the same rufescent thoracic color and the means of female identification above are sufficient to identify these males as M. communis as well. While M. c. communis is highly variable in the forms described above (and likely in more ways than currently documented), this subspecies is fairly simple to identify from M. c. alopex. For this reason, rather than presenting a subspecific variation table, a species wide table will be given below that accounts for the majority of morphs.
State records
M. c. communis has been collected in the following states. The number following each state corresponds to the amount of state-wide collections. For state records gathered from occurrences in the literature with no state-wide collection number given, the number is omitted: Florida, 337 (Florida State Collection of Arthropods, 2026; Illinois Natural History Survey, 2026; Bentley & Osborn, 2026; Ikerd, 2019; Dombroskie & Whisenant, 2026; University of Nebraska State Museum, 2026; University of Minnesota Insect Collection, 2026; Johnson, 2020), Kansas, 335 (Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; University of Nebraska State Museum, 2026; Dombroskie & Whisenant, 2026; University of Minnesota Insect Collection, 2026; Johnson, 2020), Mississippi, 107 (Mississippi Entomological Museum, 2026; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; Johnson, 2020), Texas, 105 (Ikerd, 2019; Illinois Natural History Survey, 2026; Bentley & Osborn, 2026; University of Nebraska State Museum, 2026; Johnson, 2020; Dombroskie & Whisenant, 2026), Nebraska, 92 (University of Nebraska State Museum, 2026; Ikerd, 2019; Illinois Natural History Survey, 2026; Bentley & Osborn, 2026; Johnson, 2020), Illinois, 72 (Illinois Natural History Survey, 2026; Bentley & Osborn, 2026; Johnson, 2020), Colorado, 46 (Florida State Collection of Arthropods, 2026; Bentley & Osborn, 2026; Illinois Natural History Survey, 2026; University of Nebraska State Museum, 2026; Johnson, 2020), North Carolina, 35 (Florida State Collection of Arthropods, 2026; Bentley & Osborn, 2026; University of Nebraska State Museum, 2026; University of Minnesota Insect Collection, 2026; Johnson, 2020), Minnesota, 27 (University of Minnesota Insect Collection, 2026; Bentley & Osborn, 2026), Georgia, 26 (Dombroskie & Whisenant, 2026; Bentley & Osborn, 2026; University of Nebraska State Museum, 2026; Johnson, 2020), New Mexico, 12 (Ikerd, 2019; Illinois Natural History Survey, 2026; Bentley & Osborn, 2026), Oklahoma, 12 (Bentley & Osborn, 2026), Alabama, 11 (Bentley & Osborn, 2026; Dombroskie & Whisenant, 2026; Johnson, 2020), Arizona, 10 (Ikerd, 2019; University of Arizona Insect Collection, 2026a; University of Nebraska State Museum, 2026; Bentley & Osborn, 2026), South Carolina, 8 (Bentley & Osborn, 2026; Johnson, 2020), Missouri, 5 (Illinois Natural History Survey, 2026; Bentley & Osborn, 2026; Dombroskie & Whisenant, 2026), California, 5 (Florida State Collection of Arthropods, 2026; Dombroskie & Whisenant, 2026), Chihuahua, 4 (Bentley & Osborn, 2026), Ontario, 3 (Gibbs, 2026), Iowa, 3 (Bentley & Osborn, 2026), Arkansas, 3 (Bentley & Osborn, 2026; Illinois Natural History Survey, 2026), Alberta, 3 (Bentley & Osborn, 2026), Coahuila de zaragoza, 4 (Sánchez & Comisión, 2025; Bentley & Osborn, 2026), Indiana, 2 (Bentley & Osborn, 2026), Hidalgo, 1 (Sánchez & Comisión, 2025), Tamaulipas, 1 (Bentley & Osborn, 2026), Nevada, 1 (Bentley & Osborn, 2026), Wyoming, 1 (Bentley & Osborn, 2026), Louisiana, 1 (Bentley & Osborn, 2026), Ohio, 1 (Johnson, 2026), Delaware, 1 (Johnson, 2020), and North Dakota, 1 (Johnson, 2020), Massachusetts (Laberge, 1956), Tennessee (Laberge, 1956).
M. c. alopex
This subspecies of M. communis varies much less than its subspecific counterpart, the latter comprising the majority of the variability that can be seen in Laberge’s (1956) key and in the table below. However, while less variable in vestiture and color traits in general, M. c. alopex still substantially varies in size (as do all both subspecies of M. communis). The current known range of M. c. alopex, encompassing a range that can be drawn from southern California, east to Colorado, and North towards Alberta and British Columbia, is the smallest of the two subspecies with much of its southern and southeastern areas contributing to the zone of intergradation. This subspecies can be separated from M. c. communis by the traits listed in the latter, but it is worth noting that the width of the pubescent band of the second tergum seems to be the definitive characteristic for subspecific delimitation, with the other characters listed instead used for supporting the identification (as can be determined when comparing subspecific variation). Descriptions of both sexes are presented below; these descriptions are meant to be added to, and override portions of, the base-line species description above.
Female
According to Laberge (1956), the additive description of female M. c. alopex is as follows. The apical margin of the first tergum is minimally translucent reddish brown, often colorless hyaline to yellow, and wider than a narrow sliver at the rim of the tergum. The wing veins are often a dark brownish red color (not usually distinctly black as in M. c. communis) and the wing membranes are often translucent/infumate yellow. The supraclypeal area is shiny with no shagreening. The upper impunctate “triangular area” of the propodeal slope is often small and ovular, or sometimes, the sloped surface of the propodium is almost entirely punctate with no distinct upper impunctate area. The small punctures of the basal area of the first tergum are mostly separated by less than one puncture diameter.
The additive female setal description is as follows. The mandibular, labral, and clypeal hairs are often ochraceous to yellow, but these hairs are never brown as in M. c. communis. The scutellum always bears a dark patch, this patch often large and its anterior margin is notched making it appear heart-shaped in outline. The pale hairs on the dorsal thoracic areas and the head are always ochraceous with no white or rufescent hairs present as in M. c. communis. The tergal bands and pale metasomal hairs are rarely paler than those of the thorax, or if paler, than only scarcely so. The lateral thoracic pale hairs are often ochraceous and only sometimes paler than the pale hairs on its dorsal surface. The mesepisterna often bear no dark hairs (apart from intergrades, see below). The distal pale band of the second tergum is minimally wider than one-third of the medial portion of the apical area’s width (that is, when unworn) and often wider than, or equal to, one-half of the medial portion of the apical area. The distal pale bands of the third and fourth tergum are often wider than those of M. c. communis, and the dark apicomedial patch of hairs that interrupt the apical band of the fourth tergum are often worn, but when present, ochraceous and suberect (never brown as in M. c. communis). The fifth and sixth terga always bear pale tufts of lateral ochraceous hairs. The sternal hairs are often red to dark brownish red, becoming ochraceous laterally and apically, apart from the ultimate sternum, which is dark brown with ochraceous lateral and apical hairs. The leg hairs are ochraceous, differing at the inner surfaces of all tibiae and all tarsi, which are yellow to red; the apical portions of the outer surfaces of the fore tibiae and middle tibiae, which are usually brown; the fore basitarsi, which are usually brown; and the basitibial plates, which are always brown.
Male
According to Laberge (1956), the additive description of male M. c. alopex is as follows. The apical tergal rims are always distinctly widely yellow to colorless hyaline. The supraclypeal area is shiny with no shagreening. The first flagellar segment’s minimum length is often approximately one-fifth of the second segment’s maximum length or more, and only sometimes is it shorter than this.
The additive male setal description is as follows. The pale thoracic and head hairs are often ochraceous, but on very rare occasions, these hairs may be white. The distal pale band of the second tergum is always more than one-half of the width of the medial portion of the apical area when unworn, and usually is of similar length to the apical area. The band on the fifth tergum is always present, complete, and reaches the apex across the entire tergum. The sixth and seventh terga always bear lateral pale tufts of long hairs. The sternal hairs are often mostly yellow to red, becoming white laterally, however, sometimes the ultimate sternum may bear only brownish red hairs. The leg hairs are mostly pale ochraceous, differing at the inner surfaces of the tarsi, which are orangeish-red.
Variation records
In comparison to its subspecific counterpart, M. c. alopex is much less variable, differing largely amongst populations only in size. However, intergrades, when present, are often the exception (as one might expect). Specifically, in the proposed zone of intergradation from his publication, Laberge (1956) noted a few anomalous females and males, of which distinctly deviate from the paler M. c. alopex, showing the darker characters as in those of M. c. communis, occur in this area. As can be seen in fig. 13, the current known overlapping region of the two subspecies’ ranges is much larger than originally assumed by Laberge (1956), suggestive of more variants than described in his publication. Six females, examined and identified by Laberge (1956), 5 from Salt Lake City, Utah and one from Logan, Utah, are much darker than a typical M. c. alopex; all five having their bands narrower than the average of this subspecies, but wider than a normal M. c. communis. One of the five Salt Lake City females bears small dark patches of hair ventrally on all mesepisterna, a characteristic associated with those of M. c. communis (although, the latter having much more of the ventral mesepisternal surface covered in dark hairs), another one bearing a dark apicomedial patch of hairs on the fourth tergum’s apical pale band (a distinctly M. c. communis trait; the foregoing specimens similar to that of a typical M. c. alopex, differing only at the listed characters. One male in Saltair, Utah, in general, having similar vestiture to a normal M. c. alopex, exhibits black sternal hairs (similar to that of M. c. communis) while another located in Troutcreek, Utah, has its tergal rims faintly, but noticeably, clouded with brown colorations and the distal band of the second tergum is narrowed. One male in Logan, Utah, is quite similar to an average M. c. alopex, differing at the lack of a pale band on the fifth tergum and a much narrower distal band on the second tergum. It should be noted that the foregoing descriptions seem to be reflective of intergrades between the two subspecies rather than melanistic varieties of M. c. alopex, especially given that many of the specimen localities are from the current-day estimated range of intergradation. A single male collected from the northern edge of the Grand Canyon seems to represent only known M. c. alopex specimen from Arizona, with the other M. communis observations south of the canyon constituting as M. c. communis; an interesting observation made by Laberge (1956) which in turn may possibly be suggestive of an environmental variable leading to fewer intermediate specimens in the state (although, this is just speculation).
From what can be determined above, the width of the distal pale band of the second tergum is a very important characteristic of subspecific delimitation; many specimens likely going uncategorized as M. c. alopex or M. c. communis based on ambiguity surrounding this trait (as can be seen in the discrepancy of floral records between subspecific and specific documentations). Given that very little variation truly occurs in M. c. alopex, with much of the variability listed above being due to intergrades, a subspecific variation table will be omitted and a species-wide table will instead be listed below.
State records
M. c. alopex has been collected in the following states. The number following each state corresponds to the amount of state-wide collections. For state records gathered from occurrences in the literature with no state-wide collection number given, the number is omitted: California, 368 (Cheadle Center for Biodiversity and Ecological Restoration, 2026a; Cheadle Center for Biodiversity and Ecological Restoration, 2026b; Gross & Oboyski, 2026; Gibbs, 2026; San Diego Natural History Museum, 2026; Florida State Collection of Arthropods, 2026; Illinois Natural History Survey, 2026; University of Nebraska State Museum, 2026; Bentley & Osborn, 2026; Johnson, 2020; MT James Entomological Collection, Washington State University, 2026), Washington, 34 (MT James Entomological Collection, Washington State University, 2026; Bentley & Osborn, 2026), Oregon, 9 (Bentley & Osborn, 2026; Johnson, 2020), British Columbia, 3 (Bentley & Osborn, 2026), Utah, 2 (Bentley & Osborn, 2026; Johnson, 2020), Texas, 1 (University of Nebraska State Museum, 2026), Nevada, 1 (Bentley & Osborn, 2026), and Colorado, 1 (Bentley & Osborn, 2026), Arizona (Laberge, 1956), Idaho (Laberge, 1956), Alberta (Laberge, 1956).
Intraspecific variability
From the subspecific variation records above, it can seem both sexes (and both subspecies for that matter) range largely in vestiture and integumental color (although not quite to the same extent as females of M. pallidisigantus or M. druriellus, but nearing so). Given that intergrades between the two subspecies occur (as can be seen in M. c. alopex’s variation records), likely in much of the southwestern quadrant of the United States, a broad description including ranges of characteristics cannot sufficiently capture all variation and may lead to some ambiguous traits. For this reason, two tables (one for each sex) will be given below, each table containing a list of traits that constantly differ as well as their variations in regard to melanism, including characteristics that aren’t directly implicative of melanistic value (e.g. the ratio of the male’s first flagellar segment to their second). Characters as such are given their melanistic value based on which subspecies (and/or intergrades) they are most commonly observed in, and that specific subspecies’ (and/or intergrades’) level of melanism. Important traits for identification as well as characters that consistently vary are prioritized in the tables below (such as the distal pale band of the second tergum or the upper impunctate triangular propodeal area) whereas characteristics that are fairly un-reliable to predict (leg hairs) or are somewhat inconsequential to identification and have already been listed above (the tufts of the fifth and sixth sterna of females) have been omitted. The morphological attributes in the table below along with their variations are based on the descriptions and key presented in Lasberge (1956), and similar to the subspecific descriptions, are meant to be applied to, and override portions of, the base-line M. communis description given above. A few characters have binary choices and others have multiple. In such scenarios (e.g. the ninth character of the female), they will be set into the respective characters' extremes (dark or light). Note that each value per character (light, intermediate, or dark) are independent from one another (although, many are correlated). Therefore a female M. communis may have no dark hairs on the vertex (palest choice for the first female character) and also have dark hairs on the inner surfaces of the hind basitarsi (darkest choice for the seventh female character).
Table 1. Seventeen characters pertaining to the vestiture, sculpture, and integumental color of the females of M. communis each with a maximum of different melanistic variations. This table accounts for all possible morphs of the females of M. communis when the characters are either added to, or replace, portions of the base-line pale description.
| Character | Light | Intermediate | Dark |
|---|---|---|---|
| Pale head hairs | the pale hairs of the head are very pale ochraceous to white. | the pale head hairs are darker ochraceous | the head hairs are almost entirely dark brown, pale hairs only present around the fossae of the antennae. |
| Vertex hairs: | the vertex hairs range between being similar in color to the pale hairs to a few dark brown hairs being present. | the vertex bears several to abundant dark brown hairs. | the vertex hairs are entirely dark, this dark patch reaching down the side of the head and covering most of the head in dark hairs. |
| Mandibular hairs | mandibular hairs are very pale ochraceous to ochraceous-yellow | mandibular hairs are distinctly brown, be it not extremely dark | the mandibular hairs are entirely dark brown |
| Clypeal hairs | the clypeal hairs are very pale ochraceous to ochraceous-yellow | the clypeaus bears a few distinctly brown, be it not extremely dark, hairs on its apical one-half to the entire apical one-half the the clypeus is brown. | the clypeus bears only dark brown hairs |
| Scutellar dark patch | the scutellar dark patch is absent to smaller than average; when present, composed of only several hairs rather than the majority. | a large dark patch is present, either becoming anteriorly notched, making it appear heart-shaped in outline, or rounded and fringed with pale hairs on its periphery | the scutellar hairs are almost entirely dark brown to black, bearing only a thin margin of pale hairs on its anterior area (between the suture of the scutellum and mesoscutum). |
| The pale dorsal thoracic hairs | these hairs are entirely white | these hairs are entirely ochraceous, never rufescent. | these hairs are rufescent to the entirety of the dorsal thoracic hairs are dark brown with only a thin margin of pale hairs between the suture of the mesoscutum and scutellum |
| Mesepisternal hairs | these hairs are entirely pale with no darkening ventrally. | The majority to all of the hairs are black to dark brown. | |
| The distal band of the third tergum | Pale hairs are distinctly present to all of the hairs are pale. | these hairs are mostly pale, but range from having a small dark patch of hairs on each mesepisterna ventrally, to the entire ventral mesepisternal surface as well as the lower lateral half and anterior areas being covered in dark hairs. | all mesepisternal hairs are dark brown. |
| The pale hairs of the metasoma | these hairs are often entirely white | these hairs are scarcely any paler than the pale thoracic hairs, often ochraceous to only slightly pale ochraceous. | |
| The distal pale band of the second tergum | The distal pale band of the second tergum is minimally wider than one-third of the medial portion of the apical area’s width (that is, when unworn) and often wider than, or equal to, one-half of the medial portion of the apical area. | The medial portion of the distal pale band of the second tergum often measures less than, or equal to, one third of the medial portion of the apical area. This band, at times, may be faintly wider, but never measures to be one-half of the apical area’s width medially. | |
| The apical band of the fourth tergum | mostly pale ochraceous, but when unworn, bears a small medial patch of ochraceous and suberect hairs apically; these hairs never brown and often absent due to wear. | mostly white, but minimally bears a small medial darkened patch of hairs in the apical areas, these hairs often becoming dark brown to black. | |
| Sternal hairs | The sternal hairs are often red to dark brownish red, becoming ochraceous laterally and apically, apart from the ultimate sternum, which is dark brown with ochraceous lateral and apical hairs | The sternal hairs are mostly black to dark brown, often becoming pale laterally to form lateral tufts. | |
| The upper impunctate triangle of the propodeum | absent to very small and somewhat indistinctly ovular. | somewhat medium-small sized and more ovular than triangular | often quite large and distinct. |
| The apical margin of the first tergum | the apical area of the first tergum is noticeably colorless to yellow hyaline to translucent brownish red apically, not distinctly narrower than that of an average M. (Eumelissodes). | the apical area of the first tergum is almost entirely black apart from an extremely narrow hyaline rim. | |
| Wing veins | the wing veins are a dark brownish red color | very dark red | black |
| The punctures on the interband zone of the second tergum | the interband zone of the second tergum bears small distinct punctures on either side of the lateral one-fourths of the tergum | the interband zone of the second tergum is either medially impunctate, or if punctate, then bearing shallow minute indistinct punctures that are often obscured due to dense shagreening | |
| Mesepisternal hairs | Mostly dark, but margined with reddish colorations | piceous to black |
Table 2. Ten characters pertaining to the structure, integumental color, and vesiture of males of M. communis each with a maximum of three different melanistic variations. This table accounts for all possible morphs of the males of M. communis when the characters are either added to, or replace, portions of the base-line pale description.
