Melissodes clarkiae

Scientific Classification
Kingdom
Phylum
Class
Order
Family
Tribe
Genus
Subgenus
Species
Binomial Name
Melissodes clarkiae
Melissodes clarkiae Laberge is a fairly rare species of long-horned bee known to inhabit the pacific regions of the United States (Laberge, 1961; Fig. 3). Like all Melissodes, male M. clarkiae have long antennae, and the females have short antennae in comparison (see "Genus" page for more information). This species resides in the subgenus Callimelissodes Laberge and females, although females are quite distinct from all others in this subgenus in multiple characteristics, not being closely related to any single taxon in particular. Males are somewhat more usual to that of a normal Melissodes, often resembling M. ablusus, M. minusculus, M. plumosus and M. metenuus; although differing in shared characters from the former to and the latter two. Females can be separated from the rest of the genus by the simple scopal hairs, often bearing only one to three branches on either side of each rachis, and the distinctly U-shaped pygidial plate. Males are distinctive in the subgeneric Callimelissodes trait of a convex margin on the fourth sternum (although, not a distinctly large flap); flagellar segments 4-10 (and often 3 and 11 as well) bearing longitudinal depressions; the length of the penultimate segment measuring less than, or equal to, twice its width; the first flagellar segment’s minimum length measuring more than the second segment’s maximum length; and the distinctly non-tesselate, although faintly dull due to delicate reticular shagreening galeae (see “Description and Identification” for more information on both sexes) (Laberge, 1961). The female scopal hairs are of great interest in this species given their simple nature, seemingly a direct morphological adaptation to their dietary preferences and/or oligolecty (Laberge, 1961). Such adaptations have been observed across the genus including the galeal hairs of M. apicatus (Laberge, 1956), the scopae of M. fimbriatus (Laberge, 1956), and possibly the clypeal protrusion of many Apomelissodes (Laberge, 1961). A distinct pattern arises when looking at female Melissodes with simple scopae, specifically M. fimbriata, M. baileyi, and M. clarkiae, two of which are presumed to be Ongraceae oligoleges (M. fimbriatus and M. clarkiae, with M. baileyi having too few collections to draw conclusions; Laberge, 1956, 1961). The current species, however, belongs to a subgenus quite distinct from Apomelissodes in general morphology (Laberge, 1961, 1956) and genetics (Wright et al., 2020), but seemingly shares similar dietary specializations and morphological adaptations to those of the latter (see “Flower records” for more information). With the given observations, especially the fact that Apomelissodes was found to be basal to all other subgenera (Wright et al., 2020), this may lead one to believe that M. clarkiae would be basal to the remaining Callimelissodes, suggestive of a fairly recent shared ancestor. However, M. clarkiae is instead much more terminal in position, suggesting that the scopal characteristic is possibly an effect of parallel evolution rather than inheritance.
Description and Identification
Based on Laberge's (1961) description, Melissodes clarkiae are somewhat small setaceous bees. Females are often 11 millimeters in length and approximately 3.5 millimeters in width (width measured at the widest portion of the metasoma). Males are a bit smaller, being about 8 to 11 millimeters in length and 2 to 2.5 millimeters in width (width measured at the widest portion of the metasoma). The female's first flagellar segment is on average 2.02 times the size of the second flagellar segment (standard deviation 0.038). The males are the opposite where the second flagellar segment is on average 3.46 times the size of the first flagellar segment (standard deviation 0.100). Female wing length is 3.39 millimeters on average (standard deviation 0.197 millimeters), and male wing length is 2.90 millimeters on average (standard deviation 0.253 millimeters). Females have an average of 11.71 hamuli (standard deviation 0.286), while males have an average of 10.60 (standard deviation 0.267).
Female
According to Laberge (1961), the description of female M. clarkiae is as follows. The integument is black, differing at the eyes, which are often gray to bluish-gray; the wing membranes which are colorless hyaline; the wing veins, which are dark brown; the underside of F3-F10, and usually the apical area of F2, which are yellow to reddish; the apical half of the mandibles, which are rufescent; the disitarsi, which are rufescent; and the tibial spurs, which are white to yellow. The clypeus is somewhat shiny but dulled with reticular shagreening (highly so posteriorly), has no noticeable boss or apical medial carina, and has punctures that are shallow, regular, round, somewhat large, and mostly separated by less than half of a puncture’s width (clypeal punctures are smaller than that of M. lupinus). The supraclypeal area is shiny without, or with very delicate, shagreening, and has deep largely separated punctures. The flattened lateral areas of the vertex that reach relatively posterior from, and positioned medially to, the apical portions of the compound eyes are somewhat shiny, but dulled due to shagreening, and have round punctures that are tiny and separated one to two puncture diameters. The ultimate flagellar segment is noticeably longer than it is broad, and appears rounded rather than flattened. When looking in facial view, the eyes distinctly converge near the mandibles and are marginally more than one-third as wide as they are long. The four maxillary palpal segments are in a ratio of approximately 3.2:3.2:3.4:1.0, with the penultimate segment being the largest (overall length is greater than the palpi of M. lupinus). The galeae are somewhat shiny above, but dulled due to minute reticular shagreening, often becoming less shagreened in the apical one-third, and have largely spaced punctures with straight hairs arising from them. The mesoscutum is posteromedially, laterally, and anteriorly shiny and often has a small impuncate area posteromedially. The mesoscutal punctures are deep, round, sparse and largely spaced posteromedially, mostly separated by two puncture diameters minimally and usually more, and laterally and anteriorly, are crowded. The scutellum is somewhat shiny but dulled due to very faint and minute shagreening, and has punctures that are moderately smaller than those of the surrounding mesoscutal area, but typically more crowded. The metanotum is shiny without shagreening dorsomedially and has punctures that are noticeably smaller than the mesoscutal punctures, and are mostly separated by less than, or equal to, one-half of a puncture’s diameter. The lateral areas of the mesepisterna are shiny with faint, or no, shagreeing and have round deep punctures that are mostly separated by half a puncture diameter. The dorsal propodeal surface is reticulorugose apart from the medial area and the rugae in the posterior area of the dorsal surface can partly resemble somewhat indistinct large punctures. The posterior propodeal surface has noticeable round punctures that are separated by one-half to one puncture diameter, apart from the upper impunctate triangle. The lateral propodeal surfaces are matte and dull due to faint minute tessellation and the punctures take on a crowded appearance.
The basal three-fifths of the first tergum have round, shallow, small, crowded punctures that are present both basally and laterally. The apical punctures tend to be generally sparser than those of basal area, and the apical two-fifths of the first tergum are entirely impunctate, but the posterior margin of the punctate area has crowded punctures. The second tergum is dulled due to dense reticular shagreening and the basal area of the second tergum has small round punctures that are mostly separated by more than one, and usually more than two, puncture diameters. The second tergum’s interband zone has shallow irregular punctures that are nearly consistently spaced across the entire tergum (not distinctly sparser or denser in a particular area) mostly separated by one to two puncture diameters. The apical area is entirely impunctate. The third and fourth terga are similar to that of the second, however, the interband zone punctures tend to be more crowded. The pygidial plate resembles a “U” in shape (Fig. 1).
The female setal description is as follows. The head hairs are mostly cinereous apart from the darker vertex hairs which tend to take on a dark brownish hue. The hairs on the thorax are mostly pale cinereous apart from the large dark brown mesoscutal patch of hairs that usually reaches, or almost reaches, anteriorly to the anterior margins of the tegulae, and the large dark brown medial patch of hairs on the scutellum. The first tergum has long basal and lateral hairs that are pale ochraceous. The basal area of the second tergum has long pale ochraceous hairs while the interband zone has suberect comparably simple, short, dark hairs that are dark ochraceous to brown. The distal pale pubescent band of the second tergum is wide, often becomes medially interrupted, arched, and laterally reaches, or almost reaches, the apes of the tergum. The apical area of the second tergum is narrowly glabrous marginally, but anterior to this margin bears suberect to appressed, simple, ochraceous to brown hairs (more than that of M. lupinus). The third tergum, is similar to that of the second, apart from the wider uninterrupted distal band that is noticeably spaced from the apical margin (not reaching the apex), and the interband zone, which bears appressed, sparse, pale pubescence. The pale ochraceous band on the fourth tergum is wide, positioned apically (no pale simple hairs near the medial area of the apical margin). The fifth and sixth terga are covered in pale brown hairs apart from the large lateral tufts, which are cinereous. The sternal hairs are golden brown, but laterally, these hairs become whitish. The hairs on the legs are pale ochraceous to white, differing at the fore basitarsi and usually the middle and hind basitarsi, which are dark brown; the distitarsi, which are usually dark brown; the inner surfaces of the hind basitarsi and hind tibiae, which are reddish-orange to yellow; the inner surfaces of the fore and middle tibiae, which are red to yellow; the inner surfaces of the middle bastitarsi, which are red to yellow; the scopae, which are mostly white, but become paler brown on and around the basitibial plates. The scopal hairs are long and usually consist of only one or two branches on either side of the rachis and the rachis protrudes beyond the branched area (Fig. 2).