| Character | Light | Intermediate | Dark |
|---|---|---|---|
| The apical rims of the terga | always distinctly widely yellow to colorless hyaline. | smokey brown hyaline. | black to reddish brown opaque colored. |
| The ratio of F1/F2 | The first flagellar segment’s minimum length is often approximately one-fifth of the second segment’s maximum length or more | The first flagellar segment’s minimum length is shorter than one-fifth, but longer than one-sixth of the second segment’s maximum length. | The first flagellar segment’s minimum length is often approximately one-sixth of the second segment’s maximum length, but may be faintly shorter. |
| The integumental color of the tegulae | reddish to fuscous anteriorly. | piceous to black | |
| The pale hairs of the head | pale ochraceous to white | ochraceous | |
| Pale thoracic hairs | pale ochraceous to white | these hairs are mostly ochraceous, although on the anterior mesoscutal half, these hairs sometimes become a paler rufescent color. | bright, deep, saturated ochraceous to somewhat rufescent |
| The dark scutaller patch of hairs | a dark patch is absent | this patch is reduced to only a few to several hairs or is instead reddish brown. | a dark scutellar patch is distinctly present and large, often shrouding the medial area of the scutellum with pale hairs fringing its margins. |
| The hairs of the mesoscutum | the mesoscutum bears no distinct patch contrasting with the pale thoracic hairs | The mesoscutum has a large reddish brown dorsal thoracic patch. | |
| The distal pale band of the second tergum | The distal pale band of the second tergum is always more than one-half of the width of the medial portion of the apical area when unworn, and usually is of similar length to the apical area. | The distal pale band of the second tergum is approximately one-half of the width of the apical area to slightly more, never narrower. | The second tergum’s distal pale pubescent band is commonly narrower than, or somewhat similar in width to, one-half of the medial portion of the apical area; rarely is this band noticeably wider than half the apical area’s width. |
| The pale pubescent band of the fifth tergum | The pale band of the fifth tergum is apical (at least laterally) and complete | The pale band on the fifth tergum is apical (at least laterally) and medially interrupted | The pale band on the fifth tergum is absent |
| The sternal hairs | The sternal hairs are yellow to red with white lateral hairs, sometimes the apical sternum is entirely brownish red | The sternal hairs are ochraceous to red on the basal sterna, and minimally brown medially with ochraceous to red hairs laterally on the distal sterna (although, all sterna may bear the distal sternal coloration). | the sternal hairs are mostly black to dark brown |
Gynandromorph records
Two gynandromorphs of M. communis have been reported (Campbell et al., 2024), the latter having full documentation and imagery, of which is presented below. All descriptions and morphometrics are derived from the data and images of Campbell et al. (2024). This specific specimen, in general, exhibits male characteristics, bearing no scopae on the hind tibiae and basitarsi (Fig. 19), its metasomal width measuring 4.01mm (although within the range of females, a measurement of such would be quite extreme), and having partially developed male genitalia (Fig. 20). However, the antennae are similar to that of a female, having 10 flagellar segments with the first being distinctly longer than the subsequent segments (Fig. 21). The labrum and the clypeus are bilaterally split between the black integumental colors of a female, and the yellow (clypeus) and cream-colored (labrum) colors of a male (Fig. 21). The mandibular colorations, however, are similar to that of a female on both sides of the bilateral split, the majority (including the bases) entirely black, becoming rufescent apically with an apical golden macula (Fig. 21). Additionally, the body length measures 11.26 mm, and although M. communis is known to range widely in size, when compared to Laberge’s measurements, a length of such is smaller than the average of a male (still within male range) and smaller than the minimum of a female. The collection of this specimen in Billings County, North Dakota is implicative of it being M. c. communis based on range alone, and while the thoracic hairs and tergal rims resemble those of a male from this subspecies, the angle from which the image of the distal pale band of the second tergum was taken makes it challenging to properly compare the band's width to the apical area. It’s quite interesting that, in this specimen, the majority of female characteristics (at least those presented by the images) are primarily aggregated on the head with the remainder of the body exhibiting relatively typical characteristics to that of a male. While bilaterality (as would be named in Torre & Friese, 1899) of the clypeal and labral coloring is quite striking, due to the remainder of the facial characteristics being distinctly female and the body expressing primarily male characters, a better interpretation would be the nomenclature proposed by that of Michez et al. (2010), classifying such specimens as “mosaics” (Campbell et al., 2024).

Fig. 19. A labeled diagram showing the lack of scopae on Campbell et al.'s M. communis gynandromorph. Photo credits: Campbell et al., 2024.

Fig. 20. A labeled diagram showing the partially developed male genitalia on Campbell et al.'s M. communis gynandromorph. Photo credits: Campbell et al., 2024.

Fig. 21. A labeled diagram showing the distincly female-structured antennae, female colored mandibles, and bilateral sexual diamorphism split on the clypeus and labrum on Campbell et al.'s M. communis gynandromorph. Photo credits: Campbell et al., 2024.
Location and Habitat
Melissodes communis, as the species epithet and vernacular name suggests, is a commonly recorded and fairly abundant Melissodes that inhabits the majority of the United States, Mexico, and southern parts
of Canada (Laberge, 1956; see fig. 13 below). As described above, two subspecies of M. communis are present across its range, a largely Eastern and Southern subspecies (M. c. communis) and a Western subspecies
(M. c. alopex). In his revision, Laberge (1956) documented the former to inhabit the following states: Alabama, Arkansas, Arizona, Colorado, Delaware, Florida, Georgia, Illinois, Indiana, Iowa, Kansas, Louisiana,
Massachusetts, Minnesota, Mississippi, Missouri, Nebraska, New Mexico, North Carolina, North Dakota, Oklahoma, South Carolina, South Dakota, Tennessee, Texas, Wyoming, Chihuahua, and Tamaulipas; and the latter
to inhabit Arizona, California, Colorado, Idaho, Nevada, Oregon, Utah, Washington, Alberta, and British Columbia. While the current range of both subspecies are similar, M. c. communis has been additionally
found in California (Florida State Collection of Arthropods, 2026), Nevada (Bentley & Osborn, 2026; although, this observation lacks a recorded identifier; GBIF record 657848537), and Onterio (Gibbs, 2026;
GBIF record 4540067639), while M. c. alopex has yet to be recorded outside the states listed by Laberge (1956). In his publication, using a dataset that had yet to document M. c. communis in the western areas
from which it’s currently known, Laberge (1956) proposed an estimated distribution of the two subspecies as well as their hypothesized zone of intergradation based on intermediate specimen locations. It is of
note that this original zone of intergradation proposed by Laberge (1956) has no distinct overlap between the two subspecies, but rather bridges the medial portion of the western edge of M. c. communis’ estimated
range and the medial portion of the eastern edge of M. c. alopex’s (encompassing the majority of Utah and parts of Colorado, Fig. 22); further suggestive of the intergradation zone being determined based on
anomalous specimens that fit neither subspecific classification (such specimens are described in the “Variation records” of M. c. communs and M. c. alopex above). However, the additional collections of M. c.
communis in states westward of its original proposed range are suggestive of a possible enlargement of the zone of intergradation than that postulated by Laberge. As stated in Laberge’s (1956) publication, the
male holotype of M. c. alopex was likely collected in the original hypothesized intergradation zone (Ducesne, Utah), with its vestiture and integumental color resembling that of a typical M. c. alopex male from
California; the presence of such specimens (distinctly one subspecies with no intermediate characteristics) being expected to occur in intergradation areas. Given that the Nevadan and Californian M. c. communis
specimens were definitely identified to subspecies, this may be representative of the pattern Laberge (1956) described in regards to the holotype M. c. alopex male, suggesting that the zone of integration may encompass the southwestern quadrant of the United States
(Fig. 23). However, the lack of data throughout Arizona and the Northwestern corner of New Mexico (areas that may include the expanded zone of intergradation) make it challenging to be certain whether this is
true or not.
Due to the polylectic nature of M. communis (see “Flower records” for more detail), an analysis of the habitats in which this species occupies based on floral records cannot occur. Until a species specific
nesting biology study is done, the environmental features needed to support M. communis, and the environments inhabited by M. communis, remain unknown.
Currently, the only phenological activity published for M. communis states that this species has been documented to be active throughout the months of March to September with peak activity, on average,
occurring in the later parts of June through August; although, different populations (and both subspecies) slightly vary in phenology (Laberge, 1956). Laberge (1956) noted that M. c. communis in Florida are
active between March and September, in Texas between April and September, and in Nebraska between June to September; each population having their peak activity between late June through August. Although too
few specimens existed at Laberge’s (1956) time of publication for population-based phenologies, M. c. alopex’s range-wide activity seems to commence around May and end in September with peak activity occurring
throughout June and July (Laberge, 1956). Newer data present a similar pattern to Laberge’s descriptions, however, species-wide, M. communis has been collected between March and November, although the end of
activity usually occurs in September or October (Fig. 24). Range wide, M. c. communis has been currently collected between March and November with peak activity occurring predominantly in July (although June and
August are also of substantial value); the state location records being fairly similar to those presented by Laberge (1956), although, May has the highest amount of collections for Florida and Texas (Fig. 25).
Current range wide M. c. alopex records are similar to those listed by Laberge (1956), however, June has the highest records of all months recorded (Fig. 26).
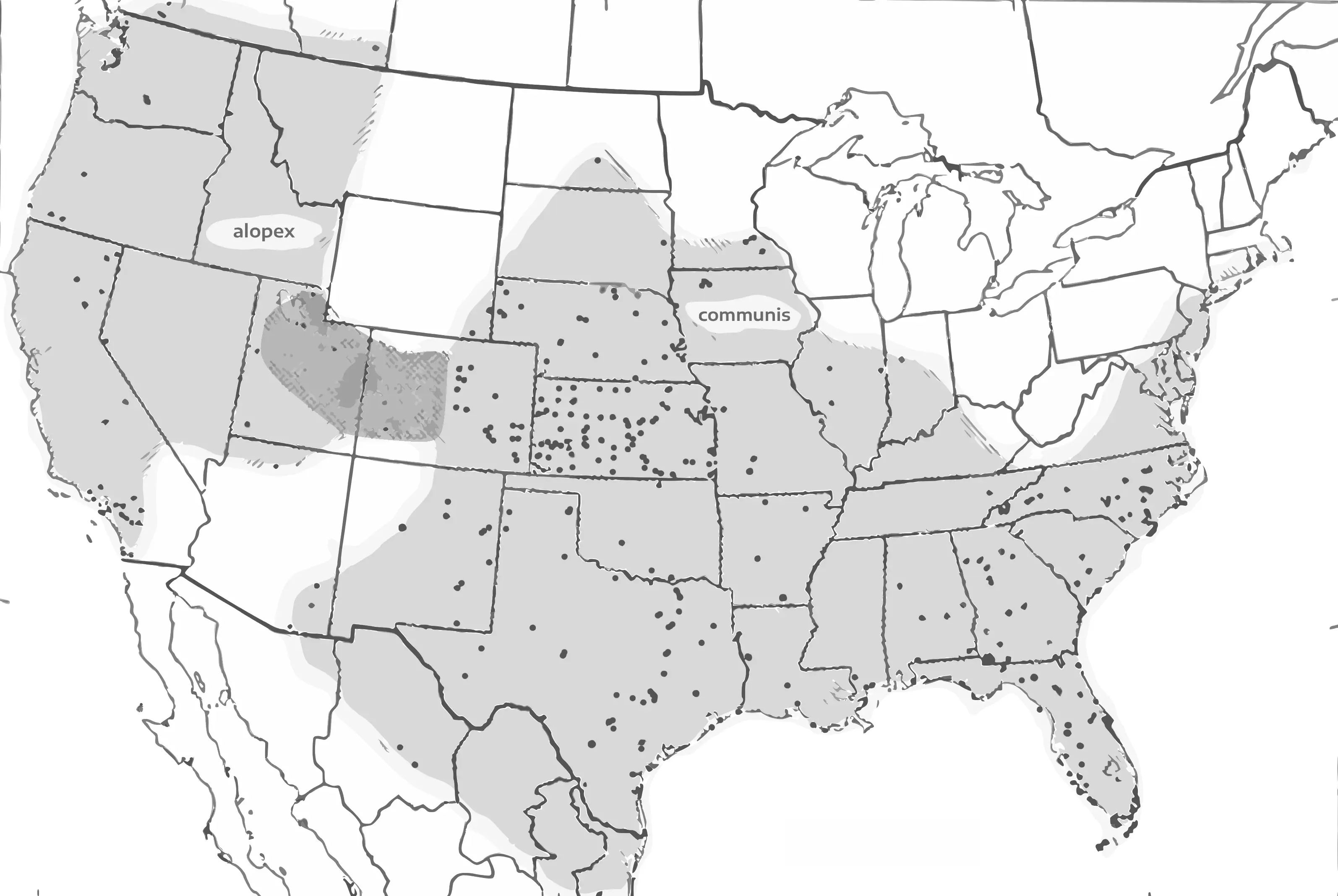
Fig. 22. Laberge's (1956) original estimated M. communis distribution map upscaled and vectorized, originally presented as "Map showing the distribution of M. (Melissodes) communis. The overlapping type of shading indicated the zone of intergradation between the two subspecies."
Fig. 23. Map showing an estimation for the known distribution for M. (Melissodes) communis. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Orange circles represent occurances of M. c. alopex, dark blue represents M. c. communis, and teal represents specimens that have only been identified to species rank. Data derived from (Laberge, 1956; Eardley & Ranwashe, 2017; Ikerd, 2019; Johnson, 2019; Johnson, 2020; Best et al., 2022; Archbold Biological Station, 2023; Carril et al., 2023; Ikerd & Engler, 2023; Purdue Entomological Research Collection, 2023; Texas A&M University Insect Collection, 2023; North Carolina State University Insect Collection, 2024; Veit & McFarland, 2024; Droege & Maffei, 2025; European Bioinformatics Institute (EMBL-EBI), GBIF Helpdesk, 2025; Holm et al., 2025; Pozo & Comisión, 2025; Quiroz & Comisión, 2025; Scott, 2025; Sánchez & Comisión, 2025; Zhuang, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Best, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026a; Cheadle Center for Biodiversity and Ecological Restoration, 2026b; Clemson University Arthropod Collection, 2026; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Dombroskie & Whisenant, 2026; Florida State Collection of Arthropods, 2026; Frost Entomological Museum, 2026; Gibbs, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Kelly et al., 2026; Kenneth S. Norris Center for Natural History, 2026; Luther College, 2026; Mertz et al., 2026; Mississippi Entomological Museum, 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Natural Science Research Laboratory, Museum of Texas Tech University (TTU), 2026; New Mexico State Collection of Arthropods, 2026; Northern Arizona University, 2026; Orrell, 2026a; San Diego Natural History Museum, 2026; The International Barcode of Life Consortium, 2026; UMMZ & LSA, 2026; University of Arizona Insect Collection, 2026a; University of Arizona Insect Collection, 2026b; University of Kentucky, 2026; University of Minnesota Insect Collection, 2026; University of Nebraska State Museum, 2026; Virginia Tech Insect Collection, 2026; Wild, 2026; Soon n.d.). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0.
Fig. 24. A figure showing the species-wide phenological activity of M. communis. The x value is the month, and the y value is the number of documented observations. Data derived from (Eardley & Ranwashe, 2017; Ikerd, 2019; Johnson, 2019; Johnson, 2020; Best et al., 2022; Archbold Biological Station, 2023; Carril et al., 2023; Ikerd & Engler, 2023; Purdue Entomological Research Collection, 2023; Texas A&M University Insect Collection, 2023; North Carolina State University Insect Collection, 2024; Veit & McFarland, 2024; Droege & Maffei, 2025; European Bioinformatics Institute (EMBL-EBI), GBIF Helpdesk, 2025; Holm et al., 2025; Pozo & Comisión, 2025; Quiroz & Comisión, 2025; Scott, 2025; Sánchez & Comisión, 2025; Zhuang, 2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Best, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026a; Cheadle Center for Biodiversity and Ecological Restoration, 2026b; Clemson University Arthropod Collection, 2026; Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Dombroskie & Whisenant, 2026; Florida State Collection of Arthropods, 2026; Frost Entomological Museum, 2026; Gibbs, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Kelly et al., 2026; Kenneth S. Norris Center for Natural History, 2026; Luther College, 2026; Mertz et al., 2026; Mississippi Entomological Museum, 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Natural Science Research Laboratory, Museum of Texas Tech University (TTU), 2026; New Mexico State Collection of Arthropods, 2026; Northern Arizona University, 2026; Orrell, 2026a; San Diego Natural History Museum, 2026; The International Barcode of Life Consortium, 2026; UMMZ & LSA, 2026; University of Arizona Insect Collection, 2026a; University of Arizona Insect Collection, 2026b; University of Kentucky, 2026; University of Minnesota Insect Collection, 2026; University of Nebraska State Museum, 2026; Virginia Tech Insect Collection, 2026; Wild, 2026; Soon n.d.). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0.
Fig. 25. A figure showing the phenological activity of M. c. communis. The x value is the month, and the y value is the number of documented observations. Data derived from (Ikerd, 2019; Johnson, 2020; Sánchez & Comisión, 2025; Bentley & Osborn, 2026; Dombroskie & Whisenant, 2026; Florida State Collection of Arthropods, 2026; Gibbs, 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Mertz et al., 2026; Mississippi Entomological Museum, 2026; Orrell, 2026a; University of Arizona Insect Collection, 2026a; University of Minnesota Insect Collection, 2026; University of Nebraska State Museum, 2026). Data licensed under CC BY 4.0, CC0 1.0, and CC BY-NC 4.0.
Fig. 26. A figure showing the phenological activity of M. c. alopex. The x value is the month, and the y value is the number of documented observations. Data derived from (Johnson, 2020; Bentley & Osborn, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026a; Cheadle Center for Biodiversity and Ecological Restoration, 2026b; Florida State Collection of Arthropods, 2026; Gibbs, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Orrell, 2026a; San Diego Natural History Museum, 2026; University of Nebraska State Museum, 2026). Data licensed under CC BY 4.0, CC0 1.0, CC BY-NC 4.0.
Bionomics
Even though M. communis is one of the most common species and has high potential to be an important pollinator for agricultural crops, there has yet to be a study concerning itself with the nesting habitats, behaviors, and overall ecology of this species. Similarly to M. bimaculatus (Harms & , Owens, 2025), M. agilis (Parker et al,. 1981), M. nigroaenea (Silva & Andrade, 2022), and likely other Eucerini, males of M. communis have been documented in sleeping aggregations (Laberge, 1956). This observation, documented by C. V. Riley in Centerville, Florida on the 29th of August (year not presented), detailed a cluster of M. c. communis males sleeping or resting on a small twig (Laberge, 1956). According to Laberge (1956) the males, along with the structure on which they slept, were collected and strung together with loose black thread and subsequently displayed in the U. S. National Museum’s collection. A similar natural observation to this, photographed by (insert name), can be seen in Fig. 18. For what can be assumed based on genus-wide similarities among other species with ecological publications, M. communis is a ground-nesting bee with a maximum of one generation per year. Unfortunately, this is the extent of current knowledge.
Flower records
Similar to most species in the subgenus M. (Melissodes), M. communis is polylectic; the trait that makes this species a great candidate for agricultural pollination. In his revision, Laberge (1956) presented two floral record lists (one for each subspecies) and one floral preference table (M. c. communis). While a similar structure will be presented here, a species-wide floral record list and graph of preference will aditionally be given, and as the collections of M. c. alopex have grown since Laberge’s (1956) publication, so too will a graph of preference for this subspecies. While the combination of the two subspecies’ records should in theory re-create the species-wide records, naturally, given that identification to species rather than subspecies, more specimens are identified to their specific rank rather than what may be their true subspecific rank (that is, unless a specimen is an intergrade). Many such records contain floral data that would be unaccounted for if only subspecific records were tallied.
Species-wide
All flower records included in this list are from reports in the literature or datasets. Each flower has a parenthesized reference listed after it, corresponding to the literary work or dataset in which it was recorded. Abutilon theophrasti (Laberge, 1956), Acacia sp. (University of Arizona Insect Collection, 2026a; GBIF record 4070494078), Achillea millefolium (Holm et al., 2025; GBIF record 5061141941), Acmispon glaber (Laberge, 1956; old synonym), Alcea rosea (Laberge, 1956; old synonym), Amorpha sp. (Laberge, 1956), Angadenia berteroi (Florida State Collection of Arthropods, 2026; GBIF record 5220125976), Argemone intermedia (Bentley & Osborn, 2026; GBIF record 658333101), Asclepias syriaca (Laberge, 1956), Asclepias tuberosa (Laberge, 1956), Aster sp. (Mississippi Entomological Museum, 2026; GBIF record 4870183650), Aureolaria pedicularia (Illinois Natural History Survey, 2026; GBIF record 3801852696), Baptisia sp. (Laberge, 1956), Baptisia tinctoria (Veit & McFarland, 2024; GBIF record 4068307346), Bejaria racemosa (Laberge, 1956; old synonym), Berteroa incana (Holm et al., 2025; GBIF record 5061148724), Bidens sp. (Veit & McFarland, 2024; GBIF record 4068297943), Bidens alba (Archbold Biological Station, 2023; GBIF record 1933510645), Blephilia ciliata (Mississippi Entomological Museum, 2026; GBIF record 4870191605), Blephilia hirsuta (Laberge, 1956), Brassica sp. (Laberge, 1956), Brazoria truncata (Laberge, 1956); Campanula sp. (Laberge, 1956), Cantinoa mutabilis (Florida State Collection of Arthropods, 2026; old synonym; GBIF record 5220126045), Capraria biflora (Florida State Collection of Arthropods, 2026; GBIF record 5220126077), Carduus nutans (Scott, 2025; GBIF record 5283848955), Carthamus tinctorius (Bentley & Osborn, 2026; GBIF record 658378533), Centaurea cuneifolia (UMMZ & LSA, 2026; old synonym; GBIF record 3009121409), Cephalanthus occidentalis (Mississippi Entomological Museum, 2026; GBIF record 5833192043), Chamaecrista fasciculata (Laberge, 1956; old synonym), Chrysopsis sp. (Mississippi Entomological Museum, 2026; GBIF record 4870183657), Chrysothamnus viscidiflorus (Best et al., 2022; GBIF record 3499812600), Cirsium sp. (Laberge, 1956), Cirsium arvense (Holm et al., 2025; GBIF record 5061147936), Cirsium discolor (Laberge, 1956),Cirsium vulgare (Laberge, 1956; old synonym), Clarkia amoena (Laberge, 1956; old synonym), Clarkia unguiculata (Laberge, 1956; old synonym), Cleomella serrulata (Laberge, 1956; old synonym), Convolvulus sp. (Laberge, 1956), Coreopsis sp. (Johnson, 2026; GBIF record 872915709), Coreopsis basalis (Florida State Collection of Arthropods, 2026; GBIF record 5220126202), Coreopsis lanceolata (Holm et al., 2025; GBIF record 5061147936), Coreopsis major (Mississippi Entomological Museum, 2026; GBIF record 5833192115), Croton sp. (Laberge, 1956), Cryptantha sp. (Illinois Natural History Survey, 2026; GBIF record 3801339802), Cucurbita sp. (Laberge, 1956), Cucurbita pepo (The International Barcode of Life Consortium, 2026; GBIF record 5863910697), Cuscuta sp. (Illinois Natural History Survey, 2026; GBIF record 3801376803), Cylindropuntia californica (Illinois Natural History Survey, 2026; old synonym; GBIF record 3801376791), Cylindropuntia imbricata (Bentley & Osborn, 2026; old synonym; GBIF record 1913918333), Cyrilla racemiflora (Laberge, 1956; old synonym), Dalea sp. (Laberge, 1956), Dalea candida (Laberge, 1956; old synonym), Dalea flavescens (Bentley & Osborn, 2026; GBIF record 658210092), Dalea multiflora (Laberge, 1956), Dalea pogonathera (Bentley & Osborn, 2026; GBIF record 658146619), Dalea purpurea (Laberge, 1956; old synonym), Dianthera americana (Laberge, 1956), Duranta erecta (Laberge, 1956; old synonym), Datura innoxia (Laberge, 1956; old synonym), Daucus carota (Florida State Collection of Arthropods, 2026; GBIF record 5220126522), Deinandra fasciculata (Cheadle Center for Biodiversity and Ecological Restoration, 2026a; GBIF record 6168506256), Desmodium sp. (Purdue Entomological Research Collection, 2023; GBIF record 3128645909), Echinacea angustifolia (Holm et al., 2025; GBIF record 5061141941), Echinacea purpurea (Luther College, 2026; GBIF record 5102737303), Echium vulgare (Laberge, 1956), Engelmannia peristenia (University of Nebraska State Museum, 2026; GBIF record 6177202459), Eriastrum virgatum (Laberge, 1956; old synonym), Ericameria ericoides (Kenneth S. Norris Center for Natural History, 2026; GBIF record 2562749820), Ericameria nauseosa (Best et al., 2022; GBIF record 3467769653), Erigeron annuus (Holm et al., 2025; GBIF record 5061147936), Eriodictyon trichocalyx (Laberge, 1956), Eriogonum nudum (Kenneth S. Norris Center for Natural History, 2026; GBIF record 2562703983), Erigeron strigosus (Florida State Collection of Arthropods, 2026; old synonym; GBIF record 5220126184), Euphorbia marginata (Illinois Natural History Survey, 2026; GBIF record 3801938742), Gaillardia sp. (University of Nebraska State Museum, 2026; GBIF record 6177195510), Gaillardia pulchella (University of Nebraska State Museum, 2026; GBIF record 6177195125), Geranium sp. (Carril et al., 2023; GBIF record 3421491127), Gilia capitata (Laberge, 1956), Gossypium herbaceum (Laberge, 1956), Grindelia sp. (Laberge, 1956), Gutierrezia sarothrae (Bentley & Osborn, 2026; GBIF record 658325978), Helenium amarum (Laberge, 1956; old synonym), Helianthus sp. (Laberge, 1956), Helianthus annuus (Laberge, 1956), Helianthus mollis (Illinois Natural History Survey, 2026; GBIF record 3802161042), Helianthus petiolaris (University of Nebraska State Museum, 2026; GBIF record 6177201642), Heliopsis helianthoides (Holm et al., 2025; GBIF record 5061147936), Heliotropium sp. (Laberge, 1956; old synonym), Herissantia crispa (Florida State Collection of Arthropods, 2026; old synonym; GBIF record 5220126081), Heterotheca angustifolia (Laberge, 1956; old synonym), Heterotheca sessiliflora (Kenneth S. Norris Center for Natural History, 2026; GBIF record 2562714447), Heterotheca subaxillaris (Mississippi Entomological Museum, 2026; GBIF record 5833191053), Heterotheca villosa (Scott, 2025; GBIF record 5283851342), Hooveria parviflora (Cheadle Center for Biodiversity and Ecological Restoration, 2026a; GBIF record 6168511180), Hypericum perforatum (Holm et al., 2025; GBIF record 5061148724), Hyrtia sp. (Laberge, 1956; however, this taxon is absent from current datasets and may be invalid), Ipomea sp. (Droege & Maffei, 2025; GBIF record 1456713834; a doubtful taxon, likely a historic synonym for Ipomoea), Ipomoea sp. (Laberge, 1956), Isocoma menziesii (Cheadle Center for Biodiversity and Ecological Restoration, 2026a; GBIF record 6168500374), Jacquemontia curtissii (Florida State Collection of Arthropods, 2026; GBIF record 5220125975), Lachnanthes caroliana (Archbold Biological Station, 2023; GBIF record 1933510663), Lactuca sp. (Laberge, 1956), Linaria vulgaris (Holm et al., 2025; GBIF record 5061141941), Linum sulcatum (Mississippi Entomological Museum, 2026; GBIF record 4870184527), Lophiola sp. (Mississippi Entomological Museum, 2026; GBIF record 5833191812), Lotus sp. (Laberge, 1956), Ludwigia peruviana (Florida State Collection of Arthropods, 2026; old synonym; GBIF record 5220126550), Lythrum alatum (Laberge, 1956), Lythrum lineare (Laberge, 1956), Macroptilium lathyroides (Holm et al., 2025; GBIF record 5061147936), Malacothamnus fasciculatus (Laberge, 1956; old synonym), Malva sylvestris (Laberge, 1956), Medicago sp. (Illinois Natural History Survey, 2026; GBIF record 3801093379), Medicago sativa (Laberge, 1956), Melanthera nivea (Florida State Collection of Arthropods, 2026; GBIF record 5220126004), Melilotus sp. (Laberge, 1956), Melilotus albus (University of Nebraska State Museum, 2026; GBIF record 6177195101), Melilotus officinalis (Laberge, 1956; old synonym), Mentha sp. (Laberge, 1956), Mesembryanthemum crystallinum (University of Nebraska State Museum, 2026; GBIF record 6177195096), Mimosa sp. (Bentley & Osborn, 2026; GBIF record 657716479), Momordica charantia (Archbold Biological Station, 2023; GBIF record 1933510689), Monarda sp. (Laberge, 1956), Monarda citriodora (Laberge, 1956), Monarda fistulosa (Laberge, 1956), Monarda pectinata (Laberge, 1956), Monarda punctata (Laberge, 1956), Monardella breweri (Laberge, 1956; old synonym), Morinda royoc (Florida State Collection of Arthropods, 2026; GBIF record 5220125979), Nepeta cataria (Laberge, 1956), Ocimum campechianum (Florida State Collection of Arthropods, 2026; GBIF record 5220126157), Oenothera glazioviana (Laberge, 1956; old synonym), Oenothera laciniata (Laberge, 1956), Opuntia sp. (Laberge, 1956), Opuntia engelmannii (Illinois Natural History Survey, 2026; old synonym; GBIF record 3801374743), Opuntia humifusa (Archbold Biological Station, 2023; GBIF record 1933510699), Opuntia littoralis (Laberge, 1956), Opuntia macrorhiza (University of Nebraska State Museum, 2026; GBIF record 6177195137), Passiflora sp. (Laberge, 1956), Passiflora incarnata (Laberge, 1956), Penstemon sp. (Illinois Natural History Survey, 2026; GBIF record 3801856829), Phacelia sp. (Bentley & Osborn, 2026; GBIF record 657635812), Phacelia ramosissima (Laberge, 1956; old synonym), Phaseolus sp. (Laberge, 1956), Phaseolus lunatus (Florida State Collection of Arthropods, 2026; old synonym; GBIF record 5220126096), Platycodon grandiflorus (Laberge, 1956), Pluchea baccharis (Archbold Biological Station, 2023; old synonym; GBIF record 1933510661), Polygala rugelii (Archbold Biological Station, 2023; GBIF record 1933510717), Polygonum hydropiperoides (Florida State Collection of Arthropods, 2026; GBIF record 5220126526), Pontederia cordata (Florida State Collection of Arthropods, 2026; GBIF record 5220126546), Proboscidea louisianica (Laberge, 1956),Pycnanthemum sp. (Laberge, 1956), Pycnanthemum flexuosum (Laberge, 1956), Pycnanthemum incanum (Illinois Natural History Survey, 2026; GBIF record 3801533354), Quincula lobata (Bentley & Osborn, 2026; GBIF record 1092785220), Ratibida columnifera (Laberge, 1956), Ratibida pinnata (University of Nebraska State Museum, 2026; GBIF record 6177202310), Rhus sp. (Laberge, 1956), Rhus copallina (Florida State Collection of Arthropods, 2026; old synonym; GBIF record 5220126110), Rhus glabra (Laberge, 1956), Richardia scabra (Florida State Collection of Arthropods, 2026; GBIF record 5220126165), Rorippa sylvestris (Purdue Entomological Research Collection, 2023; GBIF record 3128645903), Rubus sp. (The International Barcode of Life Consortium, 2026; GBIF record 4916337054), Rudbeckia sp. (Laberge, 1956), Rudbeckia subtomentosa (Illinois Natural History Survey, 2026; GBIF record 3802160064), Sabal etonia (Archbold Biological Station, 2023; GBIF record 1933510723), Sabal palmetto (Florida State Collection of Arthropods, 2026; GBIF record 5220125328), Salvia sp. (Laberge, 1956), Salvia apiana (Laberge, 1956), Salvia carnosa (Laberge, 1956), Salvia clevelandii (Cheadle Center for Biodiversity and Ecological Restoration, 2026; GBIF record 6168506806), Salvia munzii (Cheadle Center for Biodiversity and Ecological Restoration, 2026a; GBIF record 6168506853), Salvia yangii (Scott, 2025; GBIF record 5283853741), Sapindus drummondii (Bentley & Osborn, 2026; GBIF record 657850296), Scabiosa sp. (Laberge, 1956), Sidalcea reticulata (Laberge, 1956; however, this taxon is absent from current datasets and may be invalid), Silphium sp. (Mississippi Entomological Museum, 2026; GBIF record 4870191595), Silphium integrifolium (University of Nebraska State Museum, 2026; GBIF record 6177195433), Silphium laciniatum (Mississippi Entomological Museum, 2026; GBIF record 4870191032), Solanum carolinense (Holm et al., 2025; GBIF record 5061147936), Solanum elaeagnifolium (Laberge, 1956), Solidago sp. (Laberge, 1956), Solidago canadensis (University of Nebraska State Museum, 2026; GBIF record 6177203043), Solidago serotina (Laberge, 1956), Sphaeralcea sp. (Laberge, 1956), Stachys ajugoides (Laberge, 1956), Stachytarpheta urticifolia (Florida State Collection of Arthropods, 2026; GBIF record 5220125325), Stenaria nigricans (Mississippi Entomological Museum, 2026; old synonym; GBIF record 4870181486), Stephanomeria exigua (Laberge, 1956), Stokesia laevis (Florida State Collection of Arthropods, 2026; GBIF record 5220126179), Taraxacum officinale (Holm et al., 2025; GBIF record 5061147936), Tetragonotheca helianthoides (Florida State Collection of Arthropods, 2026; GBIF record 5220126534), Teucrium sp. (Laberge, 1956), Teucrium canadense (Laberge, 1956), Thelesperma sp. (Holm et al., 2025; GBIF record 5061147936), Thelesperma megapotamicum (Laberge, 1956), Trichostema arizonicum (Bentley & Osborn, 2026; GBIF record 1913918323), Trichostema lanatum (Florida State Collection of Arthropods, 2026; GBIF record 5220126151), Trifolium fragiferum (Laberge, 1956; old synonym), Trifolium pratense (Mississippi Entomological Museum, 2026; GBIF record 4870184535), Trifolium repens (Scott, 2025; GBIF record 5283860346), Tuberosa sp. (Cheadle Center for Biodiversity and Ecological Restoration, 2026b; GBIF record 4911494034), Verbascum blattaria (Holm et al., 2025; GBIF record 5061147936), Verbena sp. (Laberge, 1956), Verbena hastata (Holm et al., 2025; GBIF record 5061141941), Verbena stricta (Laberge, 1956), Verbena urticifolia (University of Nebraska State Museum, 2026; GBIF record 6177195461), Vernonia sp. (Laberge, 1956), Vernonia baldwinii (Laberge, 1956), Vernonia gigantea (Mississippi Entomological Museum, 2026; GBIF record 5102821599), Vernonia glauca (Laberge, 1956), Vicia floridana (Florida State Collection of Arthropods, 2026; GBIF record 5220126102), Vicia villosa (University of Nebraska State Museum, 2026; GBIF record 6177195142), Ximenia americana (Archbold Biological Station, 2023; GBIF record 1933510664).