Fig. 1. A comparison of the pygidial plate structure of a M. clarkiae (left), and a female M. trindois (right), illustrating the narrow U structure of the pygidial plate of the female M. clarkiae. Photo credit: Christopher Wilson (All Rights Reserved).
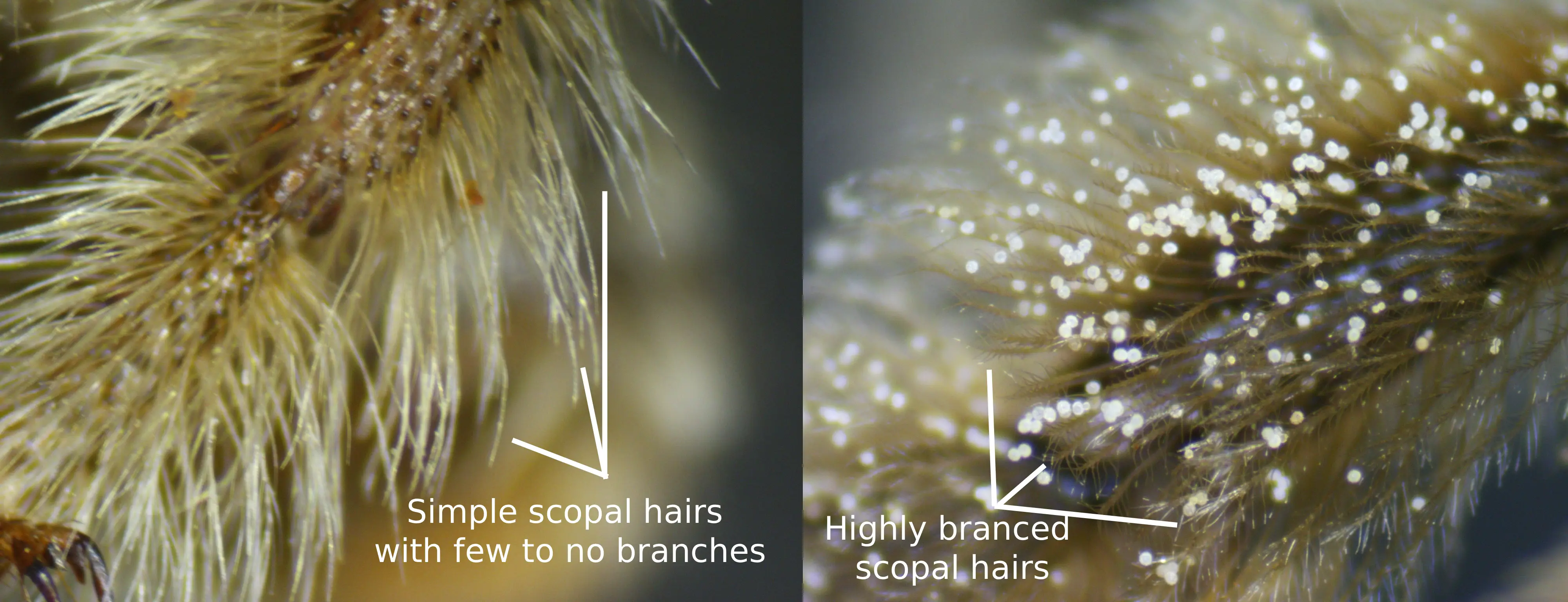
Fig. 2. A comparison of the scopal hairs of a M. clarkiae (left), and a female M. vernoniae (right), illustrating the simple nature of the scopae of the female M. clarkiae. Photo credits: Christopher Wilson (All Rights Reserved).