Fig. 27. A graph showing the raw current-day known floral data of M. (Melissodes) communis from 24 datasets. Data derived from (Best et al., 2022; Archbold Biological Station, 2023; Carril et al., 2023; Purdue Entomological Research Collection, 2023; Veit & McFarland, 2024; Droege & Maffei, 2025; Holm et al., 2025; Scott, 2025; Bentley & Osborn, 2026; Best, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026a; Cheadle Center for Biodiversity and Ecological Restoration, 2026b; Florida State Collection of Arthropods, 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Kenneth S. Norris Center for Natural History, 2026; Luther College, 2026; Mississippi Entomological Museum, 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; The International Barcode of Life Consortium, 2026; UMMZ & LSA, 2026; University of Arizona Insect Collection, 2026a; University of Nebraska State Museum, 2026). Data licensed under CC BY-NC 4.0, CC0 1.0, CC BY 4.0.
Fig. 28. A graph showing unique events of current-day known floral data of M. (Melissodes) communis from 24 datasets. Each floral taxon is filtered to only include records with unique latitudes, longitudes, or dates in an attempt to prevent single-site sampling bias. Data derived from (Best et al., 2022; Archbold Biological Station, 2023; Carril et al., 2023; Purdue Entomological Research Collection, 2023; Veit & McFarland, 2024; Droege & Maffei, 2025; Holm et al., 2025; Scott, 2025; Bentley & Osborn, 2026; Best, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026a; Cheadle Center for Biodiversity and Ecological Restoration, 2026b; Florida State Collection of Arthropods, 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Kenneth S. Norris Center for Natural History, 2026; Luther College, 2026; Mississippi Entomological Museum, 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; The International Barcode of Life Consortium, 2026; UMMZ & LSA, 2026; University of Arizona Insect Collection, 2026a; University of Nebraska State Museum, 2026). Data licensed under CC BY-NC 4.0, CC0 1.0, CC BY 4.0.
As can bee seen by the sheer number of floral families it has been collected upon (Figs. 27 & 28) along with the list of 216 species-wide floral records (212 if omitting ambiguous records), M. communis is undoubtedly polylectic (Wright, 2018). This species, while not explicitly having a floral preference as in many other Melissodes, has been collected atop the family Asteraceae most frequently (Figs. 18 & 19), an interesting occurrence given that neither subspecies seems to prefer this family (Figs. 20-23). A phenomenon as such is indicative of a data discrepancy between the number of specimens identified to specific and subspecific level. These findings, while not contradictory to those of Laberge (1956), may suggest that generally, M. communis is most abundant atop Asteraceae and that subspecific records are currently too few to reflect this. It is still of note, however, that the floral family on which Laberge’s data recorded M. c. communis (M. c. alopex’s preference was not recorded) to be most prevalent, Fabaceae (historically Leguminosae), is still of significant value in the graphs above.
M. c. communis
All flower records included in this list are from reports in the literature or datasets. Each flower has a parenthesized reference listed after it, corresponding to the literary work or dataset in which it was recorded. Abutilon theophrasti (Laberge, 1956), Acacia sp. (University of Arizona Insect Collection, 2026a; GBIF record 4070494078), Alcea rosea (Laberge, 1956; old synonym), Amorpha sp. (Laberge, 1956), Angadenia berteroi (Florida State Collection of Arthropods, 2026; GBIF record 5220125327), Argemone intermedia (Bentley & Osborn, 2026; GBIF record 658333101), Asclepias syriaca (Laberge, 1956), Asclepias tuberosa (Laberge, 1956), Aster sp. (Mississippi Entomological Museum, 2026; GBIF record 4870183650), Aureolaria pedicularia (Illinois Natural History Survey, 2026; GBIF record 3801852696), Baptisia sp. (Laberge, 1956), Bejaria racemosa (Laberge, 1956; old synonym), Blephilia ciliata (Mississippi Entomological Museum, 2026; GBIF record 4870191605), Blephilia hirsuta (Laberge, 1956), Brazoria truncata (Laberge, 1956), Campanula sp. (Laberge, 1956), Capraria biflora (Florida State Collection of Arthropods, 2026; GBIF record 5220126077), Carthamus tinctorius (Bentley & Osborn, 2026; GBIF record 658378533), Chamaecrista fasciculata (Laberge, 1956; old synonym), Chrysopsis sp. (Mississippi Entomological Museum, 2026; GBIF record 4870183657), Cirsium sp. (Laberge, 1956), Cirsium discolor (Laberge, 1956), Cirsium vulgare (Laberge, 1956; old synonym), Cleomella serrulata (Laberge, 1956; old synonym), Convolvulus sp. (Laberge, 1956), Coreopsis sp. (Johnson, 2026; GBIF record 872915709), Coreopsis basalis (Florida State Collection of Arthropods, 2026; GBIF record 5220126202), Croton sp. (Laberge, 1956), Cucurbita sp. (Laberge, 1956), Cyrilla racemiflora (Laberge, 1956; old synonym), Dalea sp. (Laberge, 1956), Dalea candida (Laberge, 1956; old synonym), Dalea flavescens (Bentley & Osborn, 2026; GBIF record 658210092), Dalea multiflora (Laberge, 1956), Dalea pogonathera (Bentley & Osborn, 2026; GBIF record 658146619), Dalea purpurea (Laberge, 1956; old synonym), Dianthera americana (Laberge, 1956), Daucus carota (Florida State Collection of Arthropods, 2026; GBIF record 5220126522), Echium vulgare (Laberge, 1956), Erigeron strigosus (Florida State Collection of Arthropods, 2026; old synonym; GBIF record 5220126184), Euphorbia marginata (Illinois Natural History Survey, 2026; GBIF record 3801938742), Gaillardia sp. (University of Nebraska State Museum, 2026; GBIF record 6177195510), Gaillardia pulchella (University of Nebraska State Museum, 2026; GBIF record 6177195125), Gossypium herbaceum (Laberge, 1956), Grindelia sp. (Laberge, 1956), Helenium amarum (Laberge, 1956; old synonym), Helianthus sp. (Laberge, 1956), Helianthus annuus (Laberge, 1956), Helianthus petiolaris (Bentley & Osborn, 2026; GBIF record 657818783), Heliopsis helianthoides (University of Nebraska State Museum, 2026; GBIF record 6177195454), Heliotropium sp. (Laberge, 1956; old synonym), Herissantia crispa (Florida State Collection of Arthropods, 2026; old synonym; GBIF record 5220126081), Heterotheca angustifolia (Laberge, 1956; old synonym), Hyrtia sp. (Laberge, 1956; however, this taxa is absent from current datasets and may be invalid), Ipomoea sp. (Laberge, 1956), Lactuca sp. (Laberge, 1956), Linum sulcatum (Mississippi Entomological Museum, 2026; GBIF record 4870184527), Ludwigia peruviana (Florida State Collection of Arthropods, 2026; old synonym; GBIF record 5220126550), Lythrum alatum (Laberge, 1956), Lythrum lineare (Laberge, 1956), Malva sylvestris (Laberge, 1956), Medicago sp. (Illinois Natural History Survey, 2026; GBIF record 3801093379), Medicago sativa (Laberge, 1956), Melanthera nivea (Florida State Collection of Arthropods, 2026; GBIF record 5220126004), Melilotus sp. (Laberge, 1956), Melilotus albus (University of Nebraska State Museum, 2026; GBIF record 6177195101), Melilotus officinalis (Laberge, 1956; old synonym), Mentha sp. (Laberge, 1956), Mimosa sp. (Bentley & Osborn, 2026; GBIF record 657716479), Monarda sp. (Laberge, 1956), Monarda citriodora (Laberge, 1956), Monarda fistulosa (Laberge, 1956), Monarda pectinata (Laberge, 1956), Monarda punctata (Laberge, 1956), Nepeta cataria (Laberge, 1956), Ocimum campechianum (Florida State Collection of Arthropods, 2026; old synonym; GBIF record 5220126157), Oenothera glazioviana (Laberge, 1956; old synonym), Oenothera laciniata (Laberge, 1956), Opuntia sp. (Laberge, 1956), Opuntia engelmannii (Illinois Natural History Survey, 2026; old synonym; GBIF record 3801374743), Opuntia macrorhiza (University of Nebraska State Museum, 2026; GBIF record 6177195137), Passiflora sp. (Laberge, 1956), Passiflora incarnata (Laberge, 1956), Phacelia sp. (Bentley & Osborn, 2026; GBIF record 657635812), Phaseolus sp. (Laberge, 1956), Phaseolus lunatus (Florida State Collection of Arthropods, 2026; old synonym; GBIF record 5220126096), Platycodon grandiflorus (Laberge, 1956; old synonym), Polygonum hydropiperoides (Florida State Collection of Arthropods, 2026; GBIF record 5220126526), Pontederia cordata (Florida State Collection of Arthropods, 2026; GBIF record 5220126546), Proboscidea louisianica (Laberge, 1956), Pycnanthemum sp. (Laberge, 1956), Pycnanthemum flexuosum (Laberge, 1956), Quincula lobata (Bentley & Osborn, 2026; GBIF record 1092785220), Ratibida columnifera (Laberge, 1956), Ratibida pinnata (Mississippi Entomological Museum, 2026; GBIF record 4870181495), Rhus sp. (Laberge, 1956), Rhus copallina (Florida State Collection of Arthropods, 2026; old synonym; GBIF record 5220126110), Rhus glabra (Laberge, 1956), Richardia scabra (Florida State Collection of Arthropods, 2026; GBIF record 5220126165), Rudbeckia sp. (Laberge, 1956), Sabal palmetto (Florida State Collection of Arthropods, 2026; GBIF record 5220125328), Salvia sp. (Laberge, 1956), Sapindus drummondii (Bentley & Osborn, 2026; GBIF record 657850296), Sidalcea reticulata (Laberge, 1956; however, this taxa is absent from current datasets and may be invalid), Silphium sp. (Mississippi Entomological Museum, 2026; GBIF record 4870191595), Silphium integrifolium (University of Nebraska State Museum, 2026; GBIF record 6177195433), Silphium laciniatum (Mississippi Entomological Museum, 2026; GBIF record 4870191032), Solanum elaeagnifolium (Laberge, 1956), Solidago sp. (Laberge, 1956), Solidago serotina (Laberge, 1956), Stachytarpheta urticifolia (Florida State Collection of Arthropods, 2026; GBIF record 5220125325), Stenaria nigricans (Mississippi Entomological Museum, 2026; old synonym; GBIF record 4870181486), Stokesia laevis (Florida State Collection of Arthropods, 2026; GBIF record 5220126179), Tetragonotheca helianthoides (Florida State Collection of Arthropods, 2026; GBIF record 5220126534), Teucrium sp. (Laberge, 1956), Teucrium canadense (Laberge, 1956), Thelesperma megapotamicum (Laberge, 1956), Trifolium pratense (Mississippi Entomological Museum, 2026; GBIF record 4870184535), Verbena sp. (Laberge, 1956), Verbena stricta (Laberge, 1956), Verbena urticifolia (University of Nebraska State Museum, 2026; GBIF record 6177195461), Vernonia sp. (Laberge, 1956), Vernonia baldwinii (Bentley & Osborn, 2026; GBIF record 657591508), Vernonia glauca (Laberge, 1956), Vicia floridana (Florida State Collection of Arthropods, 2026; GBIF record 5220126102), Vicia villosa (University of Nebraska State Museum, 2026; GBIF record 6177195142).
Fig. 29. A graph showing the raw current-day known floral data of M. (Melissodes) communis ssp. communis from 7 datasets. Data derived from (Bentley & Osborn, 2026; Florida State Collection of Arthropods, 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Mississippi Entomological Museum, 2026; University of Arizona Insect Collection, 2026a; University of Nebraska State Museum, 2026). Data licensed under CC BY 4.0 and CC0 1.0 and CC BY-NC 4.0.
Fig. 30. A graph showing unique events of current-day known floral data of M. (Melissodes) communis ssp. communis from 7 datasets. Each floral taxon is filtered to only include records with unique latitudes, longitudes, or dates in an attempt to prevent single-site sampling bias.Data derived from (Bentley & Osborn, 2026; Florida State Collection of Arthropods, 2026; Illinois Natural History Survey, 2026; Johnson, 2026; Mississippi Entomological Museum, 2026; University of Arizona Insect Collection, 2026a; University of Nebraska State Museum, 2026). Data licensed under CC BY 4.0 and CC0 1.0 and CC BY-NC 4.0.
In contrast to the species-wide findings above, M. c. communis seems instead to prefer Fabaceae, with Asteraceae taking a close second (Figs. 29 & 30). These data, while partially reflecting Laberge’s original subspecific floral preference, still distinctly highlight Asteraceae as an important family to M. c. communis (at least in comparison to other family on which it’s been collected); a significant change to the presumed preference of Asteraceae in historic literature (Laberge, 1956). Of the genera from which M. c. communis has been collected, Meliliotus (45), Dalea (31), Opunita (29), and Medicago (19) are of favorability. However, unique records (records defined as having different latitudes, longitudes, or dates) present a different pattern: Melolotus having 7 records, Dalea with 13, Opunita with 9, and Medicago with 8. While Monarda has 24 raw records, its unique tally only amounted to 5, hence the omission of favorability. From these lists, it can be seen that Dalea, having the second largest number of raw records and largest number of unique records, may be an important taxa to M. c. communis (although, the prevalence of this genus planted for the human gaze may play a role in favorability bias). Unfortunately, many specimens whose data contribute to these floral records are of an unreported sex, therefore making a statement of true preference (based on pollen collection of females) unfeasible at this date. Nonetheless, M. c. communis, as expressed by Laberge (1956), is undoubtedly polylectic, having collections from 36 different families and 87 different genera; a quite astonishing and uncommonly large pallet for a subspecies within a genus of many specialists and oligolects.
M. c. alopex
All flower records included in this list are from reports in the literature or datasets. Each flower has a parenthesized reference listed after it, corresponding to the literary work or dataset in which it was recorded. Acmispon glaber (Laberge, 1956; old synonym), Brassica sp. (Laberge, 1956), Cirsium sp. (University of Nebraska State Museum, 2026; GBIF record 6177195191), Clarkia amoena (Laberge, 1956; old synonym), Clarkia unguiculata (Laberge, 1956; old synonym), Cryptantha sp. (Illinois Natural History Survey, 2026; GBIF record 3801339802), Cuscuta sp. (Illinois Natural History Survey, 2026; GBIF record 3801376803), Cylindropuntia californica (Illinois Natural History Survey, 2026; old synonym; GBIF record 3801376791), Duranta erecta (Laberge, 1956; old synonym), Datura innoxia (Laberge, 1956; old synonym), Deinandra fasciculata (Cheadle Center for Biodiversity and Ecological Restoration, 2026a; GBIF record 6168506256), Eriastrum virgatum (Laberge, 1956; old synonym), Eriodictyon trichocalyx (Laberge, 1956), Gilia capitata (Laberge, 1956), Hooveria parviflora (Cheadle Center for Biodiversity and Ecological Restoration, 2026; GBIF record 6168511180), Isocoma menziesii (Cheadle Center for Biodiversity and Ecological Restoration, 2026a; GBIF record 6168500374), Lotus sp. (Laberge, 1956), Malacothamnus fasciculatus (Laberge, 1956; old synonym), Medicago sativa (Laberge, 1956), Melilotus sp. (Laberge, 1956), Mesembryanthemum crystallinum (University of Nebraska State Museum, 2026; GBIF record 6177195096), Monardella breweri (Laberge, 1956; old synonym), Opuntia sp. (Cheadle Center for Biodiversity and Ecological Restoration, 2026a; GBIF record 6168506589), Opuntia littoralis (Laberge, 1956), Penstemon sp. (Illinois Natural History Survey, 2026; GBIF record 3801856829), Phacelia sp. (University of Nebraska State Museum, 2026; GBIF record 6177195199), Phacelia ramosissima (Laberge, 1956; old synonym), Salvia sp. (Illinois Natural History Survey, 2026; GBIF record 3801941817), Salvia apiana (Laberge, 1956), Salvia carnosa (Laberge, 1956), Salvia clevelandii (Cheadle Center for Biodiversity and Ecological Restoration, 2026a; GBIF record 6168506806), Salvia munzii (Cheadle Center for Biodiversity and Ecological Restoration, 2026a; GBIF record 6168506853), Scabiosa sp. (Laberge, 1956), Sphaeralcea sp. (Laberge, 1956), Stachys ajugoides (Laberge, 1956), Stephanomeria exigua (Laberge, 1956), Trichostema lanatum (Florida State Collection of Arthropods, 2026; GBIF record 5220126151), Trifolium fragiferum (Laberge, 1956; old synonym).
Fig. 31. A graph showing the raw current-day known floral data of M. (Melissodes) communis ssp. alopex from 5 datasets. Data derived from (Bentley & Osborn, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026a; Florida State Collection of Arthropods, 2026; Illinois Natural History Survey, 2026; University of Nebraska State Museum, 2026). Data licensed under CC BY 4.0 and CC BY-NC 4.0 and CC0 1.0.
Fig. 32. A graph showing unique events of current-day known floral data of M. (Melissodes) communis ssp. alopex from 5 datasets. Each floral taxon is filtered to only include records with unique latitudes, longitudes, or dates in an attempt to prevent single-site sampling bias. Data derived from (Bentley & Osborn, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026a; Florida State Collection of Arthropods, 2026; Illinois Natural History Survey, 2026; University of Nebraska State Museum, 2026). Data licensed under CC BY 4.0 and CC BY-NC 4.0 and CC0 1.0.
Although the list is much smaller due to the paucity of M. c. alopex collections, it can be seen that this subspecies, in regards to floral records, is fairly similar to M. c. communis; not distinctly preferring Asteraceae as the species-wide records suggest (Figs. 31 & 32). However, M. c. alopex differs from its subspecific counterpart in that (from the data driving the graphs above), Lamiaceae has both the highest raw and unique floral records by somewhat large margins (Figs. ). It is of note that, while not represented in the graphs above due to the lack of numerical data, this subspecies has been collected from four Fabaceae taxa (Acmispon glabar, Medicago savita, Melilotus, and Trifolium fragiferum), all of which are listed Laberge’s (1956) subspecific floral records. Unfortunately, only 11 of the 18 families from which M. c. alopex has been collected currently include numerical data (those of which are listed in the graphs above), the remaining families are as follows: Fabaceae, Onagraceae, Polemoniaceae, Verbenaceae, Solanaceae, Namaceae, Caprifoliaceae (Laberge, 1956). While the true floral preference can therefore not be accounted for, it can be said that, much like M. c. communis, M. c. alopex is highly polylectic; its collections consisting of 31 separate genera, 15 of which bear some form of numerical data. Although incredibly incomplete, of the 15 genera which can currently be compared, M. c. alopex has been collected atop Salvia the most, this floral taxon having the largest tally of raw and unique records in comparison to the other genera. However, until more direct sampling efforts are made, or more data become available, a direct statement of floral preference cannot be given with certainty.
Applicable biology
Due to the nature of its generalization, Melissodes communis has been found atop numerous floral taxa, provisioning for both pollen and nectar, many of which are crops of significant agricultural importance such as cotton (Laberge, 1956; Olson et al., 2021), peanuts (Olson et al., 2021), sunflowers, (Laberge, 1956), lavender (Wilson et al., 2024), squash (Laberge, 1956; The International Barcode of Life Consortium, 2026; GBIF record 58639106970), carrots (Florida State Collection of Arthropods, 2026; GBIF record 5220126522), phasey beans (Holm et al., 2025; GBIF record 5061147936), alfalfa (Laberge, 1956), sweet clovers (Laberge, 1956), mint (Laberge, 1956), bitter melons (Archbold Biological Station, 2023; GBIF record 1933510689), catmint (Laberge, 1956), prickly pears (Laberge, 1956), passionflowers (Laberge, 1956), black berries (The International Barcode of Life Consortium, 2026; GBIF record 4916337054), sage (Laberge, 1956), and likely many others. Although no direct studies on the proficiency and efficacy of pollination by Melissodes communis have yet been published, promising results of other species in the genus are suggestive of an importance for Melissodes in agricultural settings. A research study by Parker (1981) evaluating the efficiency of different bee taxa in sunflower fields, found that flowers pollinated by Melissodes agilis, a Helianthus specialist, produce the largest yields of seeds in comparison to the remaining taxa of his study (including A. mellifera). Furthermore, a study of watermelon pollination documented that Melissodes (along with Bombus impatiens) repeatedly deposit the largest amounts of pollen onto the stigmas of watermelons each visit in comparison to A. mellifera (Campbell et al., 2018). An additional publication detailing the foraging habits of Melissodes on crops with dimorphic parental lines demonstrated that Melissodes not only visit inflorescence at a much higher rate per minute than that of A. mellifera, but also distinctly prioritize the collection of pollen over the latter (a reasonable outcome when one takes into account a female Melissodes’ need for pollen during the provision of her young; Barcala et al., 2019). Many of these accounts are suggestive of a noticeable increase in agricultural yield with the addition of Melissodes as pollinators. Given M. communis’ polylectic dietary trait along with the numerous crops on which it’s been documented, it’s not unreasonable to assume that this species may play an important role (or at least significantly contribute to) the pollination of commercial crops.
Taxonomy and Phylogeny
Melissodes communis is a highly variable species, which has undoubtedly led to its many (7) synonymies, that was originally described in 1878 by Ezra Townsend Cresson. 20 specimens (numerical data for each sex is not given although it is known that multiple of each sex were collected based on the presence of lectotypes and lectoallotypes; Laberge, 1956) comprised Creason’s original M. communis material, all of which were collected in Georgia and Illinois. It is of note that a holotype (or in this case lectotype) specimen attached to the name “Melissodes communis” (while this is what Cresson titled his examined individuals) does not truly exist, rather two type specimens, one for each subspecies, compose the name-bearing material of this taxon. Therefore, it should herein be seen that the specimens on which Cresson’s description is based are not “M. communis” (as this would not be possible), but instead M. c. communis before subspecific ranks had been acknowledged (Laberge, 1956). Within this description, Cresson (1878) described female M. c. communis as having its “tegulae piceous...abdomen shining, base and sides of first segment with cinereous pubescence; narrow band at extreme base of second segment, another, slightly arcuated, across the middle, a broad band near base of third, and another on apex of fourth, sometimes subinterrupted on posterior middle, composed of short sense appressed whitish pile…Length .45 inch.” As would be expected, this aligns quite well with the descriptions above, especially in the characters of metasomal vestiture. It is of note that “abdomen shining,” in this case, likely does not represent an entirely unshagreened surface as it may imply, but rather a comparatively shiny surface to that of noticeably dense shagreening. Descriptions of the integumental color of the tegulae also differ from the descriptions of Laberge (1956), appearing piceous instead of black (a character that seems to be a partial source of erroneous new subsequent species descriptions). Additional remarks of vestiture by Cresson (1878) include “...head and thorax clothed with a short dense cinereous pubescence, very short on clypeus…disk of mesothorax and scutellum with short sparse black pubescence; disk of meta thorax almost nude, the pubescence on sides long…four anterior legs with mixed fuscous and ochraceous pubescence, posterior tibiae and tarsi with long dense fulvous pubescence, which is darker within…” While many of these characters are known to vary (especially the leg vestiture), it is interesting to see that the females from which Cresson (1878) based his description, at least from what he wrote, all lacked dark hairs on the apical one-half of the clypeus. Cresson (1878) described the males to resemble those of the females in metasomal vestiture apart from “...the fifth abdominal segment (which sometimes) has an indistinct band of pale hairs…”, and to have their antennae “... two-thirds the length of (their) body, third joint scarcely twice the length of second, flagellum fulvous beneath…” The legs, as described by Cresson (1878) are “...clothed with ochraceous pubescence, that on tarsi beneath golden, tarsi simple, pale ferruginous at tips…Length .45 inch.” The similarity between the female and male metasomal vestiture is suggestive of a thin distal band of the second tergum (given that the range in which this band varies in females is smaller than males; the widest often measuring one-half of the apical area’s width). Pairing these descriptions with the locational variability of M. c. communis, it can be seen that the specimens, on which Cresson’s descriptions were based, would currently be classified as the slightly darker and smaller eastern variations. The designated female lectotype and male lectoallotype of Creasson’s specimens are in the Academy of Natural Science of Philadelphia (Laberge, 1956).
In 1905, Cockerell proposed a new species of Melissodes, M. hortivagans, presenting only a description of the male. The male from which his description is based was collected from Garden City, Kansas by H. W. Menke on August of 1895, and, as noted by Cockerell, most notably differs from Creasson’s (1878) description of M. c. communis in the larger size, yellow wing membranes, fewer black thoracic hairs, and the red-colored tegulae. The vestiture of the fifth tergum described by Cockerell (1905a) is of interest “...fifth and following segments with black hairs, but the hair on the apex of the fifth is party light, producing a rather ill-defined light band…”, such a characteristic likely being classified by Laberge (1956) as “interrupted”. Variations in Cockerell’s proposed species were also noted in his 1905 publication, a group of five males collected in Morton County, Kansas, bearing similarities to M. hortivagans, but in general, having the structure of Creasson’s (1878) M. c. communis specimens. One female was collected foraging with the foregoing five males, this specimen mostly resembling Creasson’s (1878) M. c. communis specimens, differing in the leg vestiture, which is slightly darker; the breadth of the metasoma and seemingly the thorax, which is wider; and the area between the lateral margin of the dark mesoscutal patch and the tegulae, which is wider. Cockerell (1905a) attributes this wider area to be due to the increased area of the thorax rather than a smaller mesoscutal patch, but, with current knowledge it seems to be a combination of these two. That is, the overall ratio of the dark thoracic patch to the remainder of the dorsal thoracic area of Cockerell’s (1905a) specimens may be smaller than the ratio of Cresson’s (1878) specimens, but the overall larger size of the former could, in theory, bear a similarly sized dark patch to the latter. As M. communis is known to vary quite widely in size, the specimens Cockerell describes align today with those of a fairly common and somewhat average M. c. communis, males bearing dark hairs on the scutellum, the dorsum of the thorax “faintly tinged with ochraceous,” pale hairs on the legs with reddish orange on the inner surface of the tarsi, and a narrow median to submedian band across the second tergum. This is further supported when looking at their collection site. What is of great interest from Cockerell’s description is the integumental color of the male tegulae, which are “...red, fuscous in front…” and of the female tegualae, which are “...dark with a narrow reddish margin…” These described variations have not been outlined by Laberge (1956), and while they may be mischaracterizations, such differences are included in the species variation table above. The male M. hortivagans holotype described by Cockerell is deposited in the University of Kansas’ Snow Entomological Museum (Laberge, 1956).
In the same year as his M. hortivagans treatment, Cockerell (1905b) proposed and described another species of Melissodes, M. martini, from two female specimens collected in Las Valles, New Mexico on the sixth of August (no year given; although, likely 1905). These females, measuring approximately 13.5 mm., most notably differ from those of Creasson’s taxon in their larger size, lighter scopal hairs, and yellow wing membranes (Cockerell, 1905b). The description of the metasoma, including vestiture, structure, structure, and sculpture, is noteworthy in Cockerell’s publication, the author stating “...(abdomen) broad but convex…which has a dullish surface…hind margin of first abdominal segment narrowly white; base of first segment with erect white hair (no black intermixed); extreme base of second with white hair; median narrow snow-white hair-bands on segments 2 to four, that on 2 linear and interrupted in the middle, those on 3 and 4 slightly angled in the middle, or that on 4 may extend to hind margin of the segment, thus becoming very broad…” Many of these characters align quite nicely with a modern-day M. c. communis female (Laberge, 1956), however, the remark about the second tergum’s distal pale band becoming medially interrupted is somewhat strange considering the opposite of this such character is determinative of female identification (from other species that is). Given that Laberge (1956) recognizes the female M. martini holotype (seemingly designated as such by Cockerell given that two females were collected together) as a historical synonym of M. c. communis, it’s likely that the medial interruption interpreted by Cockerell (1905b) is due to wear rather than a distinct species trait. Furthermore, with these females having been collected in August, it’s reasonable to assume that the specimens were not “fresh” (bearing full wing margins and unworn setae). These females also exhibit a similar tegular integumental color and wing color to the females of his species proposed above, M. hortivagans, having distinctly yellow wing membranes and dark tegulae with red colors posteriorly. It’s unknown as to why Cockerell separated these two groups of incredibly similar specimens in his treatments as it’s not directly addressed. Currently, the females of M. martini would be classified as somewhat paler M. c. communis, remarks such as “ hair of labrum pale reddish” supporting subspecific identification and “pubescence of head and thorax pale, dull whitish below, more ochraceous above, but nowhere bright colored” suggestive of the darker ochraceous specimens of western M. c. communis as described above. The female M. martini holotype is cared for by the California Academy of Sciences (Laberge, 1956) and likely resides in their collection.