Male
According to Laberge (1961), the male M. clarkiae description is as follows. The integument is black, differing at the mandibular bases and labrum (the labrum having its margin darkened), which are yellow; the clypeus, which is lemon-yellow and very narrowly darkened posteriorly (most noticeably darkened posterolaterally); the eyes, which are bluish gray to gray, and on rare occasions, greenish blue; the underside flagellar segments 2-11, which are red to yellow (these segments are dark brown on top); the wing membranes, which are hyaline; the wing veins, which are black to brown; terga 2-5, which are sometimes yellow hyaline apically, but usually they are opaque brown; the distitarsi, which are rufescent; and the tibial spurs, which are white to yellow. In facial view, the eyes noticeably converge near the mandibles and, in length, measure to be around slightly longer than 3 times their width (less than that of M. lupinus). The first flagellar segment’s minimum length is more than one-fifth of the second segment’s maximum length (usually one-fourth or longer), and segments 3-10, usually the base of F11 as well, have shiny, shallow, longitudinal dorsolateral depressions. The length of the penultimate segment is equal to, or shorter than, twice its width (usually faintly shorter) and the ultimate segment’s length is more than three times its width. The antennae are shorter than that of M. lupinus, just falling short of the pterostigma in repose. The four maxillary palpal segments decrease in length from basal to apical in a ratio of approximately 5:4:4:1, with the apical segment usually even shorter. The fourth sternum has a wide hyaline flap that is medially incredibly shallowly, or not at all, emarginate, and the fifth and third sterna are noticeably apically convex. The remainder of the sculptural characteristics are similar to that of the females except as follows: the clypeus is reticularly shagreened, and the punctures are round and mostly separated by less than, or equal to, one-half of a puncture’s diameter; the supraclypeal area is often shagreened sparsely; the galeae are shiny above, only finely reticularly shagreened in the basal one-half to two-thirds; and the basal four-fifths of the first tergum is punctate.
The medial plate of the seventh sternum isn’t laterally twisted but rather flat (not facing laterally) and the lateral portions of its apical area create a rounded angle, but before the apex of the rounded angle a distinct narrowing occurs, forming a semi-necked area that widens into a capitate knob. The ventral area of this neck and knob bear short hairs, be it fewer than that of M. plumosus. The membranous area medial to the lateral plate of the seventh sternum and below the medial plate of the same sternum is often smaller than that of M. plumosus. The apical/distal margin of the eighth sternum bears many minute hairs and the ventral tubercle in the medial areas of this same sternum is often unidentate, becoming apically rounded. The gonostyli are laterally glabrous but bear scarce short ventral hairs basally, capitate, and their lengths measure to be less than two thirds of length of the gonocoxite. The length of the spatha is approximately one-third of its width, and this structure is apically sinuate, be it less than that of M. plumosus. The penis valve has a prominent dorsolateral lamella; the basal end of the lamella ends in an inflected tooth near the spatha.
The male setal description is as follows. The head hairs are mostly cinereous, apart from the vertex, which sometimes has brownish hairs, but usually not. The thoracic hairs are also mostly cinereous, differing at the long brown posteromedial mesoscutal hairs, and the scutellar hairs, which also sometimes have brownish hairs, but usually not. The first tergum’s basal fourth-fifths bears long pale ochraceous hairs, and the apical area of this same tergum has suberect, simple, usually pale brown (sometimes ochraceous), short hairs that almost reach the apical margin. The overall vestiture of the second tergum is pale ochraceous to white. The basal pubescent band of the second tergum is white, as is the distal pale band. However, the distal pale band is medially interrupted. The apical area of the second tergum bears simple, suberect, often pale ochraceous (sometimes brown) hairs. The third and fourth terga are similar to that of the second, apart from the distal bands being uninterrupted, the basal tomentum being brown, and the apical hairs being largely brown (minimally medially and sometimes more). The fifth tergum is similar to that of the fourth, except the distal band often reaches the apex across the entire tergum. The sixth and seventh terga are entirely covered in golden-brown to golden-yellow hairs. The sternal hairs are medially darker, becoming pale ochraceous-colored laterally. The hairs on the legs are mostly pale ochraceous to white, apart from the inner surfaces of basitarsi, and usually the inner surfaces of the distitarsi as well, which are yellow to pale rufescent.
Location and Habitat
Melissodes clarkiae is a fairly rare species that has been documented across the pacific coastal regions of the United States south of Washington (Laberge, 1961; see Fig. 3). In
his revision, Laberge (1961) recorded this species to occur only in Oregon and California, and while this holds true based on current-day specimen data, M. clarkiae has been reported
further inland than its original range indicated (Best et al., 2022; Best, 2026). However, with a total of 40 present collections, 30 of which containing coordinates, the full breadth
of M. clarkiae's range may still be unknown. Although the habitats in which M. clarkiae occupy have not been listed in any publications or datasets thus far, distributions of its presumed
host plants, Ongraceae (Laberge, 1961), and the environments in which they inhabit may correspond to those of the former. Within the United States, M. clarkiae’s supposed preferred genera,
Clarkia and Oenothera (Laberge, 1961; however, only collected from the former), are fairly widespread with Clarkia having the majority of its documentations in the pacific coastal states
(see “Clarkia citations” below), a very similar range to that of M. clarkiae. Clarkia (specifically C. xantiana) tends to inhabit “grassland(s), pine-oak woodland(s) and chaparral habitats”
(Gould et al., 2013), while species of Oenothera in pacific coastal regions of the U.S. range from inhabiting sandy environments (Carlson & Meinke, 2008; Cisternas‐Fuentes et al., 2021) to
“weed-lots, garden disposal sites, and occasionally along road sides” (Carlson & Meinke, 2008); these listed habitats are not the extent to which all species of the foregoing genera can be
found. However, it is of note that M. clarkiae likely specializes on Clarkia (hence the species epithet; Laberge, 1961), and therefore probably nests in areas of similar soils to those
preferred by this genus.
Clarkia citations: (Kutner, 2019; Alexander & Gross, 2025; Kennedy, 2026; Magnaghi & Fong, 2026; Orrell & Informatics, 2026; Rancho Santa Ana Botanic Garden, 2026; Santa Barbara Botanic
Garden, 2026; UCLA Herbarium, 2026; University of California Riverside, 2026). Data licensed under CC BY-NC 4.0 and CC BY 4.0.
Fig. 3. Map showing an estimation for the known distribution for M. (Callimelissodes) clarkiae. Each point represents 1 or more occurrences; occurrences that don't have coordinates are not included. Data derived from (Ikerd, 2019; Johnson, 2020; Best et al., 2022; Bentley & Osborn, 2026; Best, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; The International Barcode of Life Consortium, 2026; University of Nebraska State Museum, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0.
Fig. 4. A figure showing the phenological activity of M. clarkiae. The x value is the month, and the y value is the number of documented observations. Data derived from (Ikerd, 2019; Johnson, 2020; Best et al., 2022; Bentley & Osborn, 2026; Best, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; The International Barcode of Life Consortium, 2026; University of Nebraska State Museum, 2026). Data licensed under CC BY 4.0, CC BY-NC 4.0, and CC0 1.0.
Bionomics
Very little can be said in regards to M. clarkiae’s nesting behaviors and ecology. From the available data, M. clarkia seems to range between elevations of 113m (The International Barcode of Life Consortium, 2026; GBIF record 2979632421) to 823m (Johnson, 2020; GBIF record 2804763317), however, only 5 records with a recorded elevation are available. Although some species in the genus, such as M. apicatus, seem to be obligates of a certain soil (sand in M. apicatus’ case; see “Melissodes apicatus” page), it’s unknown whether M. clarkiae is directly dependant upon a specific soil type, or has adjusted to many different soil series and its nesting locations are instead influenced by flora (which are then in-turn influenced by specific climates and soils).
Flower records
Although the data are too sparse to definitively state the floral preferences and/or oligolecty of M. clarkiae, the assumption of Onagraceae oligolecty with a preference on
Clarkia is currently the most accepted for the two following reasons: 1) all but one of the female M. clarkiae specimens examined by Laberge (1961) contained significant amounts of
Clarkia or Clarkia-shaped pollen (pollen in the shape of a tetrahedron) in their scopae, the remaining female containing “cobwebby” pollen (viscin threads) in her scopae with tetrahedral,
presumably Clarkia pollen, mixed; 2) the simple structure of the female’s scopal hairs lend way in carrying pollen tethered by viscin threads (such as plants in the genus Oenothera; Cruden
& Jensen, 1997), or larger triangularly shaped pollen (such as the plants in the genera Clarkia; Halbritter, 2016), but not feasibly carrying the small rounded pollen of Asteraceae (Laberge,
1961). While this is still a current plausible solution, at the time of his publication, Laberge (1961) had seven M. clarkiae females with pollen data attached, all of which carried pollen
from Onagraceae. Since 1961, nine new floral records have been documented (all female), eight of which were collected atop Clarkia (Onagraceae) and of one of which was found atop Lessingia
(Asteraceae). These data, along with Laberge’s (1961), are presented in Figs. 5 & 6. Although this Asteraceae record may be an outlier, an in-depth dietary study of M. clarkiae will need
to occur before definitive statements can be made.