Melissodes variabilis, proposed by Charles Robertson in 1905, is a peculiar historic synonym as it has only appeared twice in the literature (Robertson, 1905; Robertson, 1928), neither appearance outlining a description. Robertson’s second publication containing this taxa (Robertson, 1928) is a list of insects and the flowers they have been found atop (which has no taxonomic contributions), but his first (Robertson, 1905) consists of a key to the subfamily Euceridae in Carlinville, Illinois, the couplets from which will be used in place of a formal description. Robertson lists female M. variabilis as having “...more or less clouded or infuscated (wings)...wings yellowish…”, “...a continuous apical pale pubescent fascia (on the fourth tergum)…”, “...scopa, except on metatarsus posteriorly, pale ochraceous; mesonotum with a subquadrate black patch…”, “...abdomen without broad fasciae on segments 2-3…”, and “...fascia on segment 2 narrow, median, continuous; 3 with basal fascia; fascia on four with a patch of black pubescence on the disc; vertex more or less black in front…tegulae piceous, sometimes testaceous…” The male key commences with the first couplet separating current day Melissodes from Epimelissodes (which were all classified as Melissodes at the time of Robertson’s publication) and therefore, the reconstruction of the male M. variabilis description with begin from the couplet in relation to Melissodes (i.e. couplet 3). Robertson (1905) describes male M. variabilis as having “...base of mandibles and labrum usually colored…”, “...pubescence not black, except sometimes on vertex, mesonotum, scutel, and abdomen…”, “...(abdominal) segment 2 obscurely punctured, with basal and median whitish fasciae, continuous fasciae on 3-4…”, “...wings and nervures yellowish, more fuscous beyond middle; pubescence ochraceous; ornaments yellow…”, “...(abdominal) segment 5 usually with white fascia, 6-7 black…tegulae usually black, more or less testaceous posteriorly; apical margins of segments sometimes pale testaceous; (antennal) joint 3 about twice as long as 2.” 50 specimens comprise the material from which Robertson based his key (although unknown how many of each sex); these specimens only ranging between 11-13 mm. in length in the female sex and 12-14 mm. in the male sex (Robertson, 1905). It is of note that males seemed to measure slightly larger than females in many cases, and while M. communis is known to vary quite largely in size, generally, females are larger than their male counterparts. Given that all specimens were found in Carlinville, Illinois (Robertson, 1905), and that this state seems to be well outside the range of Laberge’s (1956), as well as the predicted current day, zone of intergradation, it’s possible that Carlinville, Illinois is an anomalous locality (although, this is just speculation and may very well be an underrepresented sample of M. communis’ true variation in this location). The tegulae of the females from Robertson’s (1905) key may represent a more common morph, that described by Laberge (1956), being testaceous to piceous rather than the rufescence and reddish colorations described in the synonyms above. It can be seen that currently, M. variabilis may be classified as a somewhat darker to intermediate M. c. communis, likely fairly similar to those of the Gulf states in size, with males bearing “pubescence ochraceous; ornaments yellow,” but also having the “apical margins of (tergal) segments sometimes pale testaceous.” It’s unknown the extent to what “pale testaceous” implies from Robertson’s publication alone, although this likely refers to somewhat hyaline tergal rims. Females unfortunately lack descriptions of many subspecific and clinal diagnostic characters; the character of most taxonomic significant documented being “...fascia on 4 with a patch of black pubescence on the disc…” which identifies these females to M. c. communis. Due to this, while the assumption of melanism above may be close, it cannot be stated with certainty where these individuals fit in the cline of M. c. communis without specimen examination. The female M. variabilis lectotype and male lectoallotype, designated by Laberge (1956), reside in the collection of the Illinois Natural History Survey (Laberge, 1956).
Melissodes manni, described by Cockerell in 1928 from a single male found at Garden Canyon, Arizona, follows a very similar pattern to many of Cockerell’s other historic synonym descriptions above. Males of this proposed species, as noted by Cockerell (1928), are most similar to M. kallstroemiae (currently M. comptoides) in gestalt and M. xanthopteralis (currently M. c. communis) in the distinctly yellow wings, differing from the former in the larger size, and from the former and latter in the presence of a broad “pure white” hair band on the fifth tergum. The description of this male given by Cockerell (1928) states that males have “...light fulvous hair (on head and thorax above), inclining to fuscous on on disc of scutellum; face with very pale yellowish hair, cheeks and under side of thorax with white…”, “...antennae long, third joint longer than second, but not nearly twice as long…”, “...tegulae ferruginous; wings strongly yellow, the apical field dusky…”, “...stigma and nervures brown…”, “...abdomen black, hind margins of first two segments obscurely reddened…segments 2 to 5 with conspicuous pure white hair-bands, widely interrupted on second, narrowly on third, and entire on fourth and fifth, but narrowed mesally by a posterior broad emargination…” Similar to his M. martini female description, Cockerell (1928) reports the male on which his description is based to exhibit a broad medial interruption of the distal pale band on the second tergum. Given that Laberge (1956) synonymized this taxon under M. c. communis, it can be assumed that the medial interruption is due to wear (just as the females of M. martini). It is of interest that M. manni is explained to have a complete distal pale band of the second tergum that becomes produced upwards medially, a typical M. c. alopex character rather than M. c. communis, but it becomes evident as to the reason of this specimens’ synonymization to the latter after examination of the apical tergal margins. The metasoma, as can be seen above, is noted to be largely black in regards to integument, the only noteworthy modification to this statement being “...hind margins of first two segments obscurely reddened…” While the width of the distal pale band of the second tergum seems to hold significant authority for subspecific delimitation (which is unfortunately not expounded upon in Cockerell’s description), the apical margins of the terga, in this case, are what allow Cockerell’s M. manni specimen to be identified to M. c. communis. In Laberge's (1956) M. c. alopex description, he notes that males of this subspecies always have widely hyaline apical rims, whereas in his M. c. communis description, he states that the apical tergal rims are often opaque reddish-brown to black; accounting for known rare variations by appending that sometimes, these can also be hyaline. From the comparison of these two traits, it can be determined that, while Cockerell’s M. manni male bears a complete white pubescent band on the fifth tergum, the integument of the terga undoubtedly assigns this specimen to M. c. communis. With the presence and completion of the fifth tergum’s pale band, along with its Arizonan collection site, it’s reasonable to assume that currently, M. manni would be classified as a light variant of M. c. communis. This holotype specimen resides in New York’s American Museum of Natural History (Laberge, 1956).
The remaining two synonym descriptions by Cockerell for M. xanthopteralis and M. hortivagans melanotica are unable to be located by the current author and will herein lack their descriptions until they become available.
M. c. communis’ many historical redescriptions seem to be a result of two events: 1) Cresson’s original specimens being collected in Georgia, 2) the distinct intrasubspecific variation of 3 male characters and 5 female characters. In Cockerell’s and Robertson’s male specimens above, the commonest way in which most are separated from Creassons specimens (often using the Georgia collections as reference) are by their larger sizes and yellow wing veins (although, Robertson drew no comparisons whereas Cockerell directly addressed them). However, as can be seen above, M. c. communis specimens from the Gulf States and Atlantic coastal areas are, on average, smaller and darker than those westward or states further inland (Laberge, 1956). This alone likely contributed to the many re-descriptions of M. c. communis given that, from what is currently known, specimens from Georgia (from which many compared) aren’t representative of individuals from across the majority of this subspecies range. Taking this into account, when comparing his males from inland and central United States to those of the small dark Georgia individuals, Cockerell’s (1905a; 1928) and Robertson’s (1905) specimens would exhibit lighter characteristics and be larger in size (which seems to be the case based on his descriptions); the two authors interpreting such differences to be indicative of separate species. Further, Cockerell differentiated many of his lighter and larger male specimens, which at the time were all believed to be separate species from one another, primarily by the band on the fifth tergum (M. manni bearing a distinctly broad band on the fifth tergum, M. hortivagans having a reduced pale band, and M. xanthopteralis lacking this band). Laberge (1956), likely through the examination of male genitalic structures, found that these differences aren’t indicative of separate species but instead separate populations of the same subspecies (see M. c. communis variation records above), thus synonymizing these taxa under M. c. communis. Subsequent females of proposed species, two described by Cockerell (M. hortivagans described only very briefly in variation notes; 1905ab) and one by Robertson (1905), most commonly differed from Creasson’s specimens by the larger size, smaller dark thoracic patch (at least for M. hortivagans), and yellow wings. The same logic applied to the males can be applied to the females as well. Cockerell likely separated M. hortivagans from M. martini by the size of the dorsal dark thoracic patch (the former being smaller as described above), but given that a true description of Robertson’s female M. variabilis was not written and neither author (Cockerell or Robertson) addressed separation of these two proposed species, a definitive answer as to why M. hortivagans and M. martini were believed to be separate from M. variabilis remains unknown. However, in his descriptions, Cockerell specifies that both M. hortivagans and M. martini females have reddened tegulae whereas Robertson lists M. variabilis’ tegulae as testaceous to piceous. Laberge (1956), likely following the synonymization of the males, grouped these females under M. c. communis as well.
While the above descriptions and comparisons address the convoluted taxonomic history of M. c. communis specimens, M. c. alopex is much simpler in this regard. This subspecies was originally described by Cockerell in 1928 as the taxon Melissodes alopex, with only the male sex being accounted for in his publication. Cockerell notes that, in his identification tables, the specimen on which his description is based keys to M. menuachus, a strange occurrence given that later in his description he erroneously states “...hind margins of abdominal segments not at all hyaline…” as pointed out by Laberge (1956), which from Cockerell’s perspective, would be quite distinct from the hyaline margins of M. menuachus. In Laberge’s (1956) revision, he re-described many characters from the male M. alopex holotype which were incorrectly interpreted in Cockerell’s original description (e.g. the “not at all hyaline” tergal rims which are actually broadly hyaline). These redescriptions will herein accompany (and overwrite portions of) the latter during its description below in an attempt to reconstruct a true interpretation of Cockerell’s specimen and understand the reasoning as to why the male was originally considered as a separate species. M. alopex is described by Cockerell (1928) as having “...second segment densely hairy at base, and with a median fulvous band; third to fifth with exposed parts densely covered with fulvous hair, except broad apical margin of third, and narrow brownish margin of fourth…”, “...eyes darker green (than M. menuachus)...”, and by Laberge (1956) as having “...rather dark ochraceous hairs on the thorax and yellowish-ochraceous hairs on the tibiae and tarsi…”, “...not strongly blackish (wings), although they are somewhat infumate, but are best described as yellowish-brown…”, and “...hind margins of the terga are broadly hyaline…” Laberge (1956) notes that Cockerell likely interpreted the tergal rims as opaque rather than distinctly hyaline due to the stretched out metasoma and wetted or greased tergal surfaces; allowing the opaque black color of the basal areas of following terga to be seen through the transparent portions of the preceding tergal rims. From the mix of these descriptions, it can be seen why Cockerell classified this male specimen as a separated species: 1) the male was collected in Arizona (similar to M. manni, but the latter has much lighter vestiture); 2) it was believed that this specimen had opaque tergal rims; 3) the general appearance was noted to bear similarity to M. menuachus, differing in the characters listed above. It is fairly strange that Cockerell did not make a comparison to M. communis or any other of his current synonyms, especially given his interpretation of the tergal rims, instead stating that the male strongly resembles that of M. sabinesis (currently Epimelissodes sabinensis). Unfortunately, all that can be assumed as to why Cockerell assumed this male to be a separated species is based on the wider and more “fulvous” bands of the terga. Laberge (1956), likely by examination of male genitalic structures, determined M. alopex falls into the species M. communis. However, from what is apparent in both Cockerell’s and Laberge’s descriptions above, the male holotype doesn’t entirely resemble those of M. c. communis, especially due to the color of the tergal bands, the hyaline tergal rims, and the width of the band on the second tergum. From this rose the second subspecies of Melissodes communis, M. c. alopex. The male M. alopex holotype is cared for by the California Academy of Sciences (Laberge, 1956) and likely resides in their collection.
Phylogeny
Melissodes communis has long been recognized to resemble those of M. comptiodes in the East (Cockerell, 1924; Robertson, 1905; Laberge, 1956), M. gilensis in the West (Cockerell, 1905; Laberge, 1956), and sometimes M. colliciatus in the South (Laberge, 1956). However, a study of the molecular phylogeny of the genus Melissodes has produced some interesting, and at times counter-intuative, results. The subgenus in which M. communis resides, M. (Melissodes), was originally presumed by Laberge (1956, 1961) to have evolved from an Ecplectica-like ancestor (as was assumed all subgenera has) with M. (Ecplectica) and M. (Melissodes) forming a tight basal clade to the remaining subgenenera. Furthermore, the diagrammatic representation illustrated by Laberge (1961) depicted the remaining subgenera (apart from Callimelissodes) to form a tight clade from which the basal taxon of this clade was unknown. In 2020, Wright et al., using molecular data, reconstructed a phylogenetic tree which, instead, places M. (Apomelissodes) basal to all subgenera and the clade created by M. (Melissodes) and M. (Ecplectica) directly beneath it. This genetic-backed reconstruction is quite significant not only to the morphological evolution of Melissodes, but it also sheds light on how the dietary differences among subgenera have evolved. As outlined in the bionomics section, M. communis is highly polylectic, similar to many species of its subgenus, however, the basal clade of M. (Apomelissodes) from which M. (Melissodes) diverge consists primarily of specialists. According to Wright (2018), the primitive attribute of the genus is likely that of generalism, therefore, at one time, all Melissodes likely had a similar dietary pattern to M. communis and its allied subgeneric species. The feasibility of the inverse, that all Melissodes are derived from a specialist, seem unlikely and therefore, all references will herein be referring to the former theory. This in turn is somewhat suggestive of a primitive nature among the species of M. (Melissodes), an interesting insight given that, using morphology, this conclusion was also come to by Laberge (1961). In Wright et al’s (2020) phylogenetic tree, M. communis sits in an offshoot within the largest clade in its subgenus and is most closely related to M. gilensis. It is interesting that the remaining similar taxa, M. comptoides and M. colliciatus, are not distinctly related to M. communis as is M. gilensis; the former having a close relation to M. thelypodii and the latter most closely related to M. morrilli.
Similar Taxa
From what can be seen in the phenology section above, M. communis is most closely related to M. gilensis, but, at times, also bears resemblance to
specimens of M. comptoides and M. colliciatus (to a higher extent in the former than the latter). Although M. communis’ description has already been
treated above, a subsequent comparison of diagnostic differences among the foregoing species may shed light on some important differences. The distribution
of M. communis, M. gilensis, M. colliciatus, and M. comptoides overlap in Arizona, New Mexico, and Texas; the paucity of these states being a direct result
of M. collicaitus’ Central American range and M. comptoides’ Eastern United States range. Therefore, pairwise lists of known overlapping ranges among the
three species will be given below along with the citations of the contributing datasets (see “Citation block”). The ranges of M. communis and M. gilensis
overlap in a range that can be drawn from southern Nevada, Arizona, and New Mexico, north to Colorado, east to Kansas, and south to southern Texas; much of
Mexico is likely inhabited by both species, but the lack of M. communis data from this region makes a definitive statement unjustifiable. The ranges of M.
communis and M. colliciatus overlap in Arizona, New Mexico, Texas, Chiapas, and likely much of Mexico; as stated above, a definitive statement cannot be
made due to the lack of M. communis data in this region. The areas in which the ranges of M. communis and M. comptoides overlap are far too great to list
in full, therefore, an outline inclusive range will be given. M. communis and M. comptoides can both be found in a range that can be drawn from South
Dakota, Southeast to Arizona, West along the border of the United States through the Gulf States to the tip of Florida, and North to Pennsylvania. These
species share a strikingly similar phylogeny and flower generalization (too few records exist of M. colliciatus to be certain, although from what has been
reported, this species seems to be polytypic similar to the foregoing taxa) with peak activity occurring around August for most species (however, M.
gilensis’ peak occurs during July through August; see citation block below for datasets). For these reasons, comparisons focusing purely on diagnostic
traits will be given below. All descriptions and comparisons are based on the work of Laberge (1956).
Citation block: Data derived from (Eardley & Ranwashe, 2017; Ikerd, 2019; Johnson, 2019; Johnson, 2020; Best et al., 2022;
Archbold Biological Station, 2023; Carril et al., 2023; Ikerd & Engler, 2023; Purdue Entomological Research Collection, 2023; South
Dakota State University, Severin-McDaniel Insect Research Collection, 2023; Texas A&M University Insect Collection, 2023; Vandame et
al., 2023; VanDyk et al., 2023; North Carolina State University Insect Collection, 2024; Veit & McFarland, 2024; Droege & Maffei, 2025;
European Bioinformatics Institute (EMBL-EBI), GBIF Helpdesk, 2025; Holm et al., 2025; Hughes et al., 2025; Luis & Comisión, 2025; Pozo &
Comisión, 2025; Quiroz & Comisión, 2025; Romero & Comisión, 2025; Scott, 2025; Sánchez & Comisión, 2025; Vergara & Comisión, 2025; Zhuang,
2025; A.J. Cook Arthropod Research Collection, 2026; Bentley & Osborn, 2026; Best, 2026; Cheadle Center for Biodiversity and Ecological
Restoration, 2026a; Cheadle Center for Biodiversity and Ecological Restoration, 2026b; Clemson University Arthropod Collection, 2026;
Colorado State University, C.P. Gillette Museum of Arthropod Diversity, 2026; Dombroskie & Whisenant, 2026; Florida State Collection of
Arthropods, 2026; Frost Entomological Museum, 2026; Gibbs, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey,
2026; István, 2026; Johnson, 2026; Kelly et al., 2026; Kenneth S. Norris Center for Natural History, 2026; Luther College, 2026; Mertz et al.,
2026; Mississippi Entomological Museum, 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern
Biology, 2026; Natural Science Research Laboratory, Museum of Texas Tech University (TTU), 2026; New Mexico State Collection of Arthropods,
2026; Northern Arizona University, 2026; Orrell, 2026a; Orrell, 2026b; San Diego Natural History Museum, 2026; The International Barcode of Life
Consortium, 2026; UMMZ & LSA, 2026; University of Arizona Insect Collection, 2026a; University of Arizona Insect Collection, 2026b; University of
Kentucky, 2026; University of Minnesota Insect Collection, 2026; University of Nebraska State Museum, 2026; Virginia Tech Insect Collection, 2026;
Wild, 2026; Soon, n.d.). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0.
Female
Females of M. communis vary far too much to warrant individual morph comparisons (unlike those of M. bimatris). Therefore, this comparison will focus on the three main groups listed in the female description while addressing the variability and how this may affect similarities among species. The taxon most often confused with M. communis (likely due to its abundance and similarly large variability), is M. comptoides. As shown above, M. communis is found across the majority of the United States and Mexico, whereas M. comptoides resides only in the Eastern U. S. While separation of former from the latter is quite simple in the Western United States (and likely parts of Mexico) due to the latter lacking a presence, the areas in which they overlap can lead to some confusion during identification. Females of M. communis in the first group (all dark thoracic hairs, the darkest group) are quite easily separated from M. comptiodes by the dark vestiture; females of the latter always bearing a few light hairs minimally on the pronotal lobes and often the majority of these hairs are light. However, it should be of note that, as stated in M. c. communis’ variation records, this morph is only represented by one female found in Kansas and is likely a fairly rare occurrence. The second group of females (the mesepisterna have dark ventral hairs, intermediate group), can be separated from those of M. comptoides by the dark thoracic patch, of which is usually absent in M. comptoides (although, specimens of the former are known to bear dark patches, therefore, this should be a supporting characteristic; Fig. 33); and the mesoscutum, which is less coarsely punctate than that of M. comptoides (Fig. 34). Furthermore, M. comptoides often bear punctures on the apical areas of the second and third terga that are usually three to four times the width of the hairs that arise from them, whereas M. communis have these areas either impunctate or bear minute punctures that are less than two times the width of the hairs that arise from them (Fig. 35). The final group of females (no dark mesepisternal hairs, the palest group) can be separated in a similar manner to the intermediate individuals above. Certain females of M. communis, more specifically reddened Mexican M. c. communis, can strongly resemble those of M. colliciatus in overlapping ranges. Given that most morphs of M. communis bear little resemblance to this species (unlike M. comptoides), a comparison only between rufescent females and M. colliciatus will be given. M. c. communis can be readily identified from M. colliciatus by the complete, narrow distal pale band of the second tergum (see Fig. 13) and the posteromedial mesoscutal area bearing sparse punctures; punctures separated by distinctly more than one puncture diameter to near-impunctate (Fig. 36). For contrast, M. colliciatus has a somewhat medially narrowed to sometimes medially interrupted band that is often much wider than M. communis’ and the punctures on the posteromedial mesoscutal surface are often less distinct as well as very dense; punctures separated by approximately one puncture diameter. Aditionally, M. colliciatus often lacks dark dorsal thoracic patches on the mesoscutum and scutellum (or if bearing dark patches, then these patches often small and dark brown), whereas M. c. communis almost always bears a distinctly large dark patch (Fig. 37) M. gilensis, as can be seen in the phylogenetics section, is likely one of the most similar Melissodes to M. communis, specifically those of M. c. alopex. Given that it’s known that the only characteristic in which M. c. alopex varies widely is in size (the majority of the above variation in M. comptoides’ comparison being a result of M. c. communis), a single comparison will be presented. M. communis can be separated from those of M. gilensis by the pale hairs on the pronotal lobes and anterior tegular halves (Fig. 38), and by the pale ochraceous tergal hairs being a similar color to, or paler than, the pale thoracic hairs. For contrast, M. gilensis often bears at least one and often many long dark hairs on the pronotal lobes, often bears appressed brown hairs on the anterior tegular areas, and the pale bands (most distinctly the third and fourth) of the terga have a distinct yellow hue, appearing yellower than the pale hairs of the thorax.

Fig. 33. A comparison of the dorsal thoracic colors between females of M. communis and M. comptoides, demonstrating the presence of a dark patch on M. communis and often the absence of one on M. comptoides (although not always, see text above). Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 34. A comparison of the mesoscutal punctures of females of M. communis and M. comptoides, demonstrating the coarsely punctate mesoscutum of M. comptoides and the sparser punctation pattern of M. communis. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 35. A comparison of the apical punctures on the second and third tergum of females of M. communis and M. comptoides, demonstrating the small and sonewhat indistinct punctures of M. communis and the large and distinct punctures of M. comptoides. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 36. A comparison of the mesoscutal punctures of females of M. communis and M. colliciatus, demonstrating the coarsely punctate mesoscutum of M. colliciatus and the sparser punctation pattern of M. communis. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 37. A comparison of the dorsal thoracic colors between females of M. communis and M. colliciatus, demonstrating the presence of a dark patch on M. communis and often the absence of one on M. colliciatus (although not always, see text above). Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 38. A comparison of the hairs on the pronotal lobes of females of M. communis and M. gilensis, demonstrating the lack of dark hairs on M. communis and the presence of such hairs on M. gilensis. Photo credits: Christopher Wilson (All Rights Reserved).
Male
Although not as variable as their female counterparts, a few morphs of M. communis males exist: 1) those that have a complete or interrupted band across the fifth tergum; 2) those that lack a band on the fifth tergum; 3) those that have hyaline apical tergal margins; 4) those that have opaque tergal margins. These characteristics, from what can be determined in the subspecies section of the male description above, are of importance to subspecific delimitation. It can be herein seen that much of the variation in male characteristics that are pertinent to identification occur between subspecies, however, intergrades do exist and will be accounted for in the comparisons below. Similar to the females, the most commonly confused species with males of Melissodes communis (specifically M. c. communis) is M. comptoides. Where their ranges overlap, males of M. communis can either have an interrupted or absent band on the fifth tergum (very rarely do they have a complete band), and often opaque reddish tergal rims (see M. c. communis description for more). M. communis can be separated from M. comptoides by the apical areas of the second and third terga, which lack deep distinct coarse punctures (if punctures present, then they are minute and fairly indistinct; Fig. 39); the minimum length of the first flagellar segment, which is often approximately equal to, or more than, one sixth of the maximum length of the second (Fig. 40); and the mesoscutum, which bears sparse, large punctures on the posteromedial surface; punctures separated by more than one puncture diameter (Fig. 41). For contrast, M. comptoides males often have coarse, distinct punctures (minimally so near the posterior areas of the distal pale bands); the minimum length of the first flagellar segment is always less than one-sixth of the second segment’s maximum length (often less than one-seventh); and the posteromedial mesoscutal are punctures small and mostly separated by one puncture diameter (sometimes less). M. comtoides males also never bear a complete distal band on the fifth tergum (these hairs often black), whereas M. communis males (although only M. c. communis males occupy this range) will have this band present on occasion (Fig. 42). The band across the fifth tergum of both species can, however, vary largly; M. communis somtimes bearing no band and M. comptoides, on occasion, bearing pale lateral tufts. M. communis is also fairly similar to M. colliciatus, and although perhaps to a lesser extent than females, can be confused with the latter in overlapping ranges. Males of the former can be separated from those of the latter in three manners, each dependent upon the variation of which any given M. colliciatus or M. communis specimen exhibits: 1) by the distal pale band of the second tergum; 2) by the apical tergal rims; 3) by the distal pale band on the fifth tergum. Males of M. communis always have a complete distal band of pubescence on the second tergum (that is, unless worn; worn specimens can still be identified by the dense punctures denoting the original area of the band) and may have hyaline tergal rims (M. c. alopex always does, and while less common of M. c. communis, hyaline tergal rims are not of extreme rarity). M. colliciatus, however, always has opaque tergal rims and the distal pale band of the second tergum is often very narrowly medially interrupted (although, sometimes extremely indistinctly so). If M. communis has opaque tergal rims (as many will), then M. colliciatus can be separated from the former by the distal band on the fifth tergum, which is often entirely complete (or if interrupted, then only narrowly so) and by the lack of dark mesoscutal hairs. The only males of M. communis that bear a distinctly complete band across the fifth tergum are those of M. c. alopex and these can be separated via the first character. The greatest degree of similarity M. communis possesses to a particular species would likely be those of M. gilensis. Occurring mostly in the Western U.S. regions and Mexico, M. gilensis bears a striking resemblance to M. c. alopex, and given that the wide hyaline tergal rims separate M. c. communis from the latter, the comparison below with revolve around the alopex subspecies. Neither taxa varies to the same extent as the foregoing species and subspecies do, the two reliably separated from one another primarily by the width of the distal band on the fourth tergum, and at times the band on the second as well. M. c. alopex’s fourth tergal band is noticeably narrower than four times the apical area’s width medially while M. gilensis’ is of equivalent width or wider than four times its apical width medially. Additionally, the distal pale band of the second tergum on M. c. alopex is often about one-half of the apical area’s medial width, or faintly more, whereas M. gilensis’ is subequal to the width of the apical area.

Fig. 39. A comparison of the apical punctures on the second and third terga the males of M. communis and M. comptoides, demonstrating the minute/indistinct punctures of M. communis and the large and developed punctures of M. comptoides. Photo credits: Christopher Wilson (All Rights Reserved).
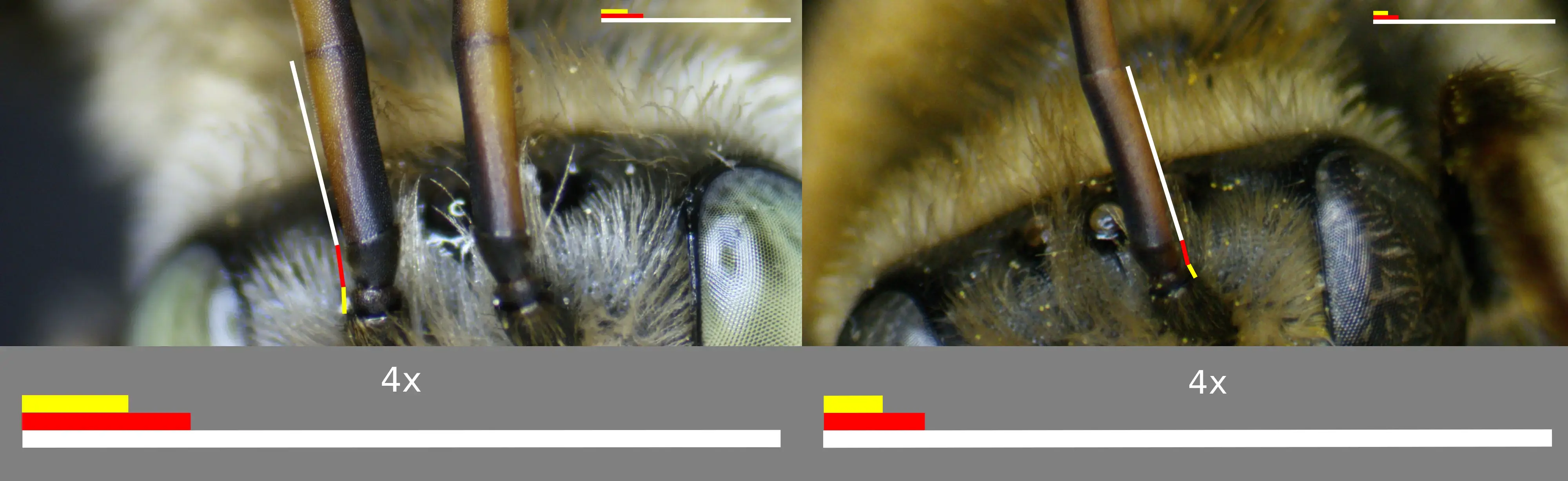
Fig. 40. A comparison of the ratio of F1/F2 of the males M. communis and M. comptoides, demonstrating the larger length of M. communis' F1. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 41. A comparison of the posteromedial mesoscutal punctures of the males M. communis and M. comptoides, showing the crowded punctation pattern of M. comptoides in contrast to the sparser punctures of M. communis. Photo credits: Christopher Wilson (All Rights Reserved).

Fig. 42. A comparison of the distal pale band of the fifth tergum of the males of M. communis and M. comptoides, showing the complete to narrowly interrupted white band of the former and the black band with lateral white tufts of the latter. Photo credits: Christopher Wilson (All Rights Reserved).
References
Wild A (2026). University of Texas, Biodiversity Center, Entomology Collection (UTIC). Version 1.307. University of Texas at Austin, Biodiversity Collections. Occurrence dataset https://doi.org/10.15468/sanyq7 accessed via GBIF.org on 2026-04-02.
Mississippi Entomological Museum (2026). Mississippi Entomological Museum. Occurrence dataset https://doi.org/10.15468/iuhxpw accessed via GBIF.org on 2026-04-02.
MT James Entomological Collection, Washington State University (2026). Washington State University Collection. Occurrence dataset https://doi.org/10.15468/c8yk6t accessed via GBIF.org on 2026-04-02.
Natural Science Research Laboratory, Museum of Texas Tech University (TTU) (2026). Museum at Texas Tech University Invertebrate Zoology Collection. Occurrence dataset https://doi.org/10.15468/ga4bmd accessed via GBIF.org on 2026-04-02.
Dombroskie J, Whisenant J (2026). Cornell University Insect Collection (CUIC). Version 1.6. Cornell University Insect Collection (CUIC). Occurrence dataset https://doi.org/10.15468/w9b6ap accessed via GBIF.org on 2026-04-02.
UMMZ Insects Division Data Group, LSA IT A (2026). University of Michigan Museum of Zoology, Division of Insects. Version 1.108. University of Michigan Museum of Zoology. Occurrence dataset https://doi.org/10.15468/tmxd7n accessed via GBIF.org on 2026-04-02.
Cheadle Center for Biodiversity and Ecological Restoration (2026a). UCSD Holway Lab - Longterm Habitat Fragmentation Study. Occurrence dataset https://doi.org/10.15468/74ktmt accessed via GBIF.org on 2026-04-02.
Droege S, Maffei C (2025). Insect Species Occurrence Data from Multiple Projects Worldwide with Focus on Bees and Wasps in North America. Version 1.27. United States Geological Survey. Sampling event dataset https://doi.org/10.15468/6autvb accessed via GBIF.org on 2026-04-02.
Cheadle Center for Biodiversity and Ecological Restoration (2026b). University of California Santa Barbara Invertebrate Zoology Collection. Occurrence dataset https://doi.org/10.15468/w6hvhv accessed via GBIF.org on 2026-04-02.
The International Barcode of Life Consortium (2026). International Barcode of Life project (iBOL). Occurrence dataset https://doi.org/10.15468/inygc6 accessed via GBIF.org on 2026-04-02.
Luther College (2026). Luther Entomological Research Collection. Occurrence dataset https://doi.org/10.15468/vy5e54 accessed via GBIF.org on 2026-04-02.
Museum of Southwestern Biology (2026). Museum of Southwestern Biology Division of Arthropods. Occurrence dataset https://doi.org/10.15468/jtovgy accessed via GBIF.org on 2026-04-02.
Best L, Engler J, Feuerborn C, Larsen J, Lindh B, Kincaid S, Melathopoulos A, Marshall C J, Marshall C (2022). Oregon Bee Atlas Survey Data: 2019. Version 1.3. Oregon State University. Occurrence dataset https://doi.org/10.15468/kuwm6h accessed via GBIF.org on 2026-04-02.
Veit M, McFarland K (2024). Bee Specimens from Michael Veit Collection. Version 1.3. Vermont Center for Ecostudies. Occurrence dataset https://doi.org/10.15468/b8sg4a accessed via GBIF.org on 2026-04-02.