All flower records included in this list are from reports in the literature or datasets. Each flower has a parenthesized reference listed after it, corresponding to the literary work or
dataset in which it was recorded. Clarkia sp. (Laberge, 1961), Clarkia amoena (Best et al., 2022; GBIF record 3467769541), Clarkia speciosa (Laberge, 1961), Lessingia sp. (MT James Entomological
Collection, Washington State University, 2026; GBIF record 5141270599).
Fig. 5. A graph showing the raw current-day known floral data of M. (Callimelissodes) clarkiae from 5 datasets and Laberge's (1961) documentations. Data derived from (Laberge, 1961; Best et al., 2022; Bentley & Osborn, 2026; Best, 2026; MT James Entomological Collection, Washington State University, 2026; The International Barcode of Life Consortium, 2026). Data licensed under CC BY-NC 4.0 and CC BY 4.0. Note that the data are of no substantial value.
Fig. 6. A graph showing unique events of current-day known floral data of M. (Callimelissodes) clarkiae from 5 datasets and Laberge's (1961) documentations. Data derived from (Laberge, 1961; Best et al., 2022; Bentley & Osborn, 2026; Best, 2026; MT James Entomological Collection, Washington State University, 2026; The International Barcode of Life Consortium, 2026). Data licensed under CC BY-NC 4.0 and CC BY 4.0. Note that the data are of no substantial value.
Taxonomy and Phylogeny
Given that M. clarkiae is in Callimelissodes, the newest recognized Melissodes subgenus proposed by Laberge (1961), and was described alongside the subgenic proposal, the taxonomic history of M. clarkiae is quite brief. However, unlike many other species in the genus (such as M. bimaculatus or M. apicatus), M. clarkiae did not undergo a species epithet reclassification with the addition of article 30.1.4.4 in the 1999 release of the International Commission on Zoological Nomenclature (ICZN). This is due to its name being a genitive latinized noun derived from the flower on which this species is presumed to specialize; the name directly translating to ‘Melissodes of the Clarkia’. As stated by ICZN “A species-group name, if a noun in the genitive case formed from a personal name that is Latin, or from a modern personal name that is or has been latinized, is to be formed in accordance with the rules of Latin grammar.” Because the species epithet of M. clarkiae is derived from its host, Clarkia (which is feminine), rather than the actual latinized name (Clarki), the suffix (-ae) instead serves as the genitive singular ending; not the plural feminine ending.
Similar Taxa
Females of Melissodes clarkiae are quite distinctive given the combination of their simple scopae, fairly small and narrow U-shaped pygidial plate, and their clypeus protruding beyond their eyes in profile by less than two-thirds of an eye’s width. Males on the other hand are somewhat similar to M. ablusus and M. minusculus in the flagellar structure, but distinctly resemble M. plumosus and M. metenuus; confusions among M. clarkiae and the latter two being fairly easy (Laberge, 1961). Similarities among these five species arise in their ranges as well, with all five inhabiting California and four inhabiting Oregon (all but M. minusculus), although, M. ablusus, M. plumnosus and M. metenuus have been found much farther Eastward (Ikerd, 2019; Johnson, 2020; Graham et al., 2021; Best et al., 2022; Ikerd & Engler, 2023; Texas A&M University Insect Collection, 2023; Droege & Maffei, 2025; Bentley & Osborn, 2026; Best, 2026; Brigham Young University, Arthropod Collection, 2026; Cheadle Center for Biodiversity and Ecological Restoration, 2026; Gibbs, 2026; Grinter et al., 2026; Gross & Oboyski, 2026; Illinois Natural History Survey, 2026; Mertz et al., 2026; MT James Entomological Collection, Washington State University, 2026; Museum of Southwestern Biology, 2026; Orrell & Informatics, 2026; The International Barcode of Life Consortium, 2026; University of Nebraska State Museum, 2026). While a full species morphological treatment has been presented above, a subsequent comparison of males among these five species may shed light on minute and important differences.