Best L (2026). Oregon Bee Atlas. Oregon State University. Occurrence dataset https://doi.org/10.15468/fqxchw accessed via GBIF.org on 2026-04-02.
University of Kentucky (2026). University of Kentucky Insect Collection. Occurrence dataset https://doi.org/10.15468/fi4vfu accessed via GBIF.org on 2026-04-02.
University of Nebraska State Museum (2026). University of Nebraska State Museum Entomology. Occurrence dataset https://doi.org/10.15468/6aq9nd accessed via GBIF.org on 2026-04-02.
Scott V (2025). UCM Entomology Collection. Version 6.6. University of Colorado Museum of Natural History. Occurrence dataset https://doi.org/10.15468/jsgtns accessed via GBIF.org on 2026-04-02.
Holm M, Houle M, Salem M (2025). U.S. Fish and Wildlife Service Great Lakes Native Bee Survey. Version 1.14. United States Fish and Wildlife Service. Sampling event dataset https://doi.org/10.15468/jr59af accessed via GBIF.org on 2026-04-02.
Frost Entomological Museum (2026). Frost Entomological Museum. Occurrence dataset https://doi.org/10.15468/epw1ws accessed via GBIF.org on 2026-04-02.
University of Minnesota Insect Collection (2026). University of Minnesota Insect Collection. Occurrence dataset https://doi.org/10.15468/ahwyqb accessed via GBIF.org on 2026-04-02.
Colorado State University, C.P. Gillette Museum of Arthropod Diversity (2026). C.P. Gillette Museum of Arthropod Diversity. Occurrence dataset https://doi.org/10.15468/k0aett accessed via GBIF.org on 2026-04-02.
Gibbs J (2026). J. B. Wallis / R. E. Roughley Museum of Entomology. Version 1.4. University of Manitoba. Occurrence dataset https://doi.org/10.5886/hgagiy accessed via GBIF.org on 2026-04-02.
Florida State Collection of Arthropods (2026). Florida State Collection of Arthropods. Occurrence dataset https://doi.org/10.15468/25xdz4 accessed via GBIF.org on 2026-04-02.
Illinois Natural History Survey (2026). Illinois Natural History Survey Insect Collection. Occurrence dataset https://doi.org/10.15468/eol0pe accessed via GBIF.org on 2026-04-02.
Johnson N (2019). Cleveland Museum of Natural History. Museum of Biological Diversity, The Ohio State University. Occurrence dataset https://doi.org/10.15468/bmfgag accessed via GBIF.org on 2026-04-02.
Virginia Tech Insect Collection (2026). Virginia Polytechnic Institute and State University Insect Collection. Occurrence dataset https://doi.org/10.15468/m2htqa accessed via GBIF.org on 2026-04-02.
Texas A&M University Insect Collection (2023). Texas A&M University Insect Collection. Occurrence dataset https://doi.org/10.15468/caprqh accessed via GBIF.org on 2026-04-02.
Quiroz Martínez H, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Abejas silvestres (Hymenoptera: Apoidea) de la región Praderas de Tokio-El Potosí - Sierra Mazmorras - Cañón de Iturbide, Coahuila y Nuevo León, México. Version 1.17. Comisión nacional para el conocimiento y uso de la biodiversidad. Occurrence dataset https://doi.org/10.15468/xf37pd accessed via GBIF.org on 2026-04-02.
Zhuang V (2025). UTEP Insects (Arctos). Version 1.99. University of Texas at El Paso Biodiversity Collections. Occurrence dataset https://doi.org/10.15468/cfddho accessed via GBIF.org on 2026-04-02.
New Mexico State Collection of Arthropods (2026). New Mexico State Collection of Arthropods. Occurrence dataset https://doi.org/10.15468/nn3cel accessed via GBIF.org on 2026-04-02.
Archbold Biological Station (2023). Archbold Biological Station Arthropod Collection. Occurrence dataset https://doi.org/10.15468/mwyr3t accessed via GBIF.org on 2026-04-02.
Northern Arizona University (2026). Northern Arizona University - Arthropod Collection. Occurrence dataset https://doi.org/10.15468/du1hci accessed via GBIF.org on 2026-04-02.
Ikerd H (2019). Bee Biology and Systematics Laboratory. USDA-ARS Pollinating Insect-Biology, Management, Systematics Research. Occurrence dataset https://doi.org/10.15468/anyror accessed via GBIF.org on 2026-04-02.
Gross J, Oboyski P (2026). Essig Museum of Entomology. Version 121.435. Berkeley Natural History Museums. Occurrence dataset https://doi.org/10.15468/0saucj accessed via GBIF.org on 2026-04-02.
University of Arizona Insect Collection (2026b). University of Arizona Insect Collection RL Minckley Insect Collection. Occurrence dataset https://doi.org/10.15468/sj3uvh accessed via GBIF.org on 2026-04-02.
Soon V. University of Tartu Natural History Museum and Botanical Garden Zooloogical Collections. University of Tartu, Natural History Museum and Botanical Garden. Occurrence dataset https://doi.org/10.15468/6hfnux accessed via GBIF.org on 2026-04-02.
Pozo de la Tijera M D C, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Fortalecimiento de las colecciones de ECOSUR. Primera fase (Abejas). Version 1.10. Comisión nacional para el conocimiento y uso de la biodiversidad. Occurrence dataset https://doi.org/10.15468/ydeiae accessed via GBIF.org on 2026-04-02.
Ikerd H, Engler J (2023). Bee Fauna of National Wildlife Refuges in the Pacific Northwest, 2010-2016. USDA-ARS Pollinating Insect-Biology, Management, Systematics Research. Occurrence dataset https://doi.org/10.15468/ppjnys accessed via GBIF.org on 2026-04-02.
San Diego Natural History Museum (2026). San Diego Natural History Museum Entomology Department. Occurrence dataset https://doi.org/10.15468/dpyqml accessed via GBIF.org on 2026-04-02.
Carril O, Wilson J, Griswold T, Ikerd H I (2023). Wild bees of Grand Staircase-Escalante National Monument. USDA-ARS Pollinating Insect-Biology, Management, Systematics Research. Occurrence dataset https://doi.org/10.7717/peerj.5867 accessed via GBIF.org on 2026-04-02.
Bentley A, Osborn R (2026). Snow Entomological Museum Collection. University of Kansas Biodiversity Institute. Occurrence dataset https://doi.org/10.15468/fhntpy accessed via GBIF.org on 2026-04-02.
University of Arizona Insect Collection (2026a). University of Arizona Insect Collection. Occurrence dataset https://doi.org/10.15468/hzkbpg accessed via GBIF.org on 2026-04-02.
North Carolina State University Insect Collection (2024). North Carolina State University Insect Collection. Occurrence dataset https://doi.org/10.15468/3ck2nw accessed via GBIF.org on 2026-04-02.
Kelly S, Dal Pos D, Sharanowski B (2026). University of Central Florida Collection of Arthropods (UCFC). University of Central Florida Collection of Arthropods (UCFC). Occurrence dataset https://doi.org/10.15468/kyulwg accessed via GBIF.org on 2026-04-02.
Eardley C, Ranwashe F (2017). ARC-PPRI: Collection of Southern African bees (1800-2007). Version 1.1. South African National Biodiversity Institute. Occurrence dataset https://doi.org/10.15468/itdvb2 accessed via GBIF.org on 2026-04-02.
Purdue Entomological Research Collection (2023). The Purdue Entomological Research Collection. Occurrence dataset https://doi.org/10.15468/hyexfq accessed via GBIF.org on 2026-04-02.
Clemson University Arthropod Collection (2026). Clemson University Arthropod Collection. Occurrence dataset https://doi.org/10.15468/fvigdc accessed via GBIF.org on 2026-04-02.
Kenneth S. Norris Center for Natural History (2026). Kenneth S. Norris Center for Natural History, University of California Santa Cruz, Insect Collection. Occurrence dataset https://doi.org/10.15468/hzkqkp accessed via GBIF.org on 2026-04-02.
Sánchez Cordero Dávila V, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Digitalización y Sistematización de las Colecciones Biológicas Nacionales del Instituto de Biología, UNAM (CNIN). Version 1.7. Comisión nacional para el conocimiento y uso de la biodiversidad. Occurrence dataset https://doi.org/10.15468/gy3jna accessed via GBIF.org on 2026-04-02.
Grinter C, Diaz-Bastin R, Fong J (2026). CAS Entomology Type (TYPE). Version 1.335. California Academy of Sciences. Occurrence dataset https://doi.org/10.15468/gak5hc accessed via GBIF.org on 2026-04-02.
Mertz W, Kung G, Xie W (2026). LACM Entomology Collection. Version 5.34. Natural History Museum of Los Angeles County. Occurrence dataset https://doi.org/10.15468/kc9hyp accessed via GBIF.org on 2026-04-02.
Hughes H, Parys K A, Huntzinger K T, Lankford C, Fortuin C, Davis J B (2025). Bees (Hymenoptera: Anthophila) of seasonal herbaceous wetlands in Mississippi. Version 2.1. Specimen Micropublications. Occurrence dataset https://doi.org/10.5886/wv0tub accessed via GBIF.org on 2026-04-02.
Johnson C (2020). hymenoptera. Version 1.3. American Museum of Natural History. Occurrence dataset https://doi.org/10.15468/mvtuf5 accessed via GBIF.org on 2026-04-02.
A.J. Cook Arthropod Research Collection (2026). The Albert J. Cook Arthropod Research Collection. Occurrence dataset https://doi.org/10.15468/mtagq6 accessed via GBIF.org on 2026-04-02.
Johnson N (2026). Triplehorn Insect Collection, The Ohio State University. Museum of Biological Diversity, The Ohio State University. Occurrence dataset https://doi.org/10.15468/efb17f accessed via GBIF.org on 2026-04-02.
European Bioinformatics Institute (EMBL-EBI), GBIF Helpdesk (2025). INSDC Sequences. Version 1.168. European Nucleotide Archive (EMBL-EBI). Occurrence dataset https://doi.org/10.15468/sbmztx accessed via GBIF.org on 2026-04-02.
South Dakota State University, Severin-McDaniel Insect Research Collection (2023). South Dakota State University, The Severin-McDaniel Insect Research Collection. Occurrence dataset https://doi.org/10.15468/hl5tkx accessed via GBIF.org on 2026-04-02.
Orrell T, Informatics and Data Science Center - Digital Stewardship (2026a). NMNH Extant Specimen Records (USNM, US). Version 1.106. National Museum of Natural History, Smithsonian Institution. Occurrence dataset https://doi.org/10.15468/hnhrg3 accessed via GBIF.org on 2026-04-02.
István M (2026). University of New Hampshire Collection of Insects and Arthropods (UNHC-UNHC). University of New Hamsphire Collection of Insects and other Arthropods. Occurrence dataset https://doi.org/10.15468/n36jmy accessed via GBIF.org on 2026-04-02.
Vandame R, Mérida J, Sagot P, Madrigal González D, Bedolla García B Y, González-Vanegas P A, Cultid-Medina C A, Barrios J M (2023). Potential host plant records recovered from ECOAB wild bee collection, Mexico. Version 1.10. Comisión nacional para el conocimiento y uso de la biodiversidad. Occurrence dataset https://doi.org/10.15468/xdzgbt accessed via GBIF.org on 2026-04-02.
Orrell T, Informatics and Data Science Center, Digital Stewardship Team (2026b). NMNH Material Samples (USNM). Version 1.88. National Museum of Natural History, Smithsonian Institution. Occurrence dataset https://doi.org/10.15468/jb9tdf accessed via GBIF.org on 2026-04-02.
Vergara Briceño C H, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Actualización de la base de datos del proyecto H278 Apoidea (Hymenoptera) del Valle de Zapotitlán de las Salinas, Puebla. Comisión nacional para el conocimiento y uso de la biodiversidad. Occurrence dataset https://doi.org/10.15468/wv3xpz accessed via GBIF.org on 2026-04-02.
Luis Martínez M A, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Computarización de la colección de abejas (Hymenoptera: Apoidea) del Museo de Zoología Alfonso L. Herrera, de la Facultad de Ciencias de la UNAM. Comisión nacional para el conocimiento y uso de la biodiversidad. Occurrence dataset https://doi.org/10.15468/wzfnf2 accessed via GBIF.org on 2026-04-02.
Romero Nápoles J, Comisión nacional para el conocimiento y uso de la biodiversidad C (2025). Catálogo de insectos de la colección del Centro de Entomología. Comisión nacional para el conocimiento y uso de la biodiversidad. Occurrence dataset https://doi.org/10.15468/ksnkfh accessed via GBIF.org on 2026-04-02.
Olson, D. M., Gibbs, J., & Schmidt, J. M.. (2021) Wild Bee Pollinators Foraging in Peanut and Cotton Adjacent to Native Wildflower Strips. Florida Entomologist, 104(3). https://doi.org/10.1653/024.104.0304
Harms, K. E., & Owens, B. E.. (2025) Backyard bees: multi-year fidelity to a sleeping roost site by male Melissodes bimaculatus (Lepeletier) (Hymenoptera: Apidae) and co-roosting by parasitic Triepeolus lunatus (Say) (Hymenoptera: Apidae) in southeastern Louisiana. Journal of the Kansas Entomological Society, 98(1). https://doi.org/10.2317/0022-8567-98.1.30
Silva, W. P., & De Andrade, R. R.. (2022) Male Sleeping Aggregation of Melissodes (Ecplectica) nigroaenea (Smith, 1854) (Hymenoptera, Apidae, Eucerini) in Brazilian Cerrado. Sociobiology, 69(2), e5459. https://doi.org/10.13102/sociobiology.v69i2.5459
Parker, F. D., Tepedino, V. J., & Bohart, G. E. (1981). Notes on the Biology of a Common Sunflower Bee, Melissodes (Eumelissodes) agilis Cresson. Journal of the New York Entomological Society, 89(1), 43–52. http://www.jstor.org/stable/25009235
Wilson, J. S., Young, J. G., & Wilson, L. T.. (2024) The Bee Communities of Young Living Lavender Farm, Mona, Utah, USA. Diversity, 16(2), 119. https://doi.org/10.3390/d16020119
Campbell, J.W., Daniels, J.C. and Ellis, J.D. (2018) ‘Fruit set and single visit stigma pollen deposition by managed bumble bees and wild bees in citrullus lanatus (cucurbitales: Cucurbitaceae)’, Journal of Economic Entomology, 111(2), pp. 989–992. doi:10.1093/jee/toy008.
Parker, F. D. (1981). How Efficient Are Been in Pollinating Sunflowers?. Journal of the Kansas Entomological Society, 54(1), 61–67.
Estravis Barcala, M. C., Palottini, F., & Farina, W. M.. (2019) Honey bee and native solitary bee foraging behavior in a crop with dimorphic parental lines. PLOS ONE, 14(10), e0223865. https://doi.org/10.1371/journal.pone.0223865
E. T. Cresson. (1878). Descriptions of New Species of North American Bees. Proceedings of the Academy of Natural Sciences of Philadelphia, 30, 181–221. http://www.jstor.org/stable/4060400
Cockerell, T. D. A. (1905a). New American bees. Proceedings of the Biological Society of Washington, 18, 177--184. https://www.biodiversitylibrary.org/part/144733
Cockerell, T. D. A. (1928). Some Bees From Utah. Psyche: A Journal of Entomology, 35(4), 232-234. https://doi.org/10.1155/1928/63153
Cockerell, T. D. A. (1924). New anthophorid bees from Arizona and Wyoming. American Museum novitates, 113, 1-3. https://digitallibrary.amnh.org/items/3d8a3f3e-dbf5-4af9-9077-2d04dbda163f
Robertson, C. (1905). Synopsis of Euceridae, Emphoridae and Anthophoridae. Transactions of the American Entomological Society, 31, 365-372. https://www.biodiversitylibrary.org/page/10444493
Cockerell, Theodore D. A. (1905b). LXVIII.—Some Bees of the Genus Melissodes from the Rocky Mountain Region. The Annals and Magazine of Natural History; Zoology, Botany, and Geology, 15, 521--529. https://doi.org/10.1080/03745480509442845
Wright, K. W., Miller, K. B., & Song, H.. (2020) A molecular phylogeny of the long-horned bees in the genus Melissodes Latreille (Hymenoptera: Apidae: Eucerinae). Insect Systematics & Evolution, 52(4), 428-443.
https://doi.org/10.1163/1876312x-bja10015
Wright, Karen W.. "The Evolution of Diet Breadth in Melissodes Bees (Apidae: Eucerini)." (2018). https://digitalrepository.unm.edu/biol_etds/301