Male
Males of M. clarkiae are similar to the former (M. ablusus & M. minusculus) and latter two (M. plumosus & M. metenuus) species in vastly different characteristics, and as Laberge (1961) noted, this species seems to bear a higher resemblance to the latter two. The reason these two distinct groups of similarity arise is due to flagellar morphology. The former group (M. ablusus, M. minusculus, and M. clarkiae) have similarly structured penultimate segments, but noticeably different microsculpturing and vestiture, whereas the latter group (M. plumosus, M. metenuus, and M. clarkiae) differ in the length of the penultimate flagellar segments, but share similar microsculpturing and fairly similar vestiture. The descriptions and comparisons below are derived from the works and keys of Laberge (1961; 1963). Males of M. clarkiae can be separated from those of M. ablusus and M. minusculus by the fairly shiny to distinctly shiny galeae with no tessellation (if shagreened, then only slightly and delicately so), and by the first flagellar segment’s minimum length measuring more than one-fifth of the second segment’s maximum length. For contrast, M. ablusus and M. minusculus both have very coarse, matte, and dulled galeae due to regular tessellation, and the first flagellar segment’s minimum length is often less than one-fifth of the second segment’s maximum length. Males can be separated from those of M. plumosus and M. metenuus by the length of the penultimate flagellar segment measuring less than, or equal to, twice its width; whereas the two former have longer penultimate segments, their lengths measuring twice their widths.
References
Alexander J, Gross J (2025). The University and Jepson Herbaria -- Vascular Plants. Version 1.10. Berkeley Natural History Museums. Occurrence dataset https://doi.org/10.15468/nmny7h accessed via GBIF.org on 2026-03-18.
Bentley A, Osborn R (2026). Snow Entomological Museum Collection. University of Kansas Biodiversity Institute. Occurrence dataset https://doi.org/10.15468/fhntpy accessed via GBIF.org on 2026-03-18.
Best L (2026). Oregon Bee Atlas. Oregon State University. Occurrence dataset https://doi.org/10.15468/fqxchw accessed via GBIF.org on 2026-03-18.
Best L, Engler J, Feuerborn C, Larsen J, Lindh B, Kincaid S, Melathopoulos A, Marshall C J, Marshall C (2022). Oregon Bee Atlas Survey Data: 2019. Version 1.3. Oregon State University. Occurrence dataset https://doi.org/10.15468/kuwm6h accessed via GBIF.org on 2026-03-18.
Brigham Young University, Arthropod Collection (2026). Brigham Young University Arthropod Museum. Occurrence dataset https://doi.org/10.15468/gqf6no accessed via GBIF.org on 2026-03-19.
Cheadle Center for Biodiversity and Ecological Restoration (2026). UCSD Holway Lab - Longterm Habitat Fragmentation Study. Occurrence dataset https://doi.org/10.15468/74ktmt accessed via GBIF.org on 2026-03-19.
Cruden, R. W., & Jensen, K. G.. (1979) Viscin Threads, Pollination Efficiency and Low Pollen-Ovule Ratios. American Journal of Botany, 66(8), 875. https://doi.org/10.2307/2442226
Droege S, Maffei C (2025). Insect Species Occurrence Data from Multiple Projects Worldwide with Focus on Bees and Wasps in North America. Version 1.27. United States Geological Survey. Sampling event dataset https://doi.org/10.15468/6autvb accessed via GBIF.org on 2026-03-19.
Gibbs J (2026). J. B. Wallis / R. E. Roughley Museum of Entomology. Version 1.4. University of Manitoba. Occurrence dataset https://doi.org/10.5886/hgagiy accessed via GBIF.org on 2026-03-19.
Gould, B., Moeller, D. A., Eckhart, V. M., Tiffin, P., Fabio, E., & Geber, M. A. (2013) Local adaptation and range boundary formation in response to complex environmental gradients across the geographical range of Clarkia xantiana ssp. xantiana. Journal of Ecology, 102(1), 95-107. https://doi.org/10.1111/1365-2745.12188
Graham M, Best L, Moldenke A (2021). Digital record of specimens, including voucher material, from the study of a pollinator habitat restoration site under a commercial solar array in Jackson County, Oregon, 2019.. Oregon State University. Occurrence dataset https://doi.org/10.5399/osu/cat_osac.5.2.4855 accessed via GBIF.org on 2026-03-19.
Grinter C, Diaz-Bastin R, Fong J (2026). CAS Entomology Type (TYPE). Version 1.333. California Academy of Sciences. Occurrence dataset https://doi.org/10.15468/gak5hc accessed via GBIF.org on 2026-03-19.
Halbritter H. 2016. Clarkia unguiculata. In: PalDat - A palynological database. https://www.paldat.org/pub/Clarkia_unguiculata/301507; accessed 2026-03-19
Ikerd H (2019). Bee Biology and Systematics Laboratory. USDA-ARS Pollinating Insect-Biology, Management, Systematics Research. Occurrence dataset https://doi.org/10.15468/anyror accessed via GBIF.org on 2026-03-18.
Ikerd H, Engler J (2023). Bee Fauna of National Wildlife Refuges in the Pacific Northwest, 2010-2016. USDA-ARS Pollinating Insect-Biology, Management, Systematics Research. Occurrence dataset https://doi.org/10.15468/ppjnys accessed via GBIF.org on 2026-03-19.
Illinois Natural History Survey (2026). Illinois Natural History Survey Insect Collection. Occurrence dataset https://doi.org/10.15468/eol0pe accessed via GBIF.org on 2026-03-19.
The International Barcode of Life Consortium (2026). International Barcode of Life project (iBOL). Occurrence dataset https://doi.org/10.15468/inygc6 accessed via GBIF.org on 2026-03-18.
Johnson C (2020). hymenoptera. Version 1.3. American Museum of Natural History. Occurrence dataset https://doi.org/10.15468/mvtuf5 accessed via GBIF.org on 2026-03-19.
Kennedy J (2026). Harvard University Herbaria: All Records. Harvard University Herbaria. Occurrence dataset https://doi.org/10.15468/o3pvnh accessed via GBIF.org on 2026-03-18.
Kutner L (2019). NatureServe Network Species Occurrence Data. Version 8.4. NatureServe. Occurrence dataset https://doi.org/10.15468/lysaex accessed via GBIF.org on 2026-03-18.
LaBerge, W.E. (1961) ‘A revision of the bees of the genus melissodes in north and Central America. part III (hymenoptera, Apidae)’, The University of Kansas science bulletin, 42(5), pp. 283–663. doi:10.5962/bhl.part.9821.
LaBerge, Wallace E. (1963), "New Species and Records of Little-known Species of Melissodes from North America (Hymenoptera:Anthophoridae)". Bulletin of the University of Nebraska State Museum. 9. http://digitalcommons.unl.edu/museumbulletin/9
Magnaghi E, Fong J (2026). CAS Botany (BOT). Version 157.641. California Academy of Sciences. Occurrence dataset https://doi.org/10.15468/7gudyo accessed via GBIF.org on 2026-03-18.
Mertz W, Kung G, Xie W (2026). LACM Entomology Collection. Version 5.33. Natural History Museum of Los Angeles County. Occurrence dataset https://doi.org/10.15468/kc9hyp accessed via GBIF.org on 2026-03-19.
MT James Entomological Collection, Washington State University (2026). Washington State University Collection. Occurrence dataset https://doi.org/10.15468/c8yk6t accessed via GBIF.org on 2026-03-19.
Museum of Southwestern Biology (2026). Museum of Southwestern Biology Division of Arthropods. Occurrence dataset https://doi.org/10.15468/jtovgy accessed via GBIF.org on 2026-03-19.
Orrell T, Informatics and Data Science Center - Digital Stewardship (2026). NMNH Extant Specimen Records (USNM, US). Version 1.106. National Museum of Natural History, Smithsonian Institution. Occurrence dataset https://doi.org/10.15468/hnhrg3 accessed via GBIF.org on 2026-03-19.
Rancho Santa Ana Botanic Garden (2026). RSA - California Botanic Garden Herbarium. Occurrence dataset https://doi.org/10.15468/0yosx9 accessed via GBIF.org on 2026-03-18.
Santa Barbara Botanic Garden (2026). SBBG - Clifton Smith Herbarium, Santa Barbara Botanic Garden. Occurrence dataset https://doi.org/10.15468/adb2bb accessed via GBIF.org on 2026-03-18.
Texas A&M University Insect Collection (2023). Texas A&M University Insect Collection. Occurrence dataset https://doi.org/10.15468/caprqh accessed via GBIF.org on 2026-03-19.
UCLA Herbarium (2026). LA - University of California, Los Angeles Herbarium. Occurrence dataset https://doi.org/10.15468/33k42a accessed via GBIF.org on 2026-03-18.
University of California Riverside (2026). UCR - University of California, Riverside Herbarium - Vascular Plants. Occurrence dataset https://doi.org/10.15468/ai1kou accessed via GBIF.org on 2026-03-18.
University of Nebraska State Museum (2026). University of Nebraska State Museum Entomology. Occurrence dataset https://doi.org/10.15468/6aq9nd accessed via GBIF.org on 2026-03-19